Abstract
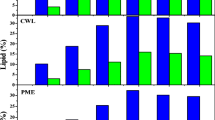
Open-field burning of rice straw contributes significantly to deterioration of air quality. Similarly, defatted algal residue is a surplus by-product of the algal biofuel industry. This study emphasizes on the two-step conversion of agro-industrial residues (rice straw and Scenedesmus obliquus deoiled biomass) into biodiesel and carotenoids using oleaginous red yeast Rhodotorula mucilaginosa R2. The effect of biological pretreatment on degradation of lignocellulosic substrates followed by catalytic saccharification was evaluated using suitable analytical techniques. Mixed biomass favoured the growth and secretion of extracellular cellulose degrading enzymes by Penicillium citrinum PKB20 resulting in the highest specific activity of endoglucanase (83.02 IU/mg), xylanase (103 IU/mg) and β-glucosidase (13.8 IU/mg) under solid-state fermentation. The hydrolysis of pre-treated substrate was achieved with a saccharification yield of 41.41 %. Batch fermentation of detoxified hydrolysate amended with synthetic yeast propagation or lipid production media (YEPD or C/N 75) resulted in maximum lipid content and lipid yield of 30.43 % and 1.19 g/L respectively. GC analysis revealed that oleic acid (29.4-50.39 %) was the predominant fatty acid. The crude carotenoid extract was composed of torularhodin, torulene and β-carotene and exhibited strong antioxidant activity.
Graphic Abstract







Similar content being viewed by others
Data Availability
The data that support the findings of this study are available in published literature. These data were derived from the following resources available in the public domain: Google Scholar, Science Direct, PubMed, Web of Science, Scopus, and so forth.
References
Bhattacharyya, P., Bhaduri, D., Adak, T., Munda, S., Satapathy, B.S., Dash, P.K., Padhy, S.R., Pattanayak, A., Routray, S., Chakraborti, M., Baig, M.J.: Characterization of rice straw from major cultivars for best alternative industrial uses to cutoff the menace of straw burning. Ind Crops Prod. 143, 111919 (2020). https://doi.org/10.1016/j.indcrop.2019.111919
Katiyar, R., Bharti, R.K., Gurjar, B.R., Kumar, A., Biswas, S., Pruthi, V.: Utilization of de-oiled algal biomass for enhancing vehicular quality biodiesel production from Chlorella sp. in mixotrophic cultivation systems. Renew. Energy. 122, 80–88 (2018). https://doi.org/10.1016/j.renene.2018.01.037
Gohain, M., Hasin, M., Eldiehy, K.S., Bardhan, P., Laskar, K., Phukon, H., Mandal, M., Kalita, D., Deka, D.: Bio-ethanol production: a route to sustainability of fuels using bio-based heterogeneous catalyst derived from waste. Process Saf. Environ. Prot. 146, 190–200 (2021). https://doi.org/10.1016/j.psep.2020.08.046
Chan, L.G., Cohen, J.L., de Moura Bell, J.M.L.N.: Conversion of agricultural streams and food-processing by-products to value-added compounds using filamentous fungi. Annu Rev Food Sci Technol. 9, 503–523 (2018). https://doi.org/10.1146/annurev-food-030117-012626
Dowhan, W.: Functional roles of lipids in membranes. In: Roberts, G.C.K. (ed.) Encyclopedia of Biophysics, pp. 868–875. Springer, Berlin (2013). https://doi.org/10.1016/S0167-7306(02)36003-4
Bardhan, P., Gupta, K., Mandal, M.: Microbes as bio-resource for sustainable production of biofuels and other bioenergy products. in: New and Future Developments in Microbial Biotechnology and Bioengineering, first ed., Elsevier, pp. 205e222: (2019). https://doi.org/10.1016/B978-0-444-64191-5.00015-8
Meng, X., Yang, J., Xu, X., Zhang, L., Nie, Q., Xian, M.: Biodiesel production from oleaginous microorganisms. Renew. Energy. 34, 1–5 (2009). https://doi.org/10.1016/j.renene.2008.04.014
Bardhan, P., Gohain, M., Daimary, N., Kishor, S., Chattopadhyay, P., Gupta, K., Chaliha, C., Kalita, E., Deka, D., Mandal, M.: Microbial lipids from cellulolytic oleaginous fungus Penicillium citrinum PKB20 as a potential feedstock for biodiesel production. Ann. Microbiol. 69, 1135–1146 (2019). https://doi.org/10.1007/s13213-019-01494-3
Wei, H., Wang, W., Yarbrough, J.M., Baker, J.O., Laurens, L., Van Wychen, S., Chen, X., Taylor, I.I., Xu, L.E., Himmel, Q., M. E., and Zhang, M.: Genomic, proteomic, and biochemical analyses of oleaginous Mucor circinelloides: evaluating its capability in utilizing cellulolytic substrates for lipid production. PLoS One. 8, 71068 (2013). https://doi.org/10.1371/journal.pone.0071068
Khot, M., Kamat, S., Zinjarde, S., Pant, A., Chopade, B., RaviKumar, A.: Single cell oil of oleaginous fungi from the tropical mangrove wetlands as a potential feedstock for biodiesel. Microb Cell Fact. 11, 71 (2012). https://doi.org/10.1186/1475-2859-11-71
Chreptowicz, K., Mierzejewska, J., Tkáčová, J., Młynek, M., Čertik, M.: Carotenoid-producing yeasts: identification and characteristics of environmental isolates with a valuable extracellular enzymatic activity. Microorganisms. 7, 653 (2019). https://doi.org/10.3390/microorganisms7120653
Dehghani, M., Karimi, K., Sadeghi, M.: Pretreatment of rice straw for the improvement of biogas production. Energ Fuel. 29, 3770–3775 (2015). https://doi.org/10.1021/acs.energyfuels.5b00718
Kainthola, J., Kalamdhad, A.S., Goud, V.V., Goel, R.: Fungal pretreatment and associated kinetics of rice straw hydrolysis to accelerate methane yield from anaerobic digestion. Bioresour. Technol. 286, 121368 (2019). https://doi.org/10.1016/j.biortech.2019.121368
Cantero, D., Jara, R., Navarrete, A., Pelaz, L., Queiroz, J., Rodríguez-Rojo, S., Cocero, M.J.: Pretreatment processes of biomass for biorefineries: current status and prospects. Annu Rev Chem Biomol Eng. 10, 289–310 (2019). https://doi.org/10.1146/annurev-chembioeng-060718-030354
Vaishnav, N., Singh, A., Adsul, M., Dixit, P., Sandhu, S.K., Mathur, A., Puri, S.K., Singhania, R.R.: Penicillium: the next emerging champion for cellulase production. Bioresour Technol Rep. 2, 131–140 (2018). https://doi.org/10.1016/j.biteb.2018.04.003
Goswami, M., Meena, S., Navatha, S., Rani, K.P., Pandey, A., Sukumaran, R.K., Prasad, R.B.N., Devi, B.P.: Hydrolysis of biomass using a reusable solid carbon acid catalyst and fermentation of the catalytic hydrolysate to ethanol. Bioresour. Technol. 188, 99–102 (2015). https://doi.org/10.1016/j.biortech.2015.03.012
Mahajan, A., Gupta, P.: Carbon-based solid acids: a review. Environ. Chem. Lett. 18, 299–314 (2020). https://doi.org/10.1007/s10311-019-00940-7
Poontawee, R., Yongmanitchai, W., Limtong, S.: Efficient oleaginous yeasts for lipid production from lignocellulosic sugars and effects of lignocellulose degradation compounds on growth and lipid production. Process Biochem. 53, 44–60 (2017). https://doi.org/10.1016/j.procbio.2016.11.013
Huang, C., Wu, H., Liu, Z.J., Cai, J., Lou, W.Y., Zong, M.H.: Effect of organic acids on the growth and lipid accumulation of oleaginous yeast Trichosporon fermentans. Biotechnol. Biofuels. 5, 1–12 (2012). https://doi.org/10.1186/1754-6834-5-4
Liu, Z., Feist, A.M., Dragone, G., Mussatto, S.I.: Lipid and carotenoid production from wheat straw hydrolysates by different oleaginous yeasts. J. Clean. Prod. 249, 119308 (2020). https://doi.org/10.1016/j.jclepro.2019.119308
Ayadi, I., Belghith, H., Gargouri, A., Guerfali, M.: Utilization of wheat bran acid hydrolysate by Rhodotorula mucilaginosa Y-MG1 for microbial lipid production as feedstock for biodiesel synthesis. BioMed Res. Int. (2019). https://doi.org/10.1155/2019/3213521
Carvalho, A.K.F., Bento, H.B., Reis, C.E., De Castro, H.F.: Sustainable enzymatic approaches in a fungal lipid biorefinery based in sugarcane bagasse hydrolysate as carbon source. Bioresour. Technol. 276, 269–275 (2019). https://doi.org/10.1016/j.biortech.2018.12.118
Park, G.W., Chang, H.N., Jung, K., Seo, C., Kim, Y.C., Choi, J.H., Woo, H.C., Hwang, I.J.: Production of microbial lipid by Cryptococcus curvatus on rice straw hydrolysates. Process Biochem. 56, 147–153 (2017). https://doi.org/10.1016/j.procbio.2017.02.020
Bardhan, P., Gupta, K., Kishor, S., Chattopadhyay, P., Chaliha, C., Kalita, E., Goud, V.V., Mandal, M.: Oleaginous yeasts isolated from traditional fermented foods and beverages of Manipur and Mizoram, India, as a potent source of microbial lipids for biodiesel production. Ann. Microbiol. 70, 1–14 (2020). https://doi.org/10.1186/s13213-020-01562-z
Lowry, O.H., Rosebrough, N.J., Farr, A.L., Randall, R.J.: Protein measurement with the Folin phenol reagent. J. Biol. Chem. 193, 265–275 (1951)
Muthusamy, S., Selvan, S.T., Arunachalam, P., Grasian, I.: Bioconversion and bioethanol production from agro-residues through fermentation process using mangrove-associated actinobacterium Streptomyces olivaceus (MSU3). Biofuels. 10, 167–179 (2019). https://doi.org/10.1080/17597269.2017.1309853
Gohain, M., Bardhan, P., Laskar, K., Sarmah, S., Mandal, M., Bora, U., Kalita, M.C., Goud, V.V., Deka, D.: Rhodotorula mucilaginosa: a source of heterogeneous catalyst for biodiesel production from yeast single cell oil and waste cooking oil. Renew. Energy. 160, 220–230 (2020). https://doi.org/10.1016/j.renene.2020.06.063
Kot, A.M., Błażejak, S., Kieliszek, M., Gientka, I., Piwowarek, K., Brzezińska, R.: Production of lipids and carotenoids by Rhodotorula gracilis ATCC 10788 yeast in a bioreactor using low-cost wastes. Biocatal. Agric. Biotechnol. (2020). https://doi.org/10.1016/j.bcab.2020.101634
Yoo, A.Y., Alnaeeli, M., Park, J.K.: Production control and characterization of antibacterial carotenoids from the yeast Rhodotorula mucilaginosa AY-01. Process Biochem. 51, 463–473 (2016). https://doi.org/10.1016/j.procbio.2016.01.008
Sluiter, A., Hames, B., Ruiz, R., Scarlata, C., Sluiter, J., Templeton, D., Crocker, D.L.: Determination of structural carbohydrates and lignin in biomass. Lab Anal. Procedure. 1617, 1–16 (2008)
Lodha, A., Pawar, S., Rathod, V.: Optimised cellulase production from fungal co-culture of Trichoderma reesei NCIM 1186 and Penicillium citrinum NCIM 768 under solid state fermentation. J. Environ. Chem. Eng. 8, 103958 (2020). https://doi.org/10.1016/j.jece.2020.103958
Kaur, J., Chugh, P., Soni, R., Soni, S.K.: A low-cost approach for the generation of enhanced sugars and ethanol from rice straw using in-house produced cellulase-hemicellulase consortium from A. niger P-19. Bioresour. Technol. Rep. 11, 100469 (2020). https://doi.org/10.1016/j.biteb.2020.100469
Suganuma, S., Nakajima, K., Kitano, M., Yamaguchi, D., Kato, H., Hayashi, S., Hara, M.: Synthesis and acid catalysis of cellulose-derived carbon-based solid acid. Solid State Sci. 12, 1029–1034 (2010). https://doi.org/10.1016/j.solidstatesciences.2010.02.038
Guo, H., Lian, Y., Yan, L., Qi, X., Smith, R.L.: Cellulose-derived superparamagnetic carbonaceous solid acid catalyst for cellulose hydrolysis in an ionic liquid or aqueous reaction system. Green Chem. 15, 2167–2174 (2013). https://doi.org/10.1039/C3GC40433A
Jin, Y., Lai, C., Li, Y., Cheng, X.: Preparation and catalytic performance of biomass-based solid acid catalyst from Pennisetum sinense for cellulose hydrolysis. Int J Biol Macromol. 165, 1149–1155 (2020). https://doi.org/10.1016/j.ijbiomac.2020.09.256
Dong, S.J., Zhang, B.X., Gao, Y.F., Hu, X.M.: An efficient process for pretreatment of lignocelluloses in functional ionic liquids. Int. J. Polym. Sci. (2015), https://doi.org/10.1155/2015/978983
Baruah, J., Deka, R.C., Kalita, E.: Greener production of microcrystalline cellulose (MCC) from Saccharum spontaneum (Kans grass): statistical optimization. Int J Biol Macromol. 154, 672–682 (2020). https://doi.org/10.1016/j.ijbiomac.2020.03.158
Phitsuwan, P., Permsriburasuk, C., Baramee, S., Teeravivattanakit, T., Ratanakhanokchai, K.: Structural analysis of alkaline pretreated rice straw for ethanol production. Int. J. Polym. Sci. (2017). https://doi.org/10.1155/2017/4876969
Li, C., Knierim, B., Manisseri, C., Arora, R., Scheller, H.V., Auer, M., Vogel, K.P., Simmons, B.A., Singh, S.: Comparison of dilute acid and ionic liquid pretreatment of switchgrass: biomass recalcitrance, delignification and enzymatic saccharification. Bioresour. Technol. 101, 4900–4906 (2010). https://doi.org/10.1016/j.biortech.2009.10.066
Alemdar, A., Sain, M.: Isolation and characterization of nanofibers from agricultural residues–wheat straw and soy hulls. Bioresour. Technol. 99, 1664–1671 (2008). https://doi.org/10.1016/j.biortech.2007.04.029
Subhedar, P.B., Gogate, P.R.: Alkaline and ultrasound assisted alkaline pretreatment for intensification of delignification process from sustainable raw-material. Ultrason Sonochem. 21, 216–225 (2014). https://doi.org/10.1016/j.ultsonch.2013.08.001
Phukan, M.M., Chutia, R.S., Konwar, B.K., Kataki, R.: Microalgae Chlorella as a potential bio-energy feedstock. Appl. Energy. 88, 3307–3312 (2011). https://doi.org/10.1016/j.apenergy.2010.11.026
Tsafrakidou, P., Bekatorou, A., Koutinas, A.A., Kordulis, C., Banat, I.M.: Petsi, T., Sotiriou, M.: Αcidogenic fermentation of wheat straw after chemical and microbial pretreatment for biofuel applications. Energy Convers. Manag. 160, 509–517 (2018). https://doi.org/10.1016/j.enconman.2018.01.046
Thota, S.P., Badiya, P.K., Yerram, S., Vadlani, P.V., Pandey, M., Golakoti, N.R., Belliraj, S.K.: Dandamudi, R.B., Ramamurthy, S.S.: Macro-micro fungal cultures synergy for innovative cellulase enzymes production and biomass structural analyses. Renew. Energy. 103, 766–773 (2017). https://doi.org/10.1016/j.renene.2016.11.010
Osório, E., Gomes, M.D.L., I, Vilela, A.C., Kalkreuth, W., de Almeida, M.A.A., Borrego, A.G., Alvarez, D.: Evaluation of petrology and reactivity of coal blends for use in pulverized coal injection (PCI). Int. J. Coal Geol. 68, 14–29 (2006). https://doi.org/10.1016/j.coal.2005.11.007
Zheng, Q., Zhou, T., Wang, Y., Cao, X., Wu, S., Zhao, M., Wang, H., Xu, M., Zheng, B., Zheng, J., Guan, X.: Pretreatment of wheat straw leads to structural changes and improved enzymatic hydrolysis. Sci. Rep. 8, 1–9 (2018). https://doi.org/10.1038/s41598-018-19517-5
Forough, N., Dzulkefly, K.A., Norhafizah, A., Nazila, M., Zamiri, R.: Biological pretreatment of rubberwood with Ceriporiopsis subvermispora for enzymatic hydrolysis and bioethanol production. BioMed Res. Int. (2013). https://doi.org/10.1155/2013/268349
Poontawee, R., Yongmanitchai, W., Limtong, S.: Lipid production from a mixture of sugarcane top hydrolysate and biodiesel-derived crude glycerol by the oleaginous red yeast, Rhodosporidiobolus fluvialis. Process Biochem. 66, 150–161 (2018). https://doi.org/10.1016/j.procbio.2017.11.020
Zhang, K., Wells, P., Liang, Y., Love, J., Parker, D.A., Botella, C.: Effect of diluted hydrolysate as yeast propagation medium on ethanol production. Bioresour. Technol. 271, 1–8 (2019). https://doi.org/10.1016/j.biortech.2018.09.080
Sharma, R., Ghoshal, G.: Optimization of carotenoids production by Rhodotorula mucilaginosa (MTCC-1403) using agro-industrial waste in bioreactor: A statistical approach. Biotechnol. Rep. (2020). https://doi.org/10.1016/j.btre.2019.e00407
Vyas, S., Chhabra, M.: Assessing oil accumulation in the oleaginous yeast Cystobasidium oligophagum JRC1 using dairy waste cheese whey as a substrate. 3 Biotech. 9, 173 (2019). https://doi.org/10.1007/s13205-019-1701-0
Beopoulos, A., Cescut, J., Haddouche, R., Uribelarrea, J.L., Molina-Jouve, C., Nicaud, J.M.: Yarrowia lipolytica as a model for bio-oil production. Prog. Lipid Res. 48, 375–387 (2009). https://doi.org/10.1016/j.plipres.2009.08.005
Siwina, S., Leesing, R.: Bioconversion of durian (Durio zibethinus Murr.) peel hydrolysate into biodiesel by newly isolated oleaginous yeast Rhodotorula mucilaginosa KKUSY14. Renew. Energy. 163, 237–245 (2020). https://doi.org/10.1016/j.renene.2020.08.138
Kumar, K.K., Deeba, F., Negi, Y.S., Gaur, N.A.: Harnessing pongamia shell hydrolysate for triacylglycerol agglomeration by novel oleaginous yeast Rhodotorula pacifica INDKK. Biotechnol. Biofuels. 13, 1–14 (2020). https://doi.org/10.1186/s13068-020-01814-9
Miao, Z., Tian, X., Liang, W., He, Y., Wang, G.: Bioconversion of corncob hydrolysate into microbial lipid by an oleaginous yeast Rhodotorula taiwanensis AM2352 for biodiesel production. Renew Energy. 161, 91–97 (2020). https://doi.org/10.1016/j.renene.2020.07.007
Mata-Gómez, L.C., Montañez, J.C., Méndez-Zavala, A., Aguilar, C.N.: Biotechnological production of carotenoids by yeasts: an overview. Microb. Cell Fact. 13, 12 (2014). https://doi.org/10.1186/1475-2859-13-12
Cheng, Y.T., Yang, C.F.: Using strain Rhodotorula mucilaginosa to produce carotenoids using food wastes. J Taiwan Inst Chem Eng. 61, 270–275 (2016). https://doi.org/10.1016/j.jtice.2015.12.027
Cipolatti, E.P., Remedi, R.D., dos Santos Sá, C., Rodrigues, A.B., Ramos, J.M.G., Burkert, C.A.V., Furlong, E.B., de Medeiros Burkert, J.F.: Use of agroindustrial byproducts as substrate for production of carotenoids with antioxidant potential by wild yeasts. Biocatal. Agric. Biotechnol. 20, 101208 (2019). https://doi.org/10.1016/j.bcab.2019.101208
Braunwald, T., Schwemmlein, L., Graeff-Hönninger, S., French, W.T., Hernandez, R., Holmes, W.E., Claupein, W.: Effect of different C/N ratios on carotenoid and lipid production by Rhodotorula glutinis. Appl. Microbiol. Biotechnol. 97, 6581–6588 (2013). https://doi.org/10.1007/s00253-013-5005-8
Marova, I., Szotkowski, M., Vanek, M., Rapta, M., Byrtusova, D., Mikheichyk, N., Haronikova, A., Certik, M., Shapaval, V.: Utilization of animal fat waste as carbon source by carotenogenic yeasts–a screening study. Eur Biotech. J. 1, 310–318 (2017). https://doi.org/10.24190/ISSN2564-615X/2017/04.08
Moliné, M., Libkind, D., van Broock, M.: Production of torularhodin, torulene, and β-carotene by Rhodotorula yeasts, Microbial carotenoids from fungi. Humana Press, Totowa, pp. 275–283, (2012)https://doi.org/10.1007/978-1-61779-918-1_19
Kaur, P., Ghoshal, G., Jain, A.: Bio-utilization of fruits and vegetables waste to produce β-carotene in solid-state fermentation: characterization and antioxidant activity. Process Biochem. 76, 155–164 (2019). https://doi.org/10.1016/j.procbio.2018.10.007
Saha, N., Samanta, A.K., Chaudhuri, S., Dutta, D.: Characterization and antioxidant potential of a carotenoid from a newly isolated yeast. Food Sci. Biotechnol. 24, 117–124 (2015). https://doi.org/10.1007/s10068-015-0017-z
Moreira, M.D., Melo, M.M., Coimbra, J.M., dos Reis, K.C., Schwan, R.F., Silva, C.F.: Solid coffee waste as alternative to produce carotenoids with antioxidant and antimicrobial activities. Waste Manage. 82, 93–99 (2018). https://doi.org/10.1016/j.wasman.2018.10.017
Pailliè-Jiménez, M.E., Stincone, P., Brandelli, A.: Natural pigments of microbial origin. Sustain. Food Syst, Front (2020). https://doi.org/10.3389/fsufs.2020.590439
Patel, A., Arora, N., Mehtani, J., Pruthi, V., Pruthi, P.A.: Assessment of fuel properties on the basis of fatty acid profiles of oleaginous yeast for potential biodiesel production. Renew Sust Energ Rev. 77, 604–616 (2017). https://doi.org/10.1016/j.rser.2017.04.016
Poontawee, R., Limtong, S.: Feeding strategies of two-stage fed-batch cultivation processes for microbial lipid production from sugarcane top hydrolysate and crude glycerol by the oleaginous red yeast Rhodosporidiobolus fluvialis. Microorganisms. 8, 151 (2020). https://doi.org/10.3390/microorganisms8020151
Thiru, M., Sankh, S., Rangaswamy, V.: Process for biodiesel production from Cryptococcus curvatus. Bioresour. Technol. 102, 10436–10440 (2011). https://doi.org/10.1016/j.biortech.2011.08.102
Knothe, G., Razon, L.F.: Biodiesel fuels. Prog Energ Combust. 58, 36–59 (2017). https://doi.org/10.1016/j.pecs.2016.08.001
Maina, S., Pateraki, C., Kopsahelis, N., Paramithiotis, S., Drosinos, E.H., Papanikolaou, S., Koutinas, A.: Microbial oil production from various carbon sources by newly isolated oleaginous yeasts. Eng. Life Sci. 17, 333–344 (2017). https://doi.org/10.1002/elsc.201500153
Khot, M., Ghosh, D.: Lipids of Rhodotorula mucilaginosa IIPL32 with biodiesel potential: oil yield, fatty acid profile, fuel properties. J. Basic Microbiol. 57, 345–352 (2017). https://doi.org/10.1002/jobm.201600618
Acknowledgements
The authors wish to thank Dr. Tuhin Ghosh for help in TGA. They also thank SAIC-Tezpur University and IIT-Guwahati for assisting in performing analyses.
Funding
The Authors MM and VVG acknowledges Sunrise Project (Ref: NECBH/2019-20/197) under North East Centre for Biological Sciences and Healthcare Engineering (NECBH) Twinning Outreach Programme hosted by Indian Institute of Technology Guwahati (IITG), Guwahati, Assam funded by Department of Biotechnology (DBT), Ministry of Science and Technology, Govt. of India with number BT/COE/34/SP28408/2018 for providing necessary financial support. The author PB expresses gratitude to DBT for providing fellowship through DBT-JRF (DBT/JRF/BET-16/ I/20 l6/AL/110–501 dated June 29, 2016).
Author information
Authors and Affiliations
Contributions
PB: Conceptualization, Data curation, Investigation, Methodology, and Writing - original draft, KSHE: Methodology, Writing - review & editing, ND: Methodology, Writing - review & editing, MG: Writing - review & editing, VVG: Methodology, Supervision, Writing - review & editing, DD: Supervision, MM: Funding acquisition, Project administration, Supervision, review& editing.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Bardhan, P., Eldiehy, K.S., Daimary, N. et al. Structural Characterization of Mixed Rice Straw and Deoiled Algal Cake-Based Substrate as a Potential Bioenergy Feedstock for Microbial Lipids and Carotenoid Production. Waste Biomass Valor 13, 195–212 (2022). https://doi.org/10.1007/s12649-021-01512-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12649-021-01512-z