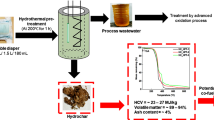
Abstract
The European Union’s circular economy strategy aims to increase the recycling and re-use of products and waste materials. According to the strategy, the use of industry waste materials and side flows is required to be more effective. In this research, a chemical precipitation method to simultaneously remove ammonium and phosphate from the reject water of anaerobic digestion plant using calcined paper mill sludge and fly ash as a precipitant, was tested. Paper mill sludge is a waste material formed in the paper-making process, and fly ash is another waste material formed in the power plant. Objective of this research was to test whether these industrial waste streams could be used as low cost precipitation chemicals for ammonium and phosphate removal from wastewaters and whether the precipitate could be suitable for fertilizer use. Results indicated that calcined paper mill sludge had high removal efficiency for both ammonium (97%) and phosphate (73%). Fly ash also had good removal efficiency for both ammonium nitrogen (74%) and phosphate (59%) at 20 ± 2 °C. The precipitates contained high concentrations of nitrogen and phosphate and could be used as a recycled fertilizer. Other possible mechanisms for the removal of phosphate and ammonium were considered.
Graphic Abstract

Similar content being viewed by others
Avoid common mistakes on your manuscript.
Statement of Novelty
For the first time industrial waste materials fly ash and paper mill sludge were used to precipitate ammonium and phosphate from authentic anaerobic digestion reject water. Results of this paper indicated that both waste materials had good removal efficiency towards ammonium nitrogen and phosphate. Therefore these waste materials could be used as a low cost coagulant and the resulting precipitate could be used as a fertilizer.
Introduction
The European Union’s circular economy strategy aims to increase the recycling and re-use of products and waste materials [1]. According to regulations, the use of industry waste materials and side flows should be more effective. The strategy is closely linked to the EU Waste Framework Directive (2008/98/EC) [2] that includes a five-step waste hierarchy in which different options for dealing with waste are described. The first goal of the waste hierarchy is to prevent formation of waste streams. If this is not possible, the next step is to prepare waste for re-use, followed by recycling and other recovery (e.g., energy recovery). Finally, if none of the first four steps are feasible, waste should be disposed without harming the environment. According to Sokka et al. [3] re-use and symbiosis products manufactured from side flows or waste materials in the local industry carry a small environmental load.
According to European Union’s Wastewater Framework Directive 2000/60/EC, the treatment of wastewater is fundamental to ensuring public health and environmental protection [4]. According to the Council Directive (91/271/EEC), the purpose is to protect the environment from adverse effects of urban waste water discharges and discharges from certain industrial sectors [5]. Municipalities have responsibility to arrange water and sewage services for municipal residents in Finland [6], and there are currently around 1500 water and sewage service providers across the country [7]. Population density is around 18 inhabitants/km2 in Finland and a approximately 80% of Finland’s 5.5 million inhabitants are connected to the sewage system of Finnish waterworks [8]. Due to the previous issues, improving the waterworks in an area of dispersed settlement is one of the key challenges. Finnish law governs environment and waterworks, including industrial wastewaters [9,10,11], which are commonly treated in situ in Finland. However, some industrial wastewaters are led to the municipal wastewater treatment plant. The quality and amount of industrial wastewaters can affect the effectiveness of wastewater treatment plant. In addition to Finnish national legislation, EU’s legislations must also be taken into account when considering industrial wastewaters [4, 12].
Anaerobic digestion is a widely used sustainable method for the management of organic solid wastes and sludge. An anaerobic digestion plant can use various biomass materials for renewable energy production. Agricultural wastes such as straw and grass are suitable and important raw materials. Pig and cow slurry, fur animal manure, stomach and gut content, and potato processing waste (stems and peals) are also useful raw materials in biogas production [13]. The composition of reject water can differ significantly because of the numerous raw materials used in the gasification process [14]. The anaerobic digestate, including reject water, used for agriculture purposes is specifically regulated by both European and Finnish national legislations [15], namely, Regulation (EU) No. 2003/2003 of the European Parliament and of the Council related to fertilizers [16], Animal by-products Regulation (EU) No. 1069/2009 of the European Parliament and of the Council [17], and the Act on Fertilizer Products 539/2006 [18], among others. The separated reject water contains much nitrogen, phosphorus, and potassium. Therefore, the reject water can be used as a fertilizer, but it should be converted into a more slow-release solid form to decrease transport costs and to reduce nutrient leak to waterways.
Phosphorus and nitrogen are the main nutrients in wastewaters that promote the growth of organic matter and algae, which cause eutrophication in water bodies. Discharges of phosphorus and nitrogen are known to occur in, for example, agricultural wastewater and the reject water of anaerobic digestion plant. Agriculture is the largest branch of industry that uses and recycles phosphorus and nitrogen in Finland. The effective recycling of nutrients decreases the use of traditional inorganic fertilizers. Therefore, the side streams and waste materials of the industry should be utilized as effectively as possible [19, 20]. Currently, phosphorus is removed from waste water by collateral precipitation using aluminum or iron precipitants before the biological process, which reduces the potential to use the precipitate, for example, as a fertilizer. Nitrogen is typically present in the form of ammonium. Ammonium ions are not easy to precipitate and are therefore removed in the biological process by nitrification and denitrification [21]. One challenge is the pH, as ammonium volatilizes easily as ammonia gas in alkaline conditions. Ammonium–ammonia equilibrium is strongly pH dependent. One good solution is to convert phosphate and ammonium nitrogen into a product that enhances nutrient value. Chemical precipitation as struvite (MgNH4PO4·6H2O) from wastewater is one method of phosphorus and ammonium nitrogen recovery [22, 23]. Commercial magnesium salts are commonly used for struvite formation, but different types of magnesium-containing by-products have also been tested for that use. Inexpensive magnesium sources, such as magnesite [24], brucite [25], a by-product of marine salt manufacturing and of the thermal treatment of meat waste, bone meal, etc. [26], MgO-saponification wastewater [27], seawater [28, 29], and wood ash from a residential fireplace [30] to produce struvite, have been tested. The precipitation of ammonium nitrogen and phosphate with CaO leads to the formation of a struvite-like calcium mineral according to Frazier et al. [31]. Wajima and Rakovan [32] investigated the removal of phosphate from an aqueous solution using calcined waste paper mill sludge.
Fly ash is fine-grained, inorganic residue that is left behind after combustion at a thermal power plant. It mainly contains Ca, Mg and Si—compounds. Paper mill sludge is a waste material formed in the paper making process. It mainly contains calcium carbonate and cellulose fibres. Currently a waste tax of 85 €/ton has to be paid of these waste streams if they are stored at waste disposal sites (Waste Tax Act 1126/2016) [33]. Due to the high Mg and Ca contents, they could be used for ammonium and phosphate removal from wastewaters. However, neither calcined paper mill sludge nor fly ash has been used to simultaneously precipitate ammonium and phosphate from authentic reject waters previously.
In this research, the suitability of two waste materials (calcined calcium-containing paper mill sludge and calcium- and magnesium-rich fly ash) to be used as low cost chemical precipitants in the simultaneous removal of nitrogen and phosphorous from authentic reject water of the anaerobic digestion plant, were investigated. The effects of temperature and pH on the precipitation efficiency were studied and the nutrient contents of the formed precipitates were analyzed. Moreover, other possible nutrient removal mechanisms and possibility to utilize the precipitate as a fertilizer were considered.
Materials and Methods
Materials
Precipitants
Fly ash originated from a 20 MW thermal power plant located in northern Finland. The power plant uses a bubbling fluidised bed combustion technique. The fuels used at the plant are wood and peat. The fuel ratios (dry mass) were approximately 75% wood (50% wood chips, 10% bark, and 15% recycled wood) and 25% peat. The ash sample was collected directly from the ash silo.
Paper mill sludge was provided by a Finnish pulp and paper mill located in northern Finland. It contains calcium carbonate, which must first be calcined into CaO (Eq. 1).
The calcination process was carried out in the ramp controlled muffle furnace. Calcination temperature was first elevated to 400 °C for 15 min. Subsequently, the temperature was elevated to 1000 °C and was then maintained for 1 h. Thus, carbon and organic materials were burned. The calcination process is necessary because calcium carbonate in itself is not soluble in water and therefore cannot be directly used as a precipitant.
Reject Water from an Anaerobic Digestion Plant
The reject water used in this research was from the anaerobic digestion plant (4 MNm3 biogas/a) located in western Finland. The anaerobic digestion plant uses slurry from the fodder and food industry, manure, fur waste from the leather industry, agricultural waste, and bio-waste as raw materials. Reject water is separated by mechanical treatment from the anaerobic digestion residual after hygienization at 70 °C.
Precipitation Experiments
Saturated precipitant solutions were prepared from fly ash, or calcined paper mill sludge. Before each precipitation experiment, reject water was diluted at 1:3 with distilled water because of its abundant nutrient content. The pH value was adjusted to pH 7.0 by 1 M HNO3, and the temperature of the reject water was adjusted to a regulated temperature of 20 ± 2 °C or 40 ± 2 °C. In the experiments, reject water (500 mL) was stirred with a magnetic stirrer at a speed of 160 rpm, and a pH meter (Knick portable, Berlin, Germany) was connected to the system. Saturated precipitant was added at 1.0 min intervals 3.0 mL at a time up to 90.0 mL. The experiment was continued by adding the precipitant at 2 min intervals 15.0 mL at a time until the total volume of the added precipitant was 150.0 mL. After each addition, the pH value was read. Water samples were taken after the 15.0, 30.0, 45.0, 60.0, 90.0, and 150.0 mL additions. The precipitates were air dried to prevent the volatilization of ammonium.
The removal efficiency (Reff) of ammonium nitrogen and phosphate from the aqueous solution is defined by Eq. 2 as follows:
where C0 and Ct are the concentrations at time o and time t, respectively.
Analytical Methods
The ammonium concentration of the liquid samples was measured using an HACH HQ40d NH4-selective electrode (Model ISENH418101, Loveland, CO, USA). The phosphate concentration of the liquid samples was measured by ion chromatography (METROHM 761 Compact IC, Herisau, Switzerland). The concentrations of trace elements were analyzed using inductively coupled plasma optical emission spectrometry (Perkin Elmer Optima 5300 DV, Shelton, CT, USA), and atomic absorption spectroscopy (Perkin Elmer, AAnalyst 200, Waltham, MA, USA). The composition of the formed precipitates was measured by a field emission scanning electron microscopy with an energy dispersive X-ray spectrometer (FESEM–EDS, Zeiss Sigma, Rödermark, Germany) using a Zeiss Ultra Plus instrument with an Oxford Instruments INCA system EDS software. The main peaks were identified with standard Joint Committee on Powder Diffraction Standards (JCPDS) files. The distribution of the particle size of fly ash was measured using a MALVERN Mastersizer 3000 (Malvern, England) based on the laser diffraction technique. The analyzer utilizes static light scattering and Mie theory to calculate the particle size distribution.
X-ray diffractograms (XRD) were recorded by a PANalytical X´Pert Pro X-ray diffraction equipment using monochromatic CuKα1 radiation (λ = 1.5406 Å) at 45 kV and 40 mA. The diffractograms were collected in the 2θ range of 5°–80° at 0.017° intervals and with a scan step time of 110 s. The crystalline phases and structures were analyzed by the HighScore Plus program. X-ray photoelectron spectroscopy (XPS) analyses were performed using a Thermo Fisher Scientific ESCALAB 250Xi XPS System. The catalyst samples were placed on an indium film. With a pass energy of 20 eV and a spot size of 900 µm, the accuracy of the reported binding energies was ± 0.2 eV. Calcium, magnesium, carbon, nitrogen, phosphorus, and oxygen were measured. The measurement data were analyzed by Avantage v5. The monochromatic AlKα radiation (1486.7 eV) was operated at 20 mA and 15 kV. Charge compensation was used to determine the presented spectra, and the calibration of the binding energies was performed by applying the C1 s at 284.8 eV as reference. Bruker AXS S4 Pioneer (Billerica USA) was used for the X-ray fluorescence spectrometer (XRF) measurement. The XRF was performed at the Center of Microscopy and Nanotechnology, University of Oulu. The sample powders were added with 6% C-wax as a binder and pressed into pellet specimens with a diameter of 37 mm in a steel ring. The detectable element concentration was 5–10 ppm.
Modelling of Precipitation Process
The precipitation processes were modelled using the MINEQL + version 5.0 computer program (Chemical equilibrium modeling system). MINEQL + calculates the solution equilibria to solve the mass balance calculations using thermodynamic constants from the database. For the calculations, the database was extended with an equilibrium constant for struvite, which was logK = − 13.15 for Eq. (3) [30]:
The experimental pH values and the calcium, phosphate, ammonium, and magnesium concentrations were used as initial values in the calculations. The calculations were performed for an open system in normal atmospheric conditions and a temperature of 25 °C. The minerals presented in Table 1 were included in the calculations. The solubility indexes (SI) were calculated for the minerals, and the minerals were considered to be able to precipitate when SI ≥ 0.
Results and Discussion
Chemical Composition and Characterization of the Precipitants
The chemical composition of fly ash and calcined paper mill sludge is presented in Table 2. Fly ash contains 34.6 wt% of CaO and 14.7 wt% of MgO. Calcined paper mill sludge contains 97.3 wt% of CaO.
Figure 1 illustrates the distribution of the particle size of calcined paper mill sludge and fly ash. The particle size distributions of the calcined paper mill powder were Dv (10) 8.7 μm, i.e. 10% of particles are smaller than 8.7 μm (Dv = the volume median diameter), Dv (50) 32.9 μm, and Dv (90) 145 μm. All particles were smaller than 500 μm in the calcined paper mill sludge. The particle size distributions of the fly ash powder were Dv (10) 10.7 μm, Dv (50) 61.1 μm, and Dv (90) 136 μm. All particles were smaller than 200 μm in the fly ash. The particle sizes were distributed at a wider range in calcined paper mill sludge than in fly ash.
In Finland, the limit values for harmful elements in fertilizers are regulated by the Finnish Decree of the Ministry of Agriculture and Forestry on Fertilizer Products (24/2011) [34]. This Decree states the quality requirements of fertilizer products. The harmful element contents of the precipitants and the limit values of the Finnish fertilizer product decree are shown in Table 3. The harmful element contents in the paper mill sludge and fly ash were clearly lower than the Finnish limits for forest fertilizers. The limits concerning forest fertilizer is presented in the parenthesis.
Characterization of the Reject Water
Essential nutrient contents were determined from the reject water of anaerobic digestate, and they are illustrated in Table 4. However, nutrient concentrations can vary widely, depending on the raw materials [35].
Ammonium Removal
Figure 2 illustrates the NH4+ removal at two different temperatures. Calcined paper mill sludge removed 97.0% of ammonium nitrogen from the reject water at 20 ± 2 °C and 97.0% at 40 ± 2 °C. Fly ash sludge removed 73.8% of ammonium nitrogen from the reject water at 20 ± 2 °C and 72.8% at 40 ± 2 °C. The change in temperature had no effect on the removal of NH4+ using either calcined paper mill sludge or fly ash sludge. Therefore, precipitation at room temperature, 20 ± 2 °C, was preferred because no heating was required. The results of ammonium removal using fly ash are comparable with the results of other types of inexpensive magnesium sources, such as magnesium hydroxide based industrial by-product [36] and hydrolysate from hydrothermal process [37].
Phosphate Removal
Figure 3 illustrates the PO43− removal at two different temperatures. Calcined paper mill sludge removed 72.9% of phosphate from the reject water at 20 ± 2 °C and 82.2% at 40 ± 2 °C. The difference was 9.3 pp (percentage point). Fly ash sludge removed 58.6% of phosphate from the reject water at 20 ± 2 °C and 62.1% at 40 ± 2 °C. The difference was only marginal, 3.5 pp. The change in temperature had a larger effect using calcined paper mill sludge than when using fly ash sludge. Others have obtained similar phosphate removal results with fly ash [38, 39], waste bone ash [40], and calcined waste paper mill sludge [32], Moriyama et al. [41] achieved 75–85% efficiency of hydroxyapatite formation using Ca-rich media in phosphorus removal.
Residual Ca2+ and Mg2+ Concentrations
The residual Ca2+ concentrations at two different temperatures are presented in Fig. 4 and the residual Mg2+ concentrations in Fig. 5. With both precipitants, the soluble calcium and magnesium ions participated readily in the precipitation reaction. Thus, the concentration of Ca2+ and Mg 2+ ions remained low as long as ammonium and phosphate ions were available in the solution. Limited amount of ammonium and phosphate ions were available found for the precipitation reaction, and the Ca2+ concentration began to increase after their removal. Thus, the residual concentration of calcium ions was at a high level when calcined paper mill sludge was used as a precipitant. When using fly ash sludge as a precipitant, the Ca2+ residual was clearly lower. Conversely, the residual of magnesium ions was at a low level, indicating that almost all Mg2+ ions participated in the reaction. A small drop in magnesium concentration is observed after the 20 min treatment time possibly because of dolomite precipitation. Initially, only a small amount of magnesium was found in the precipitant in the case of the calcined paper mill sludge. The results were the same at 20 ± 2 °C and at 40 ± 2 °C. Thus, the temperature did not affect the residual calcium and magnesium ion concentrations, consistent with the ammonium and phosphate removal results.
Effect of pH
The pH was adjusted to 7.0 in the beginning of the experiments in order to prevent the increase of pH to a too high level during the experiments. The optimum pH for struvite precipitation is pH 7.0–9.0 as reported previously [42, 43]. The change in pH as a function of time is presented in Fig. 6. The results indicated that the pH increased throughout the treatment time as more precipitant was added to the solution. The largest pH change occurred when the calcined paper mill sludge was used (5.6 pH unit at 20 ± 2 °C and 5.1 pH unit at 40 ± 2 °C), and the smallest pH change occurred when the fly ash sludge was used (2.1 pH unit at 20 ± 2 °C and 2.5 pH unit at 40 ± 2 °C). Ammonium volatilizes easily as ammonia gas at alkaline conditions [44]. When the calcined paper mill sludge was used as the precipitant, the pH increased over 12. In this case, nitrogen probably volatilized instead of precipitated. This result could partly explain the high removal percentage of nitrogen. However, when fly ash was used as the precipitant, the pH value remained at the optimum level throughout the experiments.
Characterization of the Precipitate
The mean composition of the formed precipitates is presented in Table 5. In summarizing the results of FESEM–EDS, oxygen was the main element at all temperatures. The magnesium content was 3.7–5.4% in the fly ash sludge precipitates and below 1% in the calcined paper mill sludge precipitates. The phosphorus content was slightly below 2% in the fly ash sludge precipitates and around 1% in the calcined paper mill sludge precipitates. The potassium content varied between 1.6 and 1.9% in the fly ash sludge precipitates and between 0.7 and 1.1% in the calcined paper mill sludge precipitates. The calcium content varied at 12.4–17.2% in the fly ash sludge precipitates and at 34.4–39.1% in the calcined paper mill sludge precipitates, which were clearly larger. The change in temperature only had a marginal effect on the concentration of the individual elements. The sum of the arithmetic mean is not 100% in Table 5 because of the rounding off of the numerical values.
The FESEM image of untreated fly ash is presented in Fig. 7a, and an image of treated fly ash is illustrated in Fig. 7c. Untreated fly ash mainly contained irregularly shaped particles and thus had no clear morphology. Calcined paper mill sludge contained homogenous calcium oxide Fig. 7b. A fine, white precipitate on the surface of the particles was visible in the treated material Fig. 7c, d. The magnification is 250× at all images.
Figure 8 shows the XRD results for the four samples. The calcined paper mill sludge at 20 ± 2 °C and at 40 ± 2 °C had similar patterns, indicating that the main phases were CaCO3 (JCPDS file 04-007-8659) and Ca(OH)2 (JCPDS file 04-006-9147) and the minor phase was Ca2Al(CO3)0.25 (JCPDS file 04-019-0886). Some amorphous phases were observed at 2 Theta at 30°–40°. The fly ash sludge at 20 ± 2 °C and at 40 ± 2 °C had more small peaks and more amorphous phases. The main peaks showed the phases of CaCO3 (JCPDS file 04-007-8659) and Ca(OH)2 (JCPDS file 04-006-9147), with a minor phase of K0.9Na0.1Mg0.1Fe2.4Al1.2Si2.8O10(OH)0.7F0.3 (Annite-1M JCPDS file 04-011-6697) or FeCO3 (JCPDS file 01-080-2678). No struvite or hydroxyapatite peaks were detected, but they could exist as amorphous in the case of fly ash sludge. As confirmed by the FESEM–EDS analysis results (Table 5), the phosphorus content was quite low, < 2 wt%, and thus the struvite and hydroxyapatite content could have been too low for the XRD device to detect it. In the case of fly ash sludge as a precipitant, the calculations indicated that struvite and hydroxyapatite precipitation was possible for the whole precipitation pH range of 7.0–9.5 and dolomite precipitation for pH 9.5. However, these minerals were not detected with XRD probably because of the low magnesium and phosphorus content measured by SEM–EDS (Table 5).
The modelling did not predict the precipitation of CaCO3. This is explained as, unlike the calcined paper mill sludge precipitant, the fly ash sludge precipitant contained CaCO3, according to an additional XRD analysis. For the calcined paper mill sludge as the precipitant, the calculations predicted the possible precipitation of hydroxylapatite for the pH range of 7.0–11.0, calcite for the pH range of 7.5–12.6, and dolomite for the pH range of 8.0–12.6. However, the magnesium concentration in the calcined paper mill sludge precipitation was small, and thus the amount of dolomite that could precipitate was also small. In Fig. 6, a small drop in magnesium concentration is observed after the 20 min treatment time possibly because of dolomite precipitation. The XRD analysis (Fig. 8) confirmed the precipitation of CaCO3. The amount of hydroxyapatite could have been too low for the XRD detection because of the low phosphorus content (≤ 1.1 wt%) as determined by FESEM–EDS (Table 5).
The XPS characterization of the main nutrient contents of the precipitates is presented in Table 6. As shown in Table 6, the amount of nutrients followed the FESEM–EDS analyzing results (Table 5). The nitrogen content was remarkable in the fly ash sludge and calcined paper mill sludge precipitations. The calcium content was much higher in the calcined paper mill sludge because of the larger initial content of the precipitant. The magnesium content was higher in the fly ash because of the same reason. The carbon content was at a surprisingly high level most likely because of the fact that the reject water still contained some organic matter. Moreover, carbon dioxide could absorb onto the surface of the sample, distorting the result.
The harmful element contents of the precipitates are presented in Table 7. Harmful elements can be found in the reject water from both natural and anthropogenic sources. Particularly, arsenic, cadmium, chromium, mercury, copper, lead, nickel, and zinc are undesirable harmful metals. As shown in Table 7, the heavy metal contents are below the limit values for the forest fertilizers of the Finnish Decree of the Ministry of Agriculture and Forestry on Fertilizer Products in all precipitates.
Adsorption
The removal of nutrients may include some adsorption reactions aside from the precipitation reaction [45, 46]. Phosphate can be adsorbed by calcite [47]. Moreover, substances such as CaO and Al2O3 contained in both calcined paper mill sludge and fly ash sludge enhance the adsorption mechanism instead of the chemical precipitation [48]. Reject water from the anaerobic digestion plant clearly contains more nitrogen than phosphorus (Table 4), thus making phosphorus the limiting element in the formation of struvite. When using calcined paper mill sludge, magnesium is the limiting element in the formation of struvite. However, the XRD results indicated that struvite peaks could not be detected, and this result could partly be caused by the amorphous phase. Nevertheless, the phosphorus content was quite low, and thus the struvite content might have been too low for the XRD device to detect it. The results indicated that both ammonium removal and phosphate removal were effective with both precipitants. Moreover, although some of the nitrogen was volatilized as ammonia gas in the precipitation with calcined paper mill sludge, the XPS results indicated that a high concentration of nitrogen was adsorbed into the precipitates. Thus, both precipitation reactions and adsorption reactions could have occurred in the experiments.
Conclusion
The utilization of industrial waste materials as secondary raw materials is crucial for sustainable new technologies. Objective of the paper was to test whether industrial waste materials (fly ash and calcined paper mill sludge) could be used as low cost precipitation chemicals in the simultaneous removal of ammonium nitrogen and phosphate from the reject water, and whether the precipitate could be suitable for fertilizer use. The results obtained in this study indicated that both waste materials had good removal efficiency towards ammonium nitrogen and phosphate. Calcined calcium-containing paper mill sludge removed 97% of ammonium nitrogen and 73% of phosphate and calcium- and magnesium-rich fly ash sludge removed 74% of ammonium nitrogen and 59% of phosphate from the reject water of anaerobic digestion plant, at 20 ± 2 °C. The results from the study indicate that nutrient-rich reject water from the anaerobic digestion plant can be converted to a solid form containing nitrogen and phosphate. The conversion can make the plant more feasible by lowering the costs for waste treatment. According to the XPS analysis, the precipitates contained high concentrations of nitrogen and phosphate and could be used as recycled fertilizer. Moreover, the harmful element contents of the precipitates were at very low level. The change in temperature had no effect on the removal of ammonium nitrogen and phosphate. However, precipitation reactions and adsorption reactions might have occurred in the experiments. An adsorption reaction could partly explain why the products such as struvite could not be seen by XRD. However, more research is needed to optimize the precipitation conditions, and solubility and growth tests of the precipitates to confirm the suitability for fertilizer use.
References
European Commission 2018a: Circular economy strategy—environment—European Commission. http://ec.europa.eu/environment/circular-economy/index_en.htm (2018). Accessed 11 Apr 2018
European Commission 2018b: Directive 2008/98/EC of the European Parliament and of the Council of 19 November 2008 on waste and repealing certain directives. https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=CELEX%3A32008L0098 (2018). Accessed 11 Apr 2018
Sokka, L., Pakarinen, S., Melanen, M.: Industrial symbiosis contributing to more sustainable energy use—an example from the forest industry in Kymenlaakso, Finland. J. Cleaner Prod. 19, 285–293 (2011). https://doi.org/10.1016/j.jclepro.2009.08.014
European Union’s Wastewater Framework Directive 2000/60/EC. https://www.eea.europa.eu/data-and-maps/indicators/urban-waste-water-treatment/urban-waste-water-treatment-assessment-4 (2000). Accessed 2 Apr 2019
The Council Directive 91/271/EEC. http://ec.europa.eu/environment/water/water-urbanwaste/index_en.html (1991). Accessed 2 Apr 2019
Act on Water Services 119/2001. https://www.finlex.fi/en/laki/kaannokset/2001/en20010119 (2001). Accessed 2 Apr 2019
Kuntaliitto: Kuntaliitto, 2007. Kunnat ja vesihuolto huomisen Suomessa. Helsinki, Finland (2007)
Herrala, M.E., Huotari, H., Haapasalo, H.J.O.: Governance of Finnish waterworks-A DEA comparison of selected models. Util. Policy 20, 64–70 (2012)
Finnish Ministry of the Environment, Environmental Protection Act 527/2014. https://www.finlex.fi/fi/laki/alkup/2014/20140527 (2014). Accessed 2 Apr 2019
Nitrate directive 931/2000. https://www.finlex.fi/fi/laki/alkup/2000/20000931 (2000). Accessed 2 Apr 2019
Act on Waste Water 157/2017. https://www.finlex.fi/fi/laki/alkup/2017/20170157 (2017). Accessed 2 Apr 2019
Regulation (EC) No 166/2006 on the establishment of a European Pollutant Release. https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=celex:32006R0166 (2006). Accessed 2 Apr 2019
Pan-in, S., Sukasem, N.: Methane production potential from anaerobic co-digestions of different animal dungs and sweet corn residuals. Energy Procedia. 138, 943–948 (2017). https://doi.org/10.1016/j.egypro.2017.10.062
Latvala, M.: Biokaasun tuotanto suomalaisessa toimintaympäristössä. Suomen Ympäristö 24/2009. Suomen ympäristökeskus. (2009)
Dahlin, J., Herbes, C., Nelles, M.: Biogas digestate marketing: qualitative insights into the supply side. Resour. Conserv. Recy. 104, 152–161 (2015). https://doi.org/10.1016/j.resconrec.2015.08.013
European Commission 2003: Regulation (EU) No 2003/2003 of the European Parliament and of the Council relating to fertilizers. http://eur-lex.europa.eu/legal-content/FI/TXT/PDF/?uri=CELEX:32003R2003&from=FI (2003). Accessed 11 Apr 2018
European Commission 2009: Animal by-products Regulation (EU) No 1069/2009 of the European Parliament and of the Council. http://eur-lex.europa.eu/legal-content/FI/ALL/?uri=CELEX:32009R1069 (2009). Accessed 11 Apr 2018
Finnish Legislation 2006: The Act on Fertilizer Products 539/2006. https://www.finlex.fi/fi/laki/ajantasa/2006/20060539 (2006). Accessed on 11 Apr 2018
Huttunen, S., Manninen, K., Leskinen, P.: Combining biogas LCA reviews with stakeholder interviews to analyse life cycle impacts at a practical level. J. Cleaner Prod. 80, 5–16 (2014). https://doi.org/10.1016/j.jclepro.2014.05.081
Marttinen, S., Venelampi, O., Iho, A., Koikkalainen, K., Lehtonen, E., Luostarinen, S., Rasa, K., Sarvi, M., Tampio, E., Turtola, E., Ylivainio, K., Grönroos, J., Kauppila, J., Koskiaho, J., Valve, H., Laine-Ylijoki, J., Lantto, R., Oasmaa, A., zu Castell -Rüdenhausen, M.: Luonnonvara-ja biotalouden tutkimus 45/2017. (2017)
Kilpimaa, S., Runtti, H., Kangas, T., Lassi, U., Kuokkanen, T.: Physical activation of carbon residue from biomass gasification: Novel sorbent for the removal of phosphates and nitrates from aqueous solution. J. Ind. Eng. Chem. 21, 1354–1364 (2015)
Kataki, S., West, H., Clarke, M., Baruah, D.C.: Phosphorus recovery as struvite: recent concerns for use of seed, alternative Mg source, nitrogen conservation and fertilizer potential. Resour. Conserv. Recy. 107, 142–156 (2016). https://doi.org/10.1016/j.resconrec.2015.12.009
Yoshino, M., Yao, M., Tsuno, H., Somiya, I.: Removal and recovery of phosphate and ammonium as struvite from supernatant in anaerobic digestion. Water Sci. Technol. 48, 171–178 (2003)
Gunay, A., Karadag, D., Tosun, I., Ozturk, M.: Use of magnesit as a magnesium source for ammonium removal from leachate. J. Hazard. Mater. 156, 619–623 (2008). https://doi.org/10.1016/j.jhazmat.2007.12.067
Huang, H.M., Xiao, X.M., Yang, L.P., Yan, B.: Removal of ammonium from rare-earth wastewater using natural brucite as a magnesium source of struvite precipitation. Water Sci. Technol. 63, 468–474 (2011)
Siciliano, A., De Rosa, S.: Recovery of ammonia in digestates of calf manure through a struvite precipitation process using unconventional reagents. Environ. Technol. 35, 841–850 (2014)
Huang, H., Chen, Y., Jiang, Y., Ding, L.: Treatment of swine wastewater combined with MgO-saponification wastewater by struvite precipitation technology. Chem. Eng. J. 254, 418–425 (2014). https://doi.org/10.1016/j.cej.2014.05.054
Crutchik, D., Sánchez, A., Garrido, J.M.: Simulation and experimental validation of multiple phosphate precipitates in a saline industrial wastewater. Sep. Purif. Technol. 118, 81–88 (2013). https://doi.org/10.1016/j.seppur.2013.06.041
Liu, B., Giannis, A., Zhang, J., Chang, V.W., Wang, J.: Characterization of induced struvite formation from source-separated urine using seawater and brine as magnesium sources. Chemosphere 93, 27–38 (2013). https://doi.org/10.1016/j.chemosphere.2013.09.025
Sakthivel, S.R., Tilley, E., Udert, K.M.: Wood ash as a magnesium source for phosphorus recovery from source-separated urine. Sci. Total Environ. 419, 68–75 (2012). https://doi.org/10.1016/j.scitotenv.2011.12.065
Frazier, A.W., Lehr, J.R., Smith, J.P.: Calcium ammonium orthophosphates. J. Agric. Food Chem. 12, 198–201 (1964)
Wajima, T., Rakovan, J.F.: Removal behavior of phosphate from aqueous solution by calcined paper sludge. Colloid Surf. A 435, 132–138 (2013)
Finnish Ministry of Finance.: The Waste Tax Act 1126/2010. https://finlex.fi/fi/laki/ajantasa/2010/20101126 2016. Accessed 2 Apr 2019
Ministry of Agriculture and Forestry Decree on fertilizer products 24/2011. Ministry of Agriculture and Forestry 24/2011 (2011)
Nkoa, R.: Agricultural benefits and environmental risks of soil fertilization with anaerobic digestates: a review. Agron. Sustain. Dev. 34, 473–492 (2014)
Lin, H., Yunqin Lin, Y., Wang, D., Pang, Y., Zhang, F., Tan, S.: Ammonium removal from digested effluent of swine wastewater by using solid residue from magnesium-hydroxide flue gas desulfurization process. J. Ind. Eng. Chem. 58, 148–154 (2018)
Yu, Y., Lei, Z., Yuan, T., Jiang, Y., Chen, N., Feng, C., Shimizu, K., Zhang, Z.: Simultaneous phosphorus and nitrogen recovery from anaerobically digested sludge using a hybrid system coupling hydrothermal pretreatment with MAP precipitation. Bioresour. Technol. 243, 634–640 (2017). https://doi.org/10.1016/j.biortech.2017.06.178
Johansson, L.: Industrial by-products and natural substrata as phosphorus sorbents. Environ. Technol. 20, 309–316 (1999)
Can, M.Y., Yildiz, E.: Phosphate removal from water by fly ash: factorial experimantal design. J. Hazard. Mater. B135, 165–170 (2006)
Darwish, M., Aris, A., Puteh, M.H., Jusoh, M.N.H., Kadir, A.A.: Waste bones ash as an alternative source of P for struvite preciptation. J. Environ. Manage. 203, 861–866 (2017)
Moriyama, K., Kojima, T., Minawa, Y., Matsumoto, S., Nakamachi, K.: Development of artificial seed crystal for crystallization of calcium phosphate. Environ. Technol. 22, 1245–1252 (2001)
Urdalen, I.: Phosphorus recovery from municipal wastewater. Norwegian University of Science and Technology, Trondheim (2013)
Rahaman, M.S., Mavinic, D.S., Bhuiyan, M.I.H., Koch, F.A.: Exploring the determination of struvite solubility product from analytical results. Environ. Technol. 27, 951–961 (2006)
Luukkonen, T., Veznikova, K., Tolonen, E.T., Runtti, H., Yliniemi, J., Hu, T., Kemppainen, K., Lassi, U.: Removal of ammonium from municipal wastewater with powdered and granulated metakaolin geopolymer. Environ. Technol. 39(4), 414–423 (2017)
Lu, S.G., Bai, S.Q., Zhu, L., Shan, H.D.: Removal mechanism of phosphate from aqueous solution by fly ash. J. Hazard. Mater. 161, 95–101 (2009). https://doi.org/10.1016/j.jhazmat.2008.02.123
Hermassi, M., Valderrama, C., Moreno, N., Font, O., Querol, X., Batis, N.H., Cortina, J.L.: Fly ash as reactive sorbent for phosphate removal from treated waste water as a potential slow release fertilizer. J. Environ. Chem. Eng. 5, 160–169 (2017). https://doi.org/10.1016/j.jece.2016.11.027
Liu, Y., Sheng, X., Dong, Y., Ma, Y.: Removal of high-concentration phosphate by calcite: effect of sulfate and pH. Desalination 289, 66–71 (2012). https://doi.org/10.1016/j.desal.2012.01.011
Uğurlu, M., Karaoğlu, M.H.: Adsorption of ammonium from an aqueous solution by fly ash and sepiolite: isotherm, kinetic and thermodynamic analysis. Micropor. Mesopor. Mat. 139, 173–178 (2011). https://doi.org/10.1016/j.micromeso.2010.10.039
Acknowledgements
Open access funding provided by University of Oulu including Oulu University Hospital. The authors would like to thank laboratory assistant, Mr. Jaakko Pulkkinen for his assistance in the laboratory analysis.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Myllymäki, P., Pesonen, J., Nurmesniemi, ET. et al. The Use of Industrial Waste Materials for the Simultaneous Removal of Ammonium Nitrogen and Phosphate from the Anaerobic Digestion Reject Water. Waste Biomass Valor 11, 4013–4024 (2020). https://doi.org/10.1007/s12649-019-00724-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12649-019-00724-8