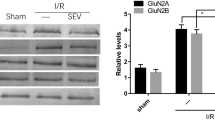
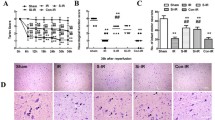
Abstract
Cerebral ischemia/reperfusion (I/R) injury is the main cause of death following trauma. The neuroprotective effect of sevoflurane (Sev) has been implicated in cerebral I/R injury. However, the mechanisms remain elusive. In this study, we aimed to explore its function in PC12 exposed to oxygen–glucose deprivation/reoxygenation (OGD/R) and in rats challenged with I/R. Sev pretreatment reduced the damage of PC12 cells after OGD/R treatment. Moreover, Sev pretreatment ameliorated neurobehavioral deficits induced by I/R treatment, reduced brain infarct volume, and decreased apoptosis of neurons in hippocampal tissues. Sev pretreatment reduced the surface expression of glutamate receptor 1 (GRIA1) in neurons, while GRIA1 reduced the neuroprotective effects of Sev pretreatment in vitro and in vivo. There was no difference in the surface expression of GRIA2 in rats with I/R and PC12 cells exposed to OGD/R. The ratio of GRIA1/GRIA2 surface expression was reduced, and calcium permeable-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid receptor (CP-AMPAR) was blocked by Sev. Together, Sev might exert beneficial effects on cerebral I/R-induced neuronal injury through inhibiting the surface expression of GRIA1 and blocking CP-AMPAR.







Similar content being viewed by others
Availability of Data and Materials
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Achzet LM, Davison CJ, Shea M, Sturgeon I, Jackson DA (2021) Oxidative stress underlies the ischemia/reperfusion-induced internalization and degradation of AMPA receptors. Int J Mol Sci 22. https://doi.org/10.3390/ijms22020717
Cai M, Tong L, Dong B, Hou W, Shi L, Dong H (2017) Kelch-like ECH-associated protein 1-dependent nuclear factor-E2-related factor 2 activation in relation to antioxidation induced by sevoflurane preconditioning. Anesthesiology 126:507–521. https://doi.org/10.1097/ALN.0000000000001485
Feske SK (2021) Ischemic Stroke Am J Med 134:1457–1464. https://doi.org/10.1016/j.amjmed.2021.07.027
Ge Y, Wang YT (2021) GluA1-homomeric AMPA receptor in synaptic plasticity and neurological diseases. Neuropharmacology 197:108708. https://doi.org/10.1016/j.neuropharm.2021.108708
Gerace E et al (2014) PARP-1 activation causes neuronal death in the hippocampal CA1 region by increasing the expression of Ca(2+)-permeable AMPA receptors. Neurobiol Dis 70:43–52. https://doi.org/10.1016/j.nbd.2014.05.023
Henley JM, Wilkinson KA (2016) Synaptic AMPA receptor composition in development, plasticity and disease. Nat Rev Neurosci 17:337–350. https://doi.org/10.1038/nrn.2016.37
Hu B, Tian T, Hao PP, Liu WC, Chen YG, Jiang TY, Xue FS (2022) The Protective Effect of Sevoflurane Conditionings against Myocardial Ischemia/reperfusion Injury: a Systematic Review and Meta-Analysis of Preclinical Trials in in-Vivo Models Front Cardiovasc Med 9:841654. https://doi.org/10.3389/fcvm.2022.841654
Ibanez L et al (2022) Multi-ancestry GWAS reveals excitotoxicity associated with outcome after ischaemic stroke. Brain 145:2394–2406. https://doi.org/10.1093/brain/awac080
Ji H, Li H, Zhang H, Cheng Z (2022) Role of microRNA-218–5p in sevoflurane-induced protective effects in hepatic ischemia/reperfusion injury mice by regulating GAB2/PI3K/AKT pathway. Mol Med Rep 25. https://doi.org/10.3892/mmr.2021.12517
Jiang JJ, Li C, Li H, Zhang L, Lin ZH, Fu BJ, Zeng YM (2016) Sevoflurane postconditioning affects post-ischaemic myocardial mitochondrial ATP-sensitive potassium channel function and apoptosis in ageing rats. Clin Exp Pharmacol Physiol 43:552–561. https://doi.org/10.1111/1440-1681.12565
Jin L, Bo XM (2021) Neuroprotection of sevoflurane against ischemia/reperfusion-induced brain injury through inhibiting GluN2A/GluN2B-PSD-95-MLK3 module. Exp Brain Res 239:2701–2709. https://doi.org/10.1007/s00221-021-06157-x
Jurcau A, Simion A (2021) Neuroinflammation in cerebral ischemia and ischemia/reperfusion injuries: from pathophysiology to therapeutic strategies. Int J Mol Sci 23. https://doi.org/10.3390/ijms23010014
Li Y, Xing N, Yuan J, Yang J (2020) Sevoflurane attenuates cardiomyocyte apoptosis by mediating the miR-219a/AIM2/TLR4/MyD88 axis in myocardial ischemia/reperfusion injury in mice. Cell Cycle 19:1665–1676. https://doi.org/10.1080/15384101.2020.1765512
Liang TY, Peng SY, Ma M, Li HY, Wang Z, Chen G (2021) Protective Effects of Sevoflurane in Cerebral Ischemia Reperfusion Injury: a Narrative Review Med Gas Res 11:152–154. https://doi.org/10.4103/2045-9912.318860
Ma H et al (2021) Sevoflurane protects the liver from ischemia-reperfusion injury by regulating Nrf2/HO-1 pathway. Eur J Pharmacol 898:173932. https://doi.org/10.1016/j.ejphar.2021.173932
Man HY (2011) GluA2-lacking, calcium-permeable AMPA receptors–inducers of plasticity? Curr Opin Neurobiol 21:291–298. https://doi.org/10.1016/j.conb.2011.01.001
Mo Z, Zeng Z, Liu Y, Zeng L, Fang J, Ma Y (2022) Activation of Wnt/beta-Catenin Signaling Pathway as a Promising Therapeutic Candidate for Cerebral Ischemia/reperfusion Injury Front Pharmacol 13:914537. https://doi.org/10.3389/fphar.2022.914537
Neag MA, Mitre AO, Catinean A, Mitre CI (2020) An overview on the mechanisms of neuroprotection and neurotoxicity of isoflurane and sevoflurane in experimental studies. Brain Res Bull 165:281–289. https://doi.org/10.1016/j.brainresbull.2020.10.011
Nunes RR, Duval Neto GF, Garcia de Alencar JC, Franco SB, de Andrade NQ, Holanda Dumaresq DM, Cavalcante SL (2013) Anesthetics, cerebral protection and preconditioning. Braz J Anesthesiol 63:119–128. https://doi.org/10.1016/S0034-7094(13)70204-6
Qu W et al (2021) Emerging role of AMPA receptor subunit GluA1 in synaptic plasticity: implications for Alzheimer’s disease. Cell Prolif 54:e12959. https://doi.org/10.1111/cpr.12959
Shen K, Limon A (2021) Transcriptomic expression of AMPA receptor subunits and their auxiliary proteins in the human brain. Neurosci Lett 755:135938. https://doi.org/10.1016/j.neulet.2021.135938
Stockwell J, Chen Z, Niazi M, Nosib S, Cayabyab FS (2016) Protein phosphatase role in adenosine A1 receptor-induced AMPA receptor trafficking and rat hippocampal neuronal damage in hypoxia/reperfusion injury. Neuropharmacology 102:254–265. https://doi.org/10.1016/j.neuropharm.2015.11.018
Stuckey SM, Ong LK, Collins-Praino LE, Turner RJ (2021) Neuroinflammation as a key driver of secondary neurodegeneration following stroke?. Int J Mol Sci 22. https://doi.org/10.3390/ijms222313101
Thushara Vijayakumar N, Sangwan A, Sharma B, Majid A, Rajanikant GK (2016) Cerebral ischemic preconditioning: the road so far. Mol Neurobiol 53:2579–2593. https://doi.org/10.1007/s12035-015-9278-z
Tong L, Cai M, Huang Y, Zhang H, Su B, Li Z, Dong H (2014) Activation of K(2)P channel-TREK1 mediates the neuroprotection induced by sevoflurane preconditioning. Br J Anaesth 113:157–167. https://doi.org/10.1093/bja/aet338
Wang D, Fang B, Wang Z, Li X, Chen F (2021) Sevoflurane Pretreatment Regulates Abnormal Expression of MicroRNAs Associated with Spinal Cord Ischemia/reperfusion Injury in Rats Ann Transl Med 9:752. https://doi.org/10.21037/atm-20-7864
Xu H, Mei XP, Xu LX (2018) The effect of pre- and after-treatment of sevoflurane on central ischemia tolerance and the underlying mechanisms J Dent Anesth. Pain Med 18:1–8. https://doi.org/10.17245/jdapm.2018.18.1.1
Xu L, Ge F, Hu Y, Yu Y, Guo K, Miao C (2021) Sevoflurane postconditioning attenuates hepatic ischemia-reperfusion injury by limiting HMGB1/TLR4/NF-kappaB pathway via modulating microRNA-142 in vivo and in vitro. Front Pharmacol 12:646307. https://doi.org/10.3389/fphar.2021.646307
Zhang K, Xu X, Hu L (2022) Sevoflurane attenuates hepatic ischemia reperfusion injury by the miR-122/Nrf2 pathway. Ann Transl Med 10:350. https://doi.org/10.21037/atm-22-115
Zhong H, Chen H, Gu C (2020) Sevoflurane post-treatment upregulated miR-203 expression to attenuate cerebral ischemia-reperfusion-induced neuroinflammation by targeting MyD88. Inflammation 43:651–663. https://doi.org/10.1007/s10753-019-01147-2
Author information
Authors and Affiliations
Contributions
YL is the guarantor of integrity of the entire study and contributed to the concepts; ZL, SYL, XNW, and TY contributed to the experimental studies, data acquisition, and statistical analysis; KM and KC contributed to data analysis and manuscript review. All authors contributed to the manuscript preparation and read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethical Approval
The procedures regarding animals were carried out as per the institutional guideline of Guide for the Care and Use of Laboratory Animals (proposed by NIH), and the protocols used in the present study were approved by the Ethics Committee for the Second Hospital of Hebei Medical University.
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Li, Y., Liang, Z., Lei, S. et al. Sevoflurane Preconditioning Downregulates GRIA1 Expression to Attenuate Cerebral Ischemia–Reperfusion-Induced Neuronal Injury. Neurotox Res 41, 29–40 (2023). https://doi.org/10.1007/s12640-022-00620-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12640-022-00620-5