Abstract
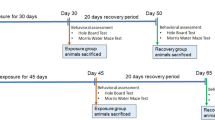
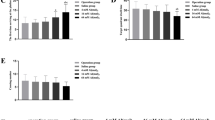
Aluminum (Al) is a neurotoxin that gradually accumulates in the brain in human life, resulting in oxidative brain injury related to Alzheimer’s disease (AD) and other diseases. In this study, the learning and memory of rats exposed to different aluminum concentrations (0.0 g/L, 2.0 g/L, 4.0 g/L, and 8.0 g/L) were studied, and the learning and memory of rats were observed by shuttle box experiment. With hematoxylin and eosin staining, Western blot, immunofluorescence, and RT-PCR, the morphology of nerve cells in the hippocampus of rat brain were observed, and the levels of activator protein-1 (AP-1) gene and protein, nerve growth factor (NGF), neurotrophin-3 (NT3), glial cell line-derived neurotrophic factor (GDNF), and brain-derived neurotrophic factor (BDNF) gene and protein level, etc. The experimental results showed that subchronic aluminum exposure damaged learning and memory in rats. The cognitive function damage in rats was more evident after increasing the aluminum intake dose. The more aluminum intake, the more pronounced the histological changes in the hippocampus will be. The expression level and protein content of neurotrophic factors in the hippocampus of rats showed a negative correlation with aluminum intake. In this experiment, we explored the mechanism of aluminum exposure in learning and memory disorders, and provided some data reference for further elucidation of the damage mechanism of aluminum on the nervous system and subsequent preventive measures.











Similar content being viewed by others
Data Availability
The analyzed data sets generated during the present study are available from the corresponding author on reasonable request.
References
Alberini CM (2009) Transcription factors in long-term memory and synaptic plasticity. Physiol Rev 89(1):121–145. https://doi.org/10.1152/physrev.00017.2008
Ameyar M, Wisniewska M, Weitzman JB (2003) A role for AP-1 in apoptosis: the case for and against. Biochimie 85(8):747–752. https://doi.org/10.1016/j.biochi.2003.09.006
Barbacid M (1994) The Trk family of neurotrophin receptors. J Neurobiol 25(11):1386–1403. https://doi.org/10.1002/neu.480251107
Cao P, Liu AD, Yang DJ, Liang J, Wang XD, Xu HB, Ma N (2019) Assessment of dietary exposure of young Chinese children to aluminium residues. Food Addit Contam A Chem Anal Control Expo Risk Assess 36(4):582–591. https://doi.org/10.1080/19440049.2019.1579369
Connor B, Dragunow M (1998) The role of neuronal growth factors in neurodegenerative disorders of the human brain. Brain Res Rev 27(1):1–39. https://doi.org/10.1016/s0165-0173(98)00004-6
Dai WL, Yan B, Bao YN, Fan JF, Liu JH (2020) Suppression of peripheral NGF attenuates neuropathic pain induced by chronic constriction injury through the T-AK1-MAPK/NF-κB signaling pathways. Cell Commun Signal 18(1):66. https://doi.org/10.1186/s12964020-00556-3
de Sousa Fernandes MS, Ordônio TF, Santos G, Santos L, Calazans CT, Gomes DA, Santos TM (2020) Effects of physical exercise on neuroplasticity and brain function: a systematic review in human and animal studies. Neural Plast 2020:8856621. https://doi.org/10.1155/2020/8856621
Eferl R, Wagner EF (2003) AP-1: a double-edged sword in tumorigenesis. Nat Rev Cancer 3(11):859–868. https://doi.org/10.1038/nrc1209
Flaten TP (2001) Aluminium as a risk factor in Alzheimer’s disease, with emphasis on drinking water. Brain Res Bull 55(2):187–196. https://doi.org/10.1016/s0361-9230(01)00459-2
Fontana X, Hristova M, Da Costa C, Patodia S, Thei L, Makwana M, SpencerDene B, Latouche M, Mirsky R, Jessen KR, Klein R, Raivich G, Behrens A (2012) c-Jun in Schwann cells promotes axonal regeneration and motoneuron survival via paracrine signaling. J Cell Biol 198(1):127–141. https://doi.org/10.1083/jcb.201205025
Gao L, Peng X, Huo S, He Y, Yan M (2014) Chin J Cell Mol Immunol 30(10):1022–1025 (PMID: 25270201)
Grimm R, Schicknick H, Riede I, Gundelfinger ED, Herdegen T, Zuschratter W, Tischmeyer W (1997) Suppression of c-fos induction in rat brain impairs retention of a brightness discrimination reaction. Learn Mem 3(5):402–413. https://doi.org/10.1101/lm.3.5.402
Hengerer B, Lindholm D, Heumann R, Rüther U, Wagner EF, Thoenen H (1990) Lesion-induced increase in nerve growth factor mRNA is mediated by c-fos. Proc Natl Acad Sci USA 87(10):3899–3903. https://doi.org/10.1073/pnas.87.10.3899
Herdegen T, Leah JD (1998) Inducible and constitutive transcription factors in the mammalian nervous system: control of gene expression by Jun, Fos and Krox, and CREB/ATF proteins. Brain Res Rev 28(3):370–490. https://doi.org/10.1016/s0165-0173(98)00018-6
Huang EJ, Reichardt LF (2003) Trk receptors: roles in neuronal signal transduction. Annu Rev Biochem 72:609–642. https://doi.org/10.1146/annurev.biochem.72.121801.161629
Izquierdo I, Bevilaqua LR, Rossato JI, Bonini JS, Medina JH, Cammarota M (2006) Different molecular cascades in different sites of the brain control memory consolidation. Trends Neurosci 29(9):496–505. https://doi.org/10.1016/j.tins.2006.07.005
Johnson GL, Lapadat R (2002) Mitogen-activated protein kinase pathways mediated by ERK, JNK, and p38 protein kinases. Science 298(5600):1911–1912. https://doi.org/10.1126/science.1072682
Johnson VJ, Sharma RP (2003) Aluminum disrupts the proinflammatory cytokine/neurotrophin balance in primary brain rotation-mediated aggregate cultures: possible rolein neurodegeneration. Neurotoxicology 24(2):261–268. https://doi.org/10.1016/S0161-813X(02)00194-8
Kaminska M, O’Sullivan M, Mery VP, Lafontaine AL, Robinson A, Gros P, Martin JG, Benedetti A, Kimoff RJ (2022) Inflammatory markers and BDNF in obstructive sleep apnea (OSA) in Parkinson’s disease (PD). Sleep Med 90:258–261. https://doi.org/10.1016/j.sleep.2021.11.018 (Advance online publication)
Kandimalla R, Vallamkondu J, Corgiat EB, Gill KD (2016) Understanding aspects of aluminum exposure in Alzheimer’s disease development. Brain Pathol 26(2):139–154. https://doi.org/10.1111/bpa.12333
Kaur A, Joshi K, Minz RW, Gill KD (2006) Neurofilament phosphorylation and disruption: a possible mechanism of chronic aluminium toxicity in Wistar rats. Toxicology 219(1–3):1–10. https://doi.org/10.1016/j.tox.2005.09.015
Keefe KM, Sheikh IS, Smith GM (2017) Targeting neurotrophins to specific populations of neurons: NGF, BDNF, and NT-3 and their relevance for treatment of spinal cord injury. Int J Mol Sci 18(3):548. https://doi.org/10.3390/ijms18030548
Kobayashi N, Nagata T, Shinagawa S, Nakayama R, Kondo K, Nakayama K, Yamada H (2012) Association between neurotrophin-3 polymorphisms and executive function in Japanese patients with amnestic mild cognitive impairment and mild Alzheimer disease. Dement Geriatr Cogn Disord 34(3–4):190–197. https://doi.org/10.1159/000343075
Krewski D, Yokel RA, Nieboer E, Borchelt D, Cohen J, Harry J, Kacew S, Lindsay J, Mahfouz AM, Rondeau V (2007) Human health risk assessment foraluminium, aluminium oxide, and aluminium hydroxide. J Toxicol Environ Health B Crit Rev 10(S1):1–269. https://doi.org/10.1080/10937400701597766
Lamballe F, Klein R, Barbacid M (1991) trkC, a new member of the trk family of tyrosine protein kinases, is a receptor for neurotrophin-3. Cell 66(5):967–979. https://doi.org/10.1016/0092-8674(91)90442-2
Lamballe F, Smeyne RJ, Barbacid M (1994) Developmental expression of trkC, the neurotrophin-3 receptor, in the mammalian nervous system. J Neurosci Off J Soc Neurosci 14(1):14–28. https://doi.org/10.1523/JNEUROSCI.14-01-00014.1994
Leal G, Comprido D, Duarte CB (2014) BDNF-induced local protein synthesis and synaptic plasticity. Neuropharmacology 76:639–656. https://doi.org/10.1016/j.neuropharm.2013.04.005
Leppä S, Bohmann D (1999) Diverse functions of JNK signaling and c-Jun in stress response and apoptosis. Oncogene 18(45):6158–6162. https://doi.org/10.1038/sj.onc.1203173
Lessmann V, Gottmann K, Malcangio M (2003) Neurotrophin secretion: current facts and future prospects. Prog Neurobiol 69(5):341–374. https://doi.org/10.1016/s0301-0082(03)00019-4
Li X, Li N, Sun HL, Yin J, Tao YC, Mao ZX, Yu ZL, Li WJ, Bogden JD (2017) Maternal lead exposure induces down-regulation of hippocampal insulin-degrading enzyme and nerve growth factor expression in mouse pups. Biome Environ Sci 30(3):215–219. https://doi.org/10.3967/bes2017.029
Liaquat L, Sadir S, Batool Z, Tabassum S, Shahzad S, Afzal A, Haider S (2019) Acute aluminum chloride toxicity revisited: study on DNA damage and histopathological, bio-chemical and neurochemical alterations in rat brain. Life Sci 217:202–211. https://doi.org/10.1016/j.lfs.2018.12.009
Maisonpierre PC, Belluscio L, Friedman B, Alderson RF, Wiegand SJ, Furth ME, Lindsay RM, Yancopoulos GD (1990) NT-3, BDNF, and NGF in the developing rat nervous system: parallel as well as reciprocal patterns of expression. Neuron 5(4):501–509. https://doi.org/10.1016/0896-6273(90)90089-x
Meng H, Wang S, Guo J, Zhao Y, Zhang S, Zhao Y, Niu Q (2019) Cognitive impairment of workers in a large-scale aluminium factory in China: a cross-sectional study. BMJ Open 9(6):e027154. https://doi.org/10.1136/bmjopen-2018-027154
Mooradian AD (1988) Effect of aging on the blood-brain barrier. Neurobiol Aging 9(1):31–39. https://doi.org/10.1016/s0197-4580(88)80013-7
Mufson EJ, Ginsberg SD, Ikonomovic MD, DeKosky ST (2003) Human cholinergic basal forebrain: chemoanatomy and neurologic dysfunction. J Chem Neuroanat 26(4):233–242. https://doi.org/10.1016/s0891-0618(03)00068-1
Nagata T, Shibata N, Shinagawa S, Nakayama R, Kuerban B, Ohnuma T, Arai H, Nakayama K, Yamada H (2013) Genetic association between neurotrophin-3 polymorphisms and Alzheimer’s disease in Japanese patients. Dement Geriatr Cogn Disord Extra 3(1):272–280. https://doi.org/10.1159/000354369
Niewiadomska G, Baksalerska-Pazera M, Riedel G (2009) The septohippocampal system, learning and recovery of function. Prog Neuropsychopharmacol Biol Psychiatry 33(5):791–805. https://doi.org/10.1016/j.pnpbp.2009.03.039
Pertusa M, García-Matas S, Mammeri H et al (2008) Expression of GDNF transgene in astrocytes improves cognitive deficits in aged rats. Neurobiol Aging 29(9):1366–1379. https://doi.org/10.1016/j.neurobiolaging.2007.02.026
Petersen RC, Caracciolo B, Brayne C, Gauthier S, Jelic V, Fratiglioni L (2014) Mild cognitive impairment: a concept in evolution. J Intern Med 275(3):214–228. https://doi.org/10.1111/joim.12190
Pierce AL, Kawas CH (2017) Dementia in the oldest old: beyond Alzheimer disease. PLoS Med 14(3):e1002263. https://doi.org/10.1371/journal.pmed.1002263
Priest ND, Talbot RJ, Newton D, Day JP, King SJ, Fifield LK (1998) Uptake by man of aluminium in a public water supply. Hum Exp Toxicol 17(6):296–301. https://doi.org/10.1177/096032719801700602
Ramos-Languren LE, Escobar ML (2013) Plasticity and metaplasticity of adult rat hippocampal mossy fibers induced by neurotrophin-3. Eur J Neurosci 37(8):1248–1259. https://doi.org/10.1111/ejn.12141
Rob PM, Niederstadt C, Reusche E (2001) Dementia in patients undergoing long-term dialysis: aetiology, differential diagnoses, epidemiology and management. CNS Drugs 15(9):691–699. https://doi.org/10.2165/00023210-200115090-00003
Shaulian E, Karin M (2002) AP-1 as a regulator of cell life and death. Nat Cell Biol 4(5):E131–E136. https://doi.org/10.1038/ncb0502-e131
Shaw CA (2018) Aluminum as a CNS and immune system toxin across the life span. Adv Exp Med Biol 1091:53–83. https://doi.org/10.1007/978-981-13-1370-7_4
Skaper SD (2018) Neurotrophic factors: an overview. Methods Mol Biol 1727:1–17. https://doi.org/10.1007/978-1-4939-7571-6_1
Stephen G, Whitehouse PJ (1992) Alzheimer disease and associated disorders: integrative, international, interdisciplinary. Alzheimer Dis Assoc Disord 6:269–270
Tessarollo L, Tsoulfas P, Martin-Zanca D, Gilbert DJ, Jenkins NA, Copeland NG, Parada LF (1993) trkC, a receptor for neurotrophin-3, is widely expressed in the developing nervous system and in non-neuronal tissues. Development 118(2):463–475 (PMID: 8223273)
Tomljenovic L (2011) Aluminum and Alzheimer’s disease: after a century of controversy, is there a plausible link? J Alzheimer’s Dis 23(4):567–598. https://doi.org/10.3233/JAD-2010-101494
Tuvikene J, Pruunsild P, Orav E, Esvald EE, Timmusk T (2016) AP-1 transcription factors mediate BDNF-positive feedback loop in cortical neurons. J Neurosci Off J Soc Neurosci 36(4):1290–1305. https://doi.org/10.1523/JNEUROSCI.3360-15.2016
Virk SA, Eslick GD (2015) Aluminum levels in brain, serum, and cerebrospinal fluid are higher in Alzheimer’s disease cases than in controls: a series of meta-analyses. J Alzheimer’s Dis 47(3):629–638. https://doi.org/10.3233/JAD-150193
Walton JR (2009) Functional impairment in aged rats chronically exposed to human range dietary aluminum equivalents. Neurotoxicology 30(2):182–193. https://doi.org/10.1016/j.neuro.2008.11.012
Wang B, Xing W, Zhao Y, Deng X (2010a) Effects of chronic aluminum exposure on memory through multiple signal transduction pathways. Environ Toxicol Pharmacol 29(3):308–313. https://doi.org/10.1016/j.etap.2010.03.007
Wang Y, Tian Q, Chen J, Wen T, Gao MQ, Zeng X, Zheng X (2010b) Effects of developmental lead exposure on hippocampal GDNF protein expression in rats. China Public Health 12:1528–1529
Wang L, Hu J, Zhao Y, Lu X, Zhang Q, Niu Q (2014) Effects of aluminium on β-amyloid (1–42) and secretases (APP-cleaving enzymes) in rat brain. Neurochem Res 39(7):1338–1345. https://doi.org/10.1007/s11064-014-1317-z
Wang J, Bast T, Wang YC, Zhang WN (2015) Hippocampus and two-way active avoidance conditioning: contrasting effects of cytotoxic lesion and temporary inactivation. Hippocampus 25(12):1517–1531. https://doi.org/10.1002/hipo.22471
Wang Z, Wei X, Yang J, Suo J, Chen J, Liu X, Zhao X (2016) Chronic exposure to aluminum and risk of Alzheimer’s disease: a meta-analysis. Neurosci Lett 610:200–206. https://doi.org/10.1016/j.neulet.2015.11.014
Wang F, Kang P, Li Z, Niu Q (2019) Role of MLL in the modification of H3K4me3 in aluminium-induced cognitive dysfunction. Chemosphere 232:121–129. https://doi.org/10.1016/j.chemosphere.2019.05.099
Wang T, Xue Y, Liang J (2021) Analysis of the correlation between brain-derived neurotrophic factor β-amyloid and tau protein and the cognitive function of elderly patients with Alzheimer’s disease. Shanxi Med J 24:3406–3408
Yokel RA, McNamara PJ (2001) Aluminium toxicokinetics: an updated mini review. Pharmacol Toxicol 88(4):159–167. https://doi.org/10.1034/j.1600-0773.2001.d01-98.x
Yumoto S, Nagai H, Matsuzaki H, Matsumura H, Tada W, Nagatsuma E, Kobayashi K (2001) Aluminium incorporation into the brain of rat fetuses and sucklings. Brain Res Bull 55(2):229–234. https://doi.org/10.1016/s0361-9230(01)00509-3
Zhang L, Shen X, Cui G, Hua F, Geng D (2003) Effects of calcitonin gene related peptide on DNA-binding activity of activated protein 1 in hippocampal CA1 region after global cerebral ischemia reperfusion in rats. Proc Xuzhou Med Coll 01:13–16
Zhang Y, Zhu W, Wang YG, Liu XJ, Jiao L, Liu X, Zhang ZH, Lu CL, He C (2006) Interaction of SH2-Bbeta with RET is involved in signaling of GDNF-induced neurite outgrowth. J Cell Sci 119(Pt 8):1666–1676. https://doi.org/10.1242/jcs.02845
Zhang L, Jin C, Liu Q, Lu X, Wu S, Yang J, Du Y, Zheng L, Cai Y (2013) Effects of subchronic aluminum exposure on spatial memory, ultrastructure and L-LTP of hippocampus in rats. J Toxicol Sci 38(2):255–268. https://doi.org/10.2131/jts.38.255
Zhang L, Jin C, Lu X, Yang J, Wu S, Liu Q, Chen R, Bai C, Zhang D, Zheng L, Du Y, Cai Y (2014) Aluminium chloride impairs long-term memory and downregulates cAMP-PKA-CREB signalling in rats. Toxicology 323:95–108. https://doi.org/10.1016/j.tox.2014.06.011
Zhang L, Bai C, Zhang D, Zhao L (2016a) Effects of subchronic aluminum exposure on learning-memory ability and c-Fos expression in hippocampal CA1 region of Wistar rats. Chin J Pharmacol Toxicol 05:553–557
Zhang L, Zhao L, Bai C, Zhang D (2016b) Effects of subchronic aluminum exposure on learning-memory ability and expression of c-Fos mRNA and c-fos protein in cerebral cortex of rats. J Neuroanat 04:447–451. https://doi.org/10.16557/J.CNKI.1000-7547.2016.04.005
Zhao G, Zhang H, Liu H (2018) Effects of low-intensity treadmill training on the gene and protein expression of hippocampal BDNF/ERK/CREB in Alzheimer’s disease transgenic mice. China J Sports Med 11:933–939. https://doi.org/10.16038/J.1000-6710.2018.11.009
Funding
This work was supported by the National Natural Science Foundation of China (81673226); by the Initiated Research Foundation for the Doctoral Program of Science and Technology Department of Liaoning Province, China (201601226); by the Natural Science Foundation of Education Department of Liaoning Province, China (L2015544, LJKZ1146); by the Natural Science Foundation for Innovation and Entrepreneurship Training Program of Education Department of Liaoning Province, China (201710164000038); by the Natural Science Foundation of Science and Technology Department of Shenyang City, China (17–231-1–44); by the Natural Science Foundation of Shenyang Medical College, China (20153043); by the Natural Science Foundation for Graduate Students of Shenyang Medical College, China (Y20180512); and by the Natural Science Foundation for Undergraduate Students of Shenyang Medical College, China (20179028).
Author information
Authors and Affiliations
Contributions
WL and JL designed the study, wrote the manuscript, and analyzed the data. JG reviewed the references. XD and LZ reviewed and revised the manuscript. All authors had read the manuscript and agree to the publication of this study.
Corresponding authors
Ethics declarations
Ethics Approval
The research was carried out following the National Institutes of Health Guide for the Care and Use of Laboratory Animals Standard.
Consent to Participate
Not applicable.
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Wei Liu and Jiaxi Liu contributed equally to this work.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Liu, W., Liu, J., Gao, J. et al. Effects of Subchronic Aluminum Exposure on Learning, Memory, and Neurotrophic Factors in Rats. Neurotox Res 40, 2046–2060 (2022). https://doi.org/10.1007/s12640-022-00599-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12640-022-00599-z