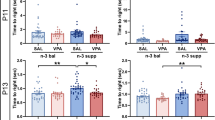
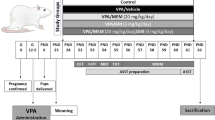
Abstract
This study examined the ameliorating effect of alpha-glycosyl isoquercitrin (AGIQ), an antioxidant, on disrupted hippocampal neurogenesis in the dentate gyrus (DG) in a rat model of autism spectrum disorder induced by prenatal valproic acid (VPA) exposure. Dams were intraperitoneally injected with 500 mg/kg VPA on gestational day 12. AGIQ was administered in the diet at 0.25 or 0.5% to dams from gestational day 13 until weaning at postnatal day (PND) 21 and then to pups until PND 63. At PND 21, VPA-exposed offspring showed decreased numbers of type-2a and type-3 neural progenitor cells (NPCs) among granule cell lineage subpopulations. AGIQ treatment at both doses rescued the reduction in type-3 NPCs. AGIQ upregulated Reln and Vldlr transcript levels in the DG at 0.5% and ≥ 0.25%, respectively, and increased the number of reelin+ interneurons in the DG hilus at 0.5%. AGIQ at 0.25% and/or 0.5% also upregulated Ntrk2, Cntf, Igf1, and Chrnb2. At PND 63, there were no changes in the granule cell lineage subpopulations in response to VPA or AGIQ. AGIQ at 0.25% increased the number of FOS+ granule cells, accompanied by Gria2 and Gria3 upregulation and increasing trend in the number of FOS+ granule cells at 0.5%. There was no definitive evidence of VPA-induced oxidative stress in the hippocampus throughout postnatal life. These results indicate that AGIQ ameliorates the VPA-induced disruption of hippocampal neurogenesis at weaning involving reelin, BDNF-TrkB, CNTF, and IGF1 signaling, and enhances FOS-mediated synaptic plasticity in adulthood, potentially through AMPA-receptor upregulation. The ameliorating effects of AGIQ may involve direct interactions with neural signaling cascades rather than antioxidant capacity.







Similar content being viewed by others
Data Availability
All data generated or analyzed during this study are included in this published article (and its supplementary information files).
References
Akane H, Saito F, Yamanaka H, Shiraki A, Imatanaka N, Akahori Y, Morita R, Mitsumori K, Shibutani M (2013) Methacarn as a whole brain fixative for gene and protein expression analyses of specific brain regions in rats. J Toxicol Sci 38:431–443. https://doi.org/10.2131/jts.38.431
Akiyama T, Washino T, Yamada T, Koda T, Maitani T (2000) Constituents of enzymatically modified isoquercitrin and enzymatically modified rutin (extract). Food Hyg Saf Sci (Shokuhin Eiseigaku Zasshi) 41:46–60. https://doi.org/10.3358/shokueishi.41.54
Baio J, Wiggins L, Christensen DL, Maenner MJ, Daniels J, Warren Z, Kurzius-Spencer M, Zahorodny W, Robinson Rosenberg C, White T, Durkin MS, Imm P, Nikolaou L, Yeargin-Allsopp M, Lee LC, Harrington R, Lopez M, Fitzgerald RT, Hewitt A, Pettygrove S, Constantino JN, Vehorn A, Shenouda J, Hall-Lande J, Van Naarden Braun K, Dowling NF (2018) Prevalence of autism spectrum disorder among children aged 8 years - autism and developmental disabilities monitoring network, 11 Sites, United States, 2014. MMWR Surveill Summ 67:1–23. https://doi.org/10.15585/mmwr.ss6706a1
Balschun D, Moechars D, Callaerts-Vegh Z, Vermaercke B, Van Acker N, Andries L, D’Hooge R (2010) Vesicular glutamate transporter VGLUT1 has a role in hippocampal long-term potentiation and spatial reversal learning. Cereb Cortex 20:684–693. https://doi.org/10.1093/cercor/bhp133
Bauer S, Kerr BJ, Patterson PH (2007) The neuropoietic cytokine family in development, plasticity, disease and injury. Nat Rev Neurosci 8:221–232. https://doi.org/10.1038/nrn2054
Berg DA, Belnoue L, Song H, Simon A (2013) Neurotransmitter-mediated control of neurogenesis in the adult vertebrate brain. Development 140:2548–2561. https://doi.org/10.1242/dev.088005
Bjørklund G, Meguid NA, El-Bana MA, Tinkov AA, Saad K, Dadar M, Hemimi M, Skalny AV, Hosnedlová B, Kizek R, Osredkar J, Urbina MA, Fabjan T, El-Houfey AA, Kałużna-Czaplińska J, Gątarek P, Chirumbolo S (2020) Oxidative stress in autism spectrum disorder. Mol Neurobiol 57:2314–2332. https://doi.org/10.1007/s12035-019-01742-2
Bristot Silvestrin R, Bambini-Junior V, Galland F, Daniele Bobermim L, Quincozes-Santos A, Torres Abib R, Zanotto C, Batassini C, Brolese G, Gonçalves CA, Riesgo R, Gottfried C (2013) Animal model of autism induced by prenatal exposure to valproate: altered glutamate metabolism in the hippocampus. Brain Res 1495:52–60. https://doi.org/10.1016/j.brainres.2012.11.048
Bromley RL, Mawer G, Clayton-Smith J, Baker GA, Liverpool and Manchester Neurodevelopment Group (2008) Autism spectrum disorders following in utero exposure to antiepileptic drugs. Neurology 71:1923–1924. https://doi.org/10.1212/01.wnl.0000339399.64213.1a
Catavero C, Bao H, Song J (2018) Neural mechanisms underlying GABAergic regulation of adult hippocampal neurogenesis. Cell Tissue Res 371:33–46. https://doi.org/10.1007/s00441-017-2668-y
Chaliha D, Albrecht M, Vaccarezza M, Takechi R, Lam V, Al-Salami H, Mamo J (2020) A systematic review of the valproic-acid-induced rodent model of autism. Dev Neurosci 42:12–48. https://doi.org/10.1159/000509109
Chen C, Magee JC, Bazan NG (2002) Cyclooxygenase-2 regulates prostaglandin E2 signaling in hippocampal long-term synaptic plasticity. J Neurophysiol 87:2851–2857. https://doi.org/10.1152/jn.2002.87.6.2851
Christensen J, Grønborg TK, Sørensen MJ, Schendel D, Parner ET, Pedersen LH, Vestergaard M (2013) Prenatal valproate exposure and risk of autism spectrum disorders and childhood autism. JAMA 309:1696–1703. https://doi.org/10.1001/jama.2013.2270
Dadalko OI, Travers BG (2018) Evidence for brainstem contributions to autism spectrum disorders. Front Integr Neurosci 12:47. https://doi.org/10.3389/fnint.2018.00047
Deckmann I, Schwingel GB, Fontes-Dutra M, Bambini-Junior V, Gottfried C (2018) Neuroimmune alterations in autism: a translational analysis focusing on the animal model of autism induced by prenatal exposure to valproic acid. NeuroImmunoModulation 25:285–299. https://doi.org/10.1159/000492113
Dlugosz P, Nimpf J (2018) The reelin receptors apolipoprotein E receptor 2 (ApoER2) and VLDL receptor. Int J Mol Sci 19:3090. https://doi.org/10.3390/ijms19103090
Elnahas EM, Abuelezz SA, Mohamad MI, Nabil MM, Abdelraouf SM, Bahaa N, Hassan GA, Ibrahim EA, Ahmed AI, Aboul-Fotouh S (2021) Validation of prenatal versus postnatal valproic acid rat models of autism: a behavioral and neurobiological study. Prog Neuropsychopharmacol Biol Psychiatry 108:110185. https://doi.org/10.1016/j.pnpbp.2020.110185
Formica JV, Regelson W (1995) Review of the biology of Quercetin and related bioflavonoids. Food Chem Toxicol 33:1061–1080. https://doi.org/10.1016/0278-6915(95)00077-1
Fowler JH, Whalley K, Murray T, O’neill MJ, McCulloch J (2004) The AMPA receptor potentiator LY404187 increases cerebral glucose utilization and c-fos expression in the rat. J Cereb Blood Flow Metab 24:1098–1109. https://doi.org/10.1097/01.WCB.0000138665.25305.7C
Freund TF, Buzsáki G (1996) Interneurons of the hippocampus. Hippocampus 6:347–470. https://doi.org/10.1002/(SICI)1098-1063(1996)6:4%3c347::AID-HIPO1%3e3.0.CO;2-I
Fujii Y, Kimura M, Ishii Y, Yamamoto R, Morita R, Hayashi SM, Suzuki K, Shibutani M (2013) Effect of enzymatically modified isoquercitrin on preneoplastic liver cell lesions induced by thioacetamide promotion in a two-stage hepatocarcinogenesis model using rats. Toxicology 305:30–40. https://doi.org/10.1016/j.tox.2013.01.002
Gasparotto Junior A, Gasparotto FM, Lourenço EL, Crestani S, Stefanello ME, Salvador MJ, da Silva-Santos JE, Marques MC, Kassuya CA (2011) Antihypertensive effects of isoquercitrin and extracts from Tropaeolum majus L.: evidence for the inhibition of angiotensin converting enzyme. J Ethnopharmacol 134:363–372. https://doi.org/10.1016/j.jep.2010.12.026
Gąssowska-Dobrowolska M, Cieślik M, Czapski GA, Jęśko H, Frontczak-Baniewicz M, Gewartowska M, Dominiak A, Polowy R, Filipkowski RK, Babiec L, Adamczyk A (2020) Prenatal exposure to valproic acid affects microglia and synaptic ultrastructure in a brain-region-specific manner in young-adult male rats: relevance to autism spectrum disorders. Int J Mol Sci 21:3576. https://doi.org/10.3390/ijms21103576
Ghowsi M, Khazali H, Sisakhtnezhad S (2018) Evaluation of TNF-α and IL-6 mRNAs expressions in visceral and subcutaneous adipose tissues of polycystic ovarian rats and effects of resveratrol. Iran J Basic Med Sci 21:165–174. https://doi.org/10.22038/ijbms.2017.24801.6167
Go HS, Kim KC, Choi CS, Jeon SJ, Kwon KJ, Han SH, Lee J, Cheong JH, Ryu JH, Kim CH, Ko KH, Shin CY (2012) Prenatal exposure to valproic acid increases the neural progenitor cell pool and induces macrocephaly in rat brain via a mechanism involving the GSK-3β/β-catenin pathway. Neuropharmacology 63:1028–1041. https://doi.org/10.1016/j.neuropharm.2012.07.028
Gonçalves JT, Schafer ST, Gage FH (2016) Adult neurogenesis in the hippocampus: from stem cells to behavior. Cell 167:897–914. https://doi.org/10.1016/j.cell.2016.10.021
Gulyás AI, Hájos N, Freund TF (1996) Interneurons containing calretinin are specialized to control other interneurons in the rat hippocampus. J Neurosci 16:3397–3411. https://doi.org/10.1523/JNEUROSCI.16-10-03397.1996
Hallmayer J, Cleveland S, Torres A, Phillips J, Cohen B, Torigoe T, Miller J, Fedele A, Collins J, Smith K, Lotspeich L, Croen LA, Ozonoff S, Lajonchere C, Grether JK, Risch N (2011) Genetic heritability and shared environmental factors among twin pairs with autism. Arch Gen Psychiatry 68:1095–1102. https://doi.org/10.1001/archgenpsychiatry.2011.76
Hamzawy MA, El-Ghandour YB, Abdel-Aziem SH, Ali ZH (2018) Leptin and camel milk abate oxidative stress status, genotoxicity induced in valproic acid rat model of autism. Int J Immunopathol Pharmacol 32:1–11. https://doi.org/10.1177/2058738418785514
He H, Mahnke AH, Doyle S, Fan N, Wang CC, Hall BJ, Tang YP, Inglis FM, Chen C, Erickson JD (2012) Neurodevelopmental role for VGLUT2 in pyramidal neuron plasticity, dendritic refinement, and in spatial learning. J Neurosci 32:15886–15901. https://doi.org/10.1523/JNEUROSCI.4505-11.2012
Hodge RD, Kowalczyk TD, Wolf SA, Encinas JM, Rippey C, Enikolopov G, Kempermann G, Hevner RF (2008) Intermediate progenitors in adult hippocampal neurogenesis: Tbr2 expression and coordinate regulation of neuronal output. J Neurosci 28:3707–3717. https://doi.org/10.1523/JNEUROSCI.4280-07.2008
Hu A, Yuan H, Wu L, Chen R, Chen Q, Zhang T, Wang Z, Liu P, Zhu X (2016) The effect of constitutive over-expression of insulin-like growth factor 1 on the cognitive function in aged mice. Brain Res 1631:204–213. https://doi.org/10.1016/j.brainres.2015.11.010
Jaworski J, Kalita K, Knapska E (2018) c-Fos and neuronal plasticity: the aftermath of Kaczmarek’s theory. Acta Neurobiol Exp (wars) 78:287–296
Jurga AM, Paleczna M, Kuter KZ (2020) Overview of general and discriminating markers of differential microglia phenotypes. Front Cell Neurosci 14:198. https://doi.org/10.3389/fncel.2020.00198
Kangawa Y, Yoshida T, Abe H, Seto Y, Miyashita T, Nakamura M, Kihara T, Hayashi SM, Shibutani M (2017) Anti-inflammatory effects of the selective phosphodiesterase 3 inhibitor, cilostazol, and antioxidants, enzymatically-modified isoquercitrin and α-lipoic acid, reduce dextran sulphate sodium-induced colorectal mucosal injury in mice. Exp Toxicol Pathol 69:179–186. https://doi.org/10.1016/j.etp.2016.12.004
Kempermann G, Song H, Gage FH (2015) Neurogenesis in the adult hippocampus. Cold Spring Harb Perspect Biol 7:a018812. https://doi.org/10.1101/cshperspect.a018812
Khalaj R, Hajizadeh Moghaddam A, Zare M (2018) Hesperetin and it nanocrystals ameliorate social behavior deficits and oxido-inflammatory stress in rat model of autism. Int J Dev Neurosci 69:80–87. https://doi.org/10.1016/j.ijdevneu.2018.06.009
Kondo M, Koyama Y, Nakamura Y, Shimada S (2018) A novel 5HT3 receptor-IGF1 mechanism distinct from SSRI-induced antidepressant effects. Mol Psychiatry 23:833–842. https://doi.org/10.1038/mp.2017.87
Lian D, He D, Wu J, Liu Y, Zhu M, Sun J, Chen F, Li L (2016) Exogenous BDNF increases neurogenesis in the hippocampus in experimental Streptococcus pneumoniae meningitis. J Neuroimmunol 294:46–55. https://doi.org/10.1016/j.jneuroim.2016.03.014
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2-ΔΔCT Method. Methods 25:402–408. https://doi.org/10.1006/meth.2001.1262
Loebrich S, Nedivi E (2009) The function of activity-regulated genes in the nervous system. Physiol Rev 89:1079–1103. https://doi.org/10.1152/physrev.00013.2009
Makino T, Kanemaru M, Okuyama S, Shimizu R, Tanaka H, Mizukami H (2013) Anti-allergic effects of enzymatically modified isoquercitrin (α-oligoglucosyl quercetin 3-O-glucoside), quercetin 3-O-glucoside, α-oligoglucosyl rutin, and quercetin, when administered orally to mice. J Nat Med 67:881–886. https://doi.org/10.1007/s11418-013-0760-5
Masiulis I, Yun S, Eisch AJ (2011) The interesting interplay between interneurons and adult hippocampal neurogenesis. Mol Neurobiol 44:287–302. https://doi.org/10.1007/s12035-011-8207-z
Matsuo K, Yabuki Y, Fukunaga K (2020) 5-aminolevulinic acid inhibits oxidative stress and ameliorates autistic-like behaviors in prenatal valproic acid-exposed rats. Neuropharmacology 168:107975. https://doi.org/10.1016/j.neuropharm.2020.107975
Mirza R, Sharma B (2019) Benefits of Fenofibrate in prenatal valproic acid-induced autism spectrum disorder related phenotype in rats. Brain Res Bull 147:36–46. https://doi.org/10.1016/j.brainresbull.2019.02.003
Müller S, Chakrapani BP, Schwegler H, Hofmann HD, Kirsch M (2009) Neurogenesis in the dentate gyrus depends on ciliary neurotrophic factor and signal transducer and activator of transcription 3 signaling. Stem Cells 27:431–441. https://doi.org/10.1634/stemcells.2008-0234
Nordenankar K, Smith-Anttila CJ, Schweizer N, Viereckel T, Birgner C, Mejia-Toiber J, Morales M, Leao RN, Wallén-Mackenzie Å (2015) Increased hippocampal excitability and impaired spatial memory function in mice lacking VGLUT2 selectively in neurons defined by tyrosine hydroxylase promoter activity. Brain Struct Funct 220:2171–2190. https://doi.org/10.1007/s00429-014-0778-9
Okada R, Masubuchi Y, Tanaka T, Nakajima K, Masuda S, Nakamura K, Maronpot RR, Yoshida T, Koyanagi M, Hayashi SM, Shibutani M (2019) Continuous exposure to α-glycosyl isoquercitrin from developmental stage facilitates fear extinction learning in rats. J Funct Foods 55:312–324. https://doi.org/10.1016/j.jff.2019.02.024
Pangrazzi L, Balasco L, Bozzi Y (2020) Natural antioxidants: a novel therapeutic approach to autism spectrum disorders? Antioxidants (Basel) 9:1186. https://doi.org/10.3390/antiox9121186
Pujadas L, Gruart A, Bosch C, Delgado L, Teixeira CM, Rossi D, de Lecea L, Martínez A, Delgado-García JM, Soriano E (2010) Reelin regulates postnatal neurogenesis and enhances spine hypertrophy and long-term potentiation. J Neurosci 30:4636–4649. https://doi.org/10.1523/JNEUROSCI.5284-09.2010
Rodier PM, Ingram JL, Tisdale B, Nelson S, Romano J (1996) Embryological origin for autism: developmental anomalies of the cranial nerve motor nuclei. J Comp Neurol 370:247–261. https://doi.org/10.1002/(SICI)1096-9861(19960624)370:2%3c247::AID-CNE8%3e3.0.CO;2-2
Rosen NE, Lord C, Volkmar FR (2021) The diagnosis of autism: from Kanner to DSM-III to DSM-5 and beyond. J Autism Dev Disord 51:4253–4270. https://doi.org/10.1007/s10803-021-04904-1
Rossignol DA, Frye RE (2014) Evidence linking oxidative stress, mitochondrial dysfunction, and inflammation in the brain of individuals with autism. Front Physiol 5:150. https://doi.org/10.3389/fphys.2014.00150
Santos MS, Foss SM, Park CK, Voglmaier SM (2014) Protein interactions of the vesicular glutamate transporter VGLUT1. PLoS ONE 9:e109824. https://doi.org/10.1371/journal.pone.0109824
Santos SD, Carvalho AL, Caldeira MV, Duarte CB (2009) Regulation of AMPA receptors and synaptic plasticity. Neuroscience 158:105–125. https://doi.org/10.1016/j.neuroscience.2008.02.037
Scharfman H, Goodman J, Macleod A, Phani S, Antonelli C, Croll S (2005) Increased neurogenesis and the ectopic granule cells after intrahippocampal BDNF infusion in adult rats. Exp Neurol 192:348–356. https://doi.org/10.1016/j.expneurol.2004.11.016
Sheppard P, Choleris E, Galea L (2019) Structural plasticity of the hippocampus in response to estrogens in female rodents. Mol Brain 12:22. https://doi.org/10.1186/s13041-019-0442-7
Sibbe M, Kuner E, Althof D, Frotscher M (2015) Stem- and progenitor cell proliferation in the dentate gyrus of the reeler mouse. PLoS ONE 10:e0119643. https://doi.org/10.1371/journal.pone.0119643
Spencer JP (2010) Beyond antioxidants: the cellular and molecular interactions of flavonoids and how these underpin their actions on the brain. Proc Nutr Soc 69:244–260. https://doi.org/10.1017/S0029665110000054
Taliaz D, Stall N, Dar DE, Zangen A (2010) Knockdown of brain-derived neurotrophic factor in specific brain sites precipitates behaviors associated with depression and reduces neurogenesis. Mol Psychiatry 15:80–92. https://doi.org/10.1038/mp.2009.67
Tallent MK (2007) Somatostatin in the dentate gyrus. Prog Brain Res 163:265–284. https://doi.org/10.1016/S0079-6123(07)63016-7
Teixeira CM, Kron MM, Masachs N, Zhang H, Lagace DC, Martinez A, Reillo I, Duan X, Bosch C, Pujadas L, Brunso L, Song H, Eisch AJ, Borrell V, Howell BW, Parent JM, Soriano E (2012) Cell-autonomous inactivation of the reelin pathway impairs adult neurogenesis in the hippocampus. J Neurosci 32:12051–12065. https://doi.org/10.1523/JNEUROSCI.1857-12.2012
von Bohlen und Halbach O (2007) Immunohistological markers for staging neurogenesis in adult hippocampus. Cell Tissue Res 329:409–420. https://doi.org/10.1007/s00441-007-0432-4
Walker DG, Lue LF (2015) Immune phenotypes of microglia in human neurodegenerative disease: challenges to detecting microglial polarization in human brains. Alzheimers Res Ther 7:56. https://doi.org/10.1186/s13195-015-0139-9
Waterhouse EG, An JJ, Orefice LL, Baydyuk M, Liao GY, Zheng K, Lu B, Xu B (2012) BDNF promotes differentiation and maturation of adult-born neurons through GABAergic transmission. J Neurosci 32:14318–14330. https://doi.org/10.1523/JNEUROSCI.0709-12.2012
Wegiel J, Kuchna I, Nowicki K, Imaki H, Wegiel J, Marchi E, Ma SY, Chauhan A, Chauhan V, Bobrowicz TW, de Leon M, Louis LA, Cohen IL, London E, Brown WT, Wisniewski T (2010) The neuropathology of autism: defects of neurogenesis and neuronal migration, and dysplastic changes. Acta Neuropathol 119:755–770. https://doi.org/10.1007/s00401-010-0655-4
Williams RJ, Spencer JP (2012) Flavonoids, cognition, and dementia: actions, mechanisms, and potential therapeutic utility for Alzheimer disease. Free Radic Biol Med 52:35–45. https://doi.org/10.1016/j.freeradbiomed.2011.09.010
Wu H, Wang X, Gao J, Liang S, Hao Y, Sun C, Xia W, Cao Y, Wu L (2017) Fingolimod (FTY720) attenuates social deficits, learning and memory impairments, neuronal loss and neuroinflammation in the rat model of autism. Life Sci 173:43–54. https://doi.org/10.1016/j.lfs.2017.01.012
Yamagata K, Andreasson KI, Kaufmann WE, Barnes CA, Worley PF (1993) Expression of a mitogen-inducible cyclooxygenase in brain neurons: regulation by synaptic activity and glucocorticoids. Neuron 11:371–386. https://doi.org/10.1016/0896-6273(93)90192-t
Yang P, Arnold SA, Habas A, Hetman M, Hagg T (2008) Ciliary neurotrophic factor mediates dopamine D2 receptor-induced CNS neurogenesis in adult mice. J Neurosci 28:2231–2241. https://doi.org/10.1523/JNEUROSCI.3574-07.2008
Zavala AR, Biswas S, Harlan RE, Neisewander JL (2006) Fos and glutamate AMPA receptor subunit coexpression associated with cue-elicited cocaine-seeking behavior in abstinent rats. Neuroscience 145:438–452. https://doi.org/10.1016/j.neuroscience.2006.12.038
Acknowledgements
The authors thank Yayoi Kohno for her technical assistance in preparing the histological specimens. We also thank Jeremy Allen, Ph.D., from Edanz (https://jp.edanz.com/ac) for editing a draft of this manuscript.
Funding
This work was supported by San-Ei Gen F.F.I., Inc. and by a research fund from the Institute of Global Innovation Research, Tokyo University of Agriculture and Technology.
Author information
Authors and Affiliations
Contributions
Kazumi Takashima: Methodology, formal analysis, investigation, data curation, writing—original draft, and visualization. Hiromu Okano: Methodology, formal analysis, investigation, data curation, and writing—review and editing. Ryota Ojiro: Investigation and writing—review and editing. Qian Tang: Investigation and writing—review and editing. Yasunori Takahashi: Investigation and writing—review and editing. Shunsuke Ozawa: Investigation and writing—review and editing. Xinyu Zou: Investigation and writing—review and editing. Mihoko Koyanagi: Resources, writing—review and editing, and project administration. Robert R. Maronpot: Validation and writing—review and editing. Toshinori Yoshida: Investigation and writing—review and editing. Makoto Shibutani: Conceptualization, writing—review and editing, visualization, supervision, and funding acquisition.
Corresponding author
Ethics declarations
Ethics Approval
Animal experiments in the present study were conducted in accordance with the National Institutes of Health guide for the care and use of laboratory animals (NIH Publications No. 8023, revised 1978), and all efforts were made to minimize animal suffering. The experimental procedures were approved by the Animal Care and Use Committee of the Tokyo University of Agriculture and Technology (Approved No.: R2-05 for main study; R03-162 for satellite study).
Competing Interest
Mihoko Koyanagi is employed by a food additive manufacturer whose product lines include AGIQ. Robert R. Maronpot is a scientific consultant at the aforementioned food additive manufacturer. The views and opinions expressed in this article are those of the authors and not necessarily those of their respective employers. Kazumi Takashima, Hiromu Okano, Ryota Ojiro, Qian Tang, Yasunori Takahashi, Shunsuke Ozawa, Xinyu Zou, Toshinori Yoshida, and Makoto Shibutani declare that no conflicts of interest exist.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Takashima, K., Okano, H., Ojiro, R. et al. Continuous Exposure to Alpha-Glycosyl Isoquercitrin from Gestation Ameliorates Disrupted Hippocampal Neurogenesis in Rats Induced by Gestational Injection of Valproic Acid. Neurotox Res 40, 2278–2296 (2022). https://doi.org/10.1007/s12640-022-00574-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12640-022-00574-8