Abstract
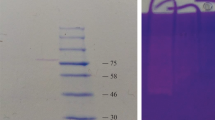
Blastocystis sp. is a commonly encountered gut protozoan with unclear pathogenicity. The presence of proteases in this organism may be related to its potential pathogenicity or other physiological differences. This study aimed to identify the various proteases that may be present in different subtypes (STs) of Blastocystis sp. using azocasein assay and gelatin zymography. In this study, cysteine, serine, aspartic protease, metalloproteases, and unknown proteases were identified in Blastocystis sp. cultures obtained from animal and water samples belonging to ST1–ST5 and ST7. Azocasein assay and gelatin zymography were conducted on different batches of protease extracts, which showed varying results. Cysteine protease was the most commonly encountered. Protease activity in general is not associated with ST or source (animal or water). However, the presence of cysteine protease alone was associated with animal samples, whereas the presence of more than two protease types was associated with water samples. Azocasein assay and gelatin zymography were conducted on different batches of protease extracts, which showed varying results. Protease activity and the types present may change over time. Blastocystis sp. shows high protease diversity, including possible novel types.








Similar content being viewed by others
Availability of data and material
All relevant data are included in this manuscript.
References
Abdel-Hameed DM, Hassanin OM (2011) Protease activity of Blastocystis hominis subtype 3 in symptomatic and asymptomatic patients. Parasitol Res 109:321–327. https://doi.org/10.1007/s00436-011-2259-x
Abou Gamra MM, Elwakil HS, El Deeb HK, Khalifa KE, Abd Elhafiz HE (2011) The potential use of 29 kDa protein as a marker of pathogenicity and diagnosis of symptomatic infections with Blastocystis hominis. Parasitol Res 108:1139–1146. https://doi.org/10.1007/s00436-010-2156-8
Ajjampur SSR, Tan KSW (2016) Pathogenic mechanisms in Blastocystis spp.—Interpreting results from in vitro and in vivo studies. Parasitol Int 65:772–779. https://doi.org/10.1016/j.parint.2016.05.007
Alfellani MA, Jacob AS, Perea NO, Krecek RC, Tanner-Mulla D, Verweij JJ, Levecke B, Tannich E, Clark CG, Stensvold CR (2013a) Diversity and distribution of Blastocystis sp. subtypes in non-human primates. Parasitology 140:966–971. https://doi.org/10.1017/S0031182013000255
Alfellani MA, Stensvold CR, Vidal-Lapiedra A, Onuoha ESU, Fagbenro-Beyioku AF, Clark CG (2013b) Variable geographic distribution of Blastocystis subtypes and its potential implications. Acta Trop 126:11–18. https://doi.org/10.1016/j.actatropica.2012.12.011
Alfellani MA, Taner-Mulla D, Jacob AS, Imeede CA, Yoshikawa H, Stensvold CR, Clark CG (2013c) Genetic diversity of Blastocystis in livestock and zoo animals. Protist 164:497–509. https://doi.org/10.1016/j.protis.2013.05.003
Bernardis FD, Agatensi L, Ross IK, Emerson GW, Lorenzini R, Sullivan PA, Cassone A (1990) Evidence for a role for secreted aspartate proteinase of Candida albicans in vulvovaginal candidiasis. J Infect Dis 161:1276–1283. https://doi.org/10.1093/infdis/161.6.1276
Carruthers VB, Blackman MJ (2005) A new release on life: emerging concepts in proteolysis and parasite invasion. Mol Microbiol 55:1617–1630. https://doi.org/10.1111/j.1365-2958.2005.04483.x
Debnath A, Tashker JS, Sajid M, McKerrow JH (2007) Transcriptional and secretory responses of Entamoeba histolytica to mucins, epithelial cells and bacteria. Int J Parasitol 37:897–906. https://doi.org/10.1016/j.ijpara.2007.01.016
Eme L, Gentekaki E, Curtis B, Archibald JM, Roger AJ (2017) Lateral gene transfer in the adaptation of the anaerobic parasite Blastocystis to the gut. Curr Biol 27:807–820. https://doi.org/10.1016/j.cub.2017.02.003
Hoevers J, Holman P, Logan K, Hommel M, Ashford R, Snowden K (2000) Restriction-fragment-length polymorphism analysis of small-subunit rRNA genes of Blastocystis hominis isolates from geographically diverse human hosts. Parasitol Res 86:57–61. https://doi.org/10.1007/s004360050010
Karamati SA, Mirjalali H, Niyyati M, Yadegar A, Aghdaei HA, Haghighi A, Tabaei SJS (2021) Association of Blastocystis ST6 with higher protease activity among symptomatic subjects. BMC Microbiol 21:1–12. https://doi.org/10.1186/s12866-021-02341-9
Katsarou-Katsari A, Vassalos CM, Tzanetou K, Spanakos G, Papadopoulou C, Vakalis N (2008) Acute urticaria associated with amoeboid forms of Blastocystis sp. subtype 3. Acta Derm Venereol 88:80–81. https://doi.org/10.2340/00015555-0338
Lidell ME, Moncada DM, Chadee K, Hansson GC (2006) Entamoeba histolytica cysteine proteases cleave the MUC2 mucin in its C-terminal domain and dissolve the protective colonic mucus gel. Proc Natl Acad Sci USA 103:9298–9303. https://doi.org/10.1073/pnas.0600623103
McKerrow JH, Caffrey C, Kelly B, Loke P, Sajid M (2006) Proteases in parasitic diseases. Annu Rev Pathol Mech Dis 1:497–536. https://doi.org/10.1146/annurev.pathol.1.110304.100151
McKerrow JH, Rosenthal PJ, Swenerton R, Doyle P (2008) Development of protease inhibitors for protozoan infections. Curr Opin Infect Dis 21:668. https://doi.org/10.1097/QCO.0b013e328315cca9
Mirza H, Tan KS (2009) Blastocystis exhibits inter-and intra-subtype variation in cysteine protease activity. Parasitol Res 104:355–361. https://doi.org/10.1007/s00436-008-1203-1
Mirza H, Wu Z, Teo JDW, Tan KSW (2012) Statin pleiotropy prevents rho kinase-mediated intestinal epithelial barrier compromise induced by Blastocystis cysteine proteases. Cell Microbiol 14:1474–1484. https://doi.org/10.1111/j.1462-5822.2012.01814.x
Miyoshi S, Shinoda S (2000) Microbial metalloproteases and pathogenesis. Microbes Infect 2:91–98. https://doi.org/10.1016/S1286-4579(00)00280-X
Nourrisson C, Wawrzyniak I, Cian A, Livrelli V, Viscogliosi E, Delbac F, Poirier P (2016) On Blastocystis secreted cysteine proteases: a legumain-activated cathepsin B increases paracellular permeability of intestinal Caco-2 cell monolayers. Parasitology 143:1713–1722. https://doi.org/10.1017/S0031182016001396
Puthia MK, Vaithilingam A, Lu J, Tan KS (2005) Degradation of human secretory immunoglobulin A by Blastocystis. Parasitol Res 97:386–389. https://doi.org/10.1007/s00436-005-1461-0
Puthia MK, Lu J, Tan KS (2008) Blastocystis ratti contains cysteine proteases that mediate interleukin-8 response from human intestinal epithelial cells in an NF-κB-dependent manner. Eukaryot Cell 7:435–443. https://doi.org/10.1128/EC.00371-07
Rajamanikam A, Govind SK (2013) Amoebic forms of Blastocystis spp. —evidence for a pathogenic role. Parasit Vectors 6:295. https://doi.org/10.1186/1756-3305-6-295
Rivera WL (2008) Phylogenetic analysis of Blastocystis isolates from animal and human hosts in the Philippines. Vet Parasitol 156:178–182. https://doi.org/10.1016/j.vetpar.2008.06.001
Salter JP, Lim K-C, Hansell E, Hsieh I, McKerrow JH (2000) Schistosome invasion of human skin and degradation of dermal elastin are mediated by a single serine protease. J Biol Chem 275:38667–38673. https://doi.org/10.1074/jbc.M006997200
Sibley LD (2013) The roles of intramembrane proteases in protozoan parasites. Biochim Biophys Acta Biomembr 1828:2908–2915. https://doi.org/10.1016/j.bbamem.2013.04.017
Sio SW, Puthia MK, Lee AS, Lu J, Tan KS (2006) Protease activity of Blastocystis hominis. Parasitol Res 99:126–130. https://doi.org/10.1007/s00436-006-0131-1
Stensvold CR, Alfellani MA, Nørskov-Lauritsen S, Prip K, Victory EL, Maddox C, Nielsen HV, Clark CG (2009) Subtype distribution of Blastocystis isolates from synanthropic and zoo animals and identification of a new subtype. Int J Parasitol 39:473–479. https://doi.org/10.1016/j.ijpara.2008.07.006
Tan T, Suresh K (2006) Predominance of amoeboid forms of Blastocystis hominis in isolates from symptomatic patients. Parasitol Res 98:189–193. https://doi.org/10.1007/s00436-005-0033-7
Toth M, Fridman R (2001) Assessment of gelatinases (MMP-2 and MMP-9 by gelatin zymography. In: Brooks S.A., Schumacher U. (eds) Metastasis Research Protocols Methods in Molecular Medicine, vol 57. Humana Press, https://doi.org/10.1385/1-59259-136-1:163
Wilkesman J, Kurz L (2009) Protease analysis by zymography: a review on techniques and patents. Recent Pat Biotechnol 3:175–184. https://doi.org/10.2174/187220809789389162
Yike I (2011) Fungal proteases and their pathophysiological effects. Mycopathologia 171:299–323. https://doi.org/10.1007/s11046-010-9386-2
Funding
This study was financially supported by the Office of the Vice Chancellor for Research and Development of the University of the Philippines Diliman.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Adao, D.E.V., Rivera, W.L. Variations in active proteases of Blastocystis sp. obtained from water and animal isolates from the Philippines. J Parasit Dis 46, 627–636 (2022). https://doi.org/10.1007/s12639-022-01472-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12639-022-01472-8