Abstract
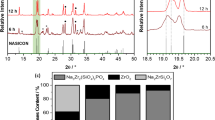
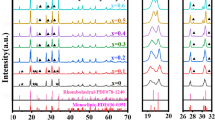
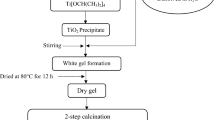
A composition of NASICON (Na3Zr2Si2PO12) was synthesized by the solid-state reaction method using a new compound Na2HPO42H2O. The X-ray diffraction patterns of all samples exhibit monoclinic Na3Zr2Si2PO12 as a major phase with a very small amount of monoclinic-ZrO2. The maximum relative density (97 %) and maximum conductivity is obtained in the samples sintered at 1200 °C (N3) which is slightly higher than β-Al2O3. The activation energy is ∼ 0.20 eV for the N3 sample which is lower than for β-Al2O3. The dilatometeric study and Arrhenius plots confirmed a phase transition of NASICON from monoclinic to rhombohedral. The micro-structural study of the samples done by scanning electron microscopy (SEM) indicated a significant influence of the processing conditions on the microstructures. Raman spectroscopy demonstrated that the sample N3 exhibits minor structural changes compared to other samples.
Similar content being viewed by others
References
Kida T, Morinaga N, Kishi S, An K M, Sim K W, Chae B Y, Kim J K, Ryu B K, Shimanoe K (2011) J Electrochem Acta 22:7484–7490
Zhang H, Zhong T, Sun R, Liang X, Lu G (2014) RSC Adv 4:55334–55340
Alpen U V, Bell M F, Wichelhaus W (1979) Mat Res Bull 14:1317–1322
McEntire BJ, Miller GR, Gordon RS (1979) Proceedings Fifth International Conference, “Sintering and Related Phenomena”, (Notre Dame Indiana)p 18
Anantharamulu N, Velchuri R, Sarojini T, Madhavi K, Prasad G, Vithal M (2009) Ind J Eng Mat Sci 16:347–354
Palomares V, Serras P, Villaluenga I, Hueso B K, Gonz J C, Rojo T (2012) Energy Environ Sci 5:5884–5901
Horwat D, Pierson J F, Billard A (2007) Surf Coat Tech 201:7060–7065
Perthuis H, Colomban Ph (2012) Ceram Int 12:39–52
Yadav P, Bhatnagar MC (2012) Ceram Int 38:1731–1735
Essoumhi A, Favotto C, Mansori M, Satre P (2004) J Solid State Chem 177:4475–4481
Dang HY, Guo XM, Huang YP, Rong JQ (2012) Int J Miner Meta Mat 19:768–773
Ponrouch A, Marchante E, Courty M, Tarascon JM, Palacin MR (2012) Energy Environ Sci 5:8572–8583
Schucker R (2010). WO/2010/135283:PCT/US2010/035198
Wang W, Jiang B, Hua L, Jiao S (2014) J Mater Chem A 2:1341–1345
Paściak G, Mielcarek W, Prociów K, Warycha J (2014) Ceram Int 40:12783–12787
Frank K, Kohler H, Guth U (2008). Ionics 14:363–369
Obata K, Matsushima S (2010) J Ceram Soc Japan 118:213–216
Fuentes RO, Lamas DG, Fernandez de Rapp ME, Figueiredo FM, Frade JR, Marques FMB, Franco JI (2004) Bull Spanish Soc Ceram Glass 43:775–779
Gordon RS, Miller GR, Mcentire BJ, Beck ED, Rasmussen JR (1981) Solid State Ionics 3 (/4):243–248
Boilot JP, Salanie JP, Desplanches G, Potier DL (1979) Mat Res Bull 14:1469–1477
Bayard ML, Barna GG (1978) J Electroanal Chem 91:201–209
Fuentes RO, Figueiredo FM, Marques FMB, Franco JI (2001) J Euro Ceram Soc 21:737–743
Ignaszak A, Pasierb P, Gajerski R, Komornicki S (2006) Mat Sci Poland 24:95–102
Fuentes RO, Figueiredo FM, Marques FMB, Franco JI (2001) Solid State Ionics 140:173–179
Takahashi T, Kuwabare K, Shibata M (1980) Solid State Ionics 1:163–175
Mali A, Petric A (2012) J Euro Ceram Soc 32:1229–1234
Traversa E, Aono H, Sadaoka Y, Montanaro L (2000) J Electroceram 3:261–272
Materials Handbook (2008) A Concise Desktop Reference, 2 nd edition by Cardarelli F, Materials & Electrochemical Research (Corporation Tucson, London Ltd):1340
Chris K, Dyer K, Moseley PT, Ogumi Z, Rand DAJ, Scrosati B, Garche J (2009) Encyclopedia of Electrochemical Power Sources. Elsevier, p 4538
Kang HB, Cho NH (1999) J Mat Sci 34:5005–5013
Hooper A (1980) J Electroanal Chem 109:161–166
Bohnke O, Ronchetti S, Mazza D (1999) Solid State Ionics 122:127–136
Hooper A (1977) J Phys D Appl Phys 10:1487–1496
Hudgens JJ, Brow RK, Tallant DR, Martin SW (1998) J Non Cryst Solids 223:21–31
Syed KM, Pang SF, Zhang Y, Zhang YH (2013) J Chem Phy 138:024901
Palanivel R, Velraj G (2007) Indian J Pure Appl Phys 45:501–508
Marzouk MA, Ouis MA, Hamdy YM (2012) Silicon 4:221–227
McMillan P (1984) Am Mineral 69:622–644
Caster AG, Kowarik S, Schwartzberg AM, Nicolet O, Lim SH, Leone SR (2009) J Raman Spectrosc 40:770–774
Stelling J, Behrens H, Wilke M, Gottlicher J, Aljanabi EC (2011) Geochim Cosmochim Acta 75:3542–3557
Andrade JS, Pinheiro AG, Vasconcelos IF, Sasaki JM (1999) J Phys Conden Matter 11:4451–4460
El-Batal FH, Khalil EM, Hamdy YM, Zidan HM, Aziz MS, Abdelghany AM (2010) Silicon 2:41–47
Marzouk MA, ElBatal HA, ElDin FME (2013) Silicon 5:283–295
Rao GV, Shashikala HD (2014) J Adv Ceram 3:109–116
Chahine A, Ettabirou M, Pascal JL (2004) Mater Lett 58:2776–2778
Fang X, Ray CS, Milkankovic AM, Day DE (2001) J Non Cryst Solids 283:162–172
Froehlich E, Mandeville JS, Weinert CM, Kreplak L, Riahi HAT (2011) Biomacromolecules 12:511–517
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jha, P.K., Pandey, O.P. & Singh, K. Optimization of High Conducting Na3Zr2Si2PO12 Phase by new Phosphate Salt for Solid Electrolyte. Silicon 9, 411–419 (2017). https://doi.org/10.1007/s12633-015-9396-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12633-015-9396-2