Abstract
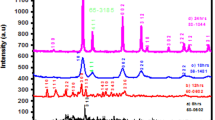
To synthesize pure γ-La2S3 at lower temperature, lanthanide complex La(Et2S2CN)3·phen, containing La-S bond, was chosen as the precursors to decompose. The obtained samples were characterized by X-ray power diffraction (XRD), field-emission scanning electron microscopy (FE-SEM) with an energy dispersive spectrometer and UV-Vis diffuse reflectance spectra. The decomposition mechanism of the lanthanide complex was studied by thermogravimetric analyses (TGA). The results show that the obtained samples are cubic phase particles with sizes among 20–50 nm and the band gap is 2.97 eV, which is bigger than that of its bulk crystal. TG/DTG results indicate that La(Et2S2CN)3·phen decomposed to γ-La2S3 via La4(Et2S2CN)3 as an intermediate product.
Similar content being viewed by others
References
P.D. Mumford and M. Cahay, Dynamic work function shift in cold cathode emitters using current carrying thin films, J. Appl. Phys., 79(1996), p.2176.
O. Eriksson, J. Wills, P.D. Mumford, et al., Electronic structure of the LaS surface and LaS/CdS interface, Phys. Rev. B, 57(1998), p.4067.
L. Tian, T. Ouyang, K.P. Loh, and J.J. Vittal, La2S3 thin films from metal organic chemical vapor deposition of single-source precursor, J. Mater. Chem., 16(2006), p.272.
D.L. Chess, C.A. Chess, J.A. Marks, and W.B. White, Phase equilibria and processing of infrared optical ceramics on the join CaLa2S4-La2S3, J. Ceram. Process. Res., 11(2010), No.4, p.465.
C.I. Merzbacher, D.L. Chess, and W.B. White, Infrared optical properties of calcium lanthanum sulfide, Mater. Lett., 64(2010), No.3, p.334.
P.N. Kumta and S.H. Risbud, Low temperature synthesis of cubic lanthanum sulfide (La2S3) powders, Mater. Sci. Eng. B, 2(1989), No.4, p.281.
S. Roméro, A. Mosset, J.C. Trombe, and P. Macaudière, Low-temperature process of the cubic lanthanide sesquisulfides: remarkable stabilization of the γ-Ce2S3 phase, J. Mater. Chem., 7(1997), p.1541.
K. Tang, C. An, P. Xie, et al., Low-temperature synthesis and characterization of β-La2S3 nanorods, J. Cryst. Growth, 245(2002), No.3–4, p.304.
H. Yuan, J. Zhang, R. Yu, and Q. Su, Synthesis of rare earth sulfides and their UV-vis absorption spectra, J. Rare Earths, 27(2009), No.2, p.308.
X.Z. Fan, S.P. Chen, G. Xie, et al., Regularity of thermochemical properties of ternary complexes RE[(pdtc)3(phen)], Chin. J. Chem., 24(2006), p.1013.
H.X. Li, Y.J. Zhu, M.L. Cheng, et al., Lanthanide chalcogenolate complexes: syntheses, structures and applications in organic chemistry, Coord. Chem. Rev, 250(2006), No.15–16, p.2059.
P.E. Nikolaev and I.G. Vasil’eva, Vapor pressure determination for solid and liquid La2S3 using boiling points, Inorg. Mater., 44(2008), No.12, p.1367.
L.A. Ivanchenko, V.I. Marchenko, Y.B. Paderno, et al., Optical properties of and chemical bonds in lanthanum sulfide of variable composition, Powder Metall. Met. Ceram, 20(1981), No.1, p.53.
S.A. Kutolin, R.N. Samoilova, L.F. Belova, et al., Electronic spectra and physicochemical properties of rare earth metal sesquisulfides, J. Struct. Chem., 13(1972), No.2, p.226.
C.M. Forster and W.B. White, Optical absorption edge in rare earth sesquisulfides, Mater. Res. Bull., 41(2006), No.2, p.448.
Author information
Authors and Affiliations
Corresponding author
Additional information
This work was financially supported by the National High-Tech Research and Development Program of China (No.2009AA03Z431).
Rights and permissions
About this article
Cite this article
Li, Ps., Li, Hy. & Jie, Wq. Preparation and characterization of γ-La2S3 nanoparticles from thermal decomposition. Int J Miner Metall Mater 18, 748–752 (2011). https://doi.org/10.1007/s12613-011-0506-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12613-011-0506-x