Abstract
Objectives
Diet can influence peripheral leukocyte telomere length (LTL), and various micronutrients have been reported to correlate with it. Zinc is known for its antioxidant properties and immunomodulatory effects. However, there are few epidemiological investigations on the relationship between dietary zinc intake and LTL. This study analyzed the association between dietary zinc and LTL and the potential role of inflammation and oxidative stress among them.
Design
Cross-sectional and community-based study.
Setting and Participants
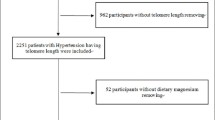
599 participants from rural communities in the Changping suburb of Beijing, China, were recruited.
Measurements
Serum lipid profile, glycosylated hemoglobin (HbA1c), oxidative stress marker, and inflammatory cytokines levels were measured. Detailed dietary data were obtained using a 24 h food recall. LTL was assessed using a real-time PCR assay. Spearman analysis, restricted cubic splines (RCS), and general linear regression models were used to determine the association between dietary zinc intake and LTL. Simple regulatory models were also applied to analyze the role of inflammation and oxidative stress among them.
Results
A total of 482 subjects were ultimately included in this analysis. Spearman analysis showed that dietary zinc intake and zinc intake under energy density were negatively correlated with LTL (r=−0.142 and −0.126, all P <0.05) and positively correlated with tumor necrosis factor-α (TNF-α) (r=0.138 and 0.202, all P <0.05) while only dietary zinc without energy adjustment had a positive correlation with superoxide dismutase (SOD). RCS (P for non-linearity=0.933) and multiple linear regression (B=−0.084, P=0.009) indicated a negative linear association between dietary zinc and LTL. The adjustment of TNF-α rather than SOD could abolish the relationship. The mediation model suggested that the unfavorable effect of dietary zinc on LTL was mediated by TNF-α.
Conclusions
High dietary zinc may correlate with telomere attrition, and TNF-α can act as a mediator in this relationship. In the future, more extensive cohort studies are needed to further explore the relationship between dietary zinc and cellular aging and the specific mechanisms.
Access this article
We’re sorry, something doesn't seem to be working properly.
Please try refreshing the page. If that doesn't work, please contact support so we can address the problem.


Similar content being viewed by others
References
Blackburn EH, Epel ES, Lin J. Human telomere biology: a contributory and interactive factor in aging, disease risks, and protection. Science 2015; 350: 1193–1198. doi: https://doi.org/10.1126/science.aab3389.
Riethmail H. Human telomere structure and biology. Annu Rev Genomics Hum Genet. 2008;9:1–19. doi: https://doi.org/10.1146/annurev.genom.8.021506.172017.
Vaziri H, Dragowska W, Allsopp RC, et al. Evidence for a mitotic clock in human hematopoietic stem cells: loss of telomeric DNA with age. Proc Natl Acad Sci USA. 1994;91:9857–9860. doi:https://doi.org/10.1073/pnas.91.21.9857.
Gao X, Yu X, Zhang C, Wang Y, Sun Y, Sun H, Zhang H, Shi Y, He X. Telomeres and Mitochondrial Metabolism: Implications for Cellular Senescence and Age-related Diseases. Stem Cell Rev Rep. 2022;18: 2315–2327. doi: https://doi.org/10.1007/s12015-022-10370-8
Cheng F, Carroll L, Joglekar MV, Januszewski AS, Wong KK, Hardikar AA, Jenkins AJ, Ma RCW. Diabetes, metabolic disease, and telomere length. Lancet Diabetes Endocrinol. 2021;9:117–126. doi:https://doi.org/10.1016/S2213-8587(20)30365-X.
Pauleck S, Sinnott JA, Zheng YL, Gadalla SM, Viskochil R, Haaland B, Cawthon RM, Hoffmeister A, Hardikar S. Association of Telomere Length with Colorectal Cancer Risk and Prognosis: A Systematic Review and Meta-Analysis. Cancers (Basel) 2023 Feb 11;15(4):1159.
Valera-Gran D, Prieto-Botella D, Hurtado-Pomares M, et al. The Impact of Foods, Nutrients, or Dietary Patterns on Telomere Length in Childhood and Adolescence: A Systematic Review. Nutrients 2022;14:3885. doi:https://doi.org/10.3390/nu14193885
Yu S, Baylin A, Ruiz-Narváez EA. Micro- and Macronutrient Intake in Elderly Costa Ricans: The Costa Rican Longevity and Healthy Aging Study (CRELES). Nutrients 2023;15:1446. doi:https://doi.org/10.3390/nu15061446.
Yu J, Liu H, He S, et al. Dietary Magnesium Intake and Leukocyte Telomere Attrition in Adults: The Regulatory Role of Serum Tumor Necrosis Factor α. Mediators Inflamm. 2020; 2020:7610436. doi:https://doi.org/10.1155/2020/7610436.
Gong H, Yu Q, Yuan M, et al. The Relationship between Dietaiy Copper intake and Telomere Length in Hypertension. J Nutr Health Aging. 2022;26:510–514. doi: https://doi.org/10.1007/s12603-022-1787-7.
Roohani N., Hurrell R., Kelishadi R., et al. Zinc and its importance for human health: An integrative review. J. Res. Med. Sci. 2013;18:144–157. doi.
Maywald M, Rink L. Zinc in Human Health and Infectious Diseases. Biomolecules 2022;12:1748. doi:https://doi.org/10.3390/biom12121748.
Kwon YJ, Lee HS, Park G, et al. Dietary Zinc Intake and All-Cause and Cardiovascular Mortality in Korean Middle-Aged and Older Adults. Nutrients 2023; 15:358. doi:https://doi.org/10.3390/nu15020358.
He P, Li H, Liu M, et al. U-shaped Association Between Dietary Zinc Intake and New-onset Diabetes: A Nationwide Cohort Study in China. J Clin Endocrinol Metab. 2022;107:e815–e824. doi:https://doi.org/10.1210/clinem/dgab636.
Vrieling F, Stienstra R. Obesity and dysregulated innate immune responses: impact of micronutrient deficiencies. Trends Immunol. 2023;44:217–230. doi:https://doi.org/10.1016/j.it.2023.01.003.
A. O’Donovan, M. S. Pantell, E. Puterman et al. Cumulative inflammatory load is associated with short leukocyte telomere length in the Health, Aging and Body Composition Study. PLoS One 2011;6:e19687. doi: https://doi.org/10.1371/journal.pone.0019687.
Armstrong E, Boonekamp J. Does oxidative stress shorten telomeres in vivo? A metaanalysis. Ageing Res Rev. 2023;85:101854.doi:https://doi.org/10.1016/j.arr.2023.101854.
Shi H, Li X, Yu H, Shi W, Lin Y, Zhou Y. Potential effect of dietary zinc intake on telomere length: A cross-sectional study of US adults. Front Nutr. 2022 16; 9: 993 425. doi:https://doi.org/10.3389/fnut.2022.993425.
Lee JY, Shin C, Baik I. Longitudinal associations between micronutrient consumption and leukocyte telomere length. J Hum Nutr Diet. 2017; 30: 236–243. doi: https://doi.org/10.1111/jhn.12403.
Milne E, O’Callaghan N, Ramankutty P, et al. Plasma micronutrient levels and telomere length in children. Nutrition. 2015;31:331–6. doi:https://doi.org/10.1016/j.nut.2014.08.005.
M. Zhou, L. Zhu, X. Cui et al. Influence of diet on leukocyte telomere length, markers of inflammation and oxidative stress in individuals with varied glucose tolerance: a Chinese population study. Nutr J. 2016;15:39. doi: https://doi.org/10.1186/s12937-016-0157-x.
Albertik G, Zimmetp Z. Definition, diagnosis and classification of diabetes mellitus and its complications. Part 1: diagnosis and classification of diabetes mellitus provisional report of a WHO consultation. Diabet Med. 1998;15:539–553. doi:https://doi.org/10.1002/(SICI)1096-9136(199807)15:7<539::AID-DIA668>3.0.CO;2-S.
Cawthon RM. Telomere length measurement by a novel monochrome multiplex quantitative PCR method. Nucleic Acids Res. 2009;37:e21. doi:https://doi.org/10.1093/nar/gkn1027.
Hayes, A. F. (2012). PROCESS: A Versatile Computational Tool for Observed Variable Mediation, Moderation, and Conditional Process Modeling 1. http://www.afhayes.com/public/process2012.pdf.
Faul F, et al. Statistical power analyses using G*Power 264 3.1: Tests for correlation and regression analyses. Behav Res Methods. 2009; 41 265(4):1149–60. doi:https://doi.org/10.3758/BRM.41.4.1149.
Wang Y, Jia XF, Zhang B, et al. Dietary Zinc Intake and Its Association with Metabolic Syndrome Indicators among Chinese Adults: An Analysis of the China Nutritional Transition Cohort Survey 2015. Nutrients 2018;10:572. doi:https://doi.org/10.3390/nu10050572.
Shim JS, Kim KN, Lee JS, et al. Dietary zinc intake and sources among Koreans: findings from the Korea National Health and Nutrition Examination Survey 2016–2019. Nutr Res Pract. 2023;17:257–268.doi:https://doi.org/10.4162/nrp.2023.17.2.257.
Kark JD, Goldberger N, Kimura M, et al. Energy intake and leukocyte telomere length in young adults. Am J Clin Nutr. 2012;95(2):479–87. doi: https://doi.org/10.3945/ajcn.111.024521.
Ceylan MN, Akdas S, Yazihan N. The Effects of Zinc Supplementation on C-Reactive Protein and Inflammatory Cytokines: A Meta-Analysis and Systematical Review. J Interferon Cytokine Res. 2021;41:81–101. doi: https://doi.org/10.1089/jir.2020.0209.
Taneja SK, Mandal R, Girhotra S. Long term excessive Zn-supplementation promotes metabolic syndrome-X in Wistar rats fed sucrose and fat rich semisynthetic diet. Indian J Exp Biol. 2006;44(9):705–18. doi.
Milton AH, Vashum KP, McEvoy M, et al. Prospective Study of Dietary Zinc Intake and Risk of Cardiovascular Disease in Women. Nutrients 2018;10:38. doi: https://doi.org/10.3390/nu10010038.
Drake I, Hindy G, Ericson U, et al. A prospective study of dietary and supplemental zinc intake and risk of type 2 diabetes depending on genetic variation in SLC30A8. Genes Nutr. 2017;12:30. doi: https://doi.org/10.1186/s12263-017-0586-y.
Shi Z, Chu A, Zhen S, et al. Association between dietary zinc intake and mortality among Chinese adults: findings from 10-year follow-up in the Jiangsu Nutrition Study. Eur J Nutr. 2018;57:2839–2846. doi: https://doi.org/10.1007/s00394-017-1551-7.
Zhu L, An P, Zhao W, et al. Low Zinc Alleviates the Progression of Thoracic Aortic Dissection by Inhibiting Inflammation. Nutrients 2023;15:1640. doi:https://doi.org/10.3390/nu15071640.
Li W, Yang X, Ding M, et al. Zinc accumulation aggravates cerebral ischemia/ reperfusion injury by promoting inflammation. Front Cell Neurosci. 2023; 17: 10658 73. doi:https://doi.org/10.3389/fncel.2023.1065873.
Akdas S, Turan B, Durak A, et al. The Relationship Between Metabolic Syndrome Development and Tissue Trace Elements Status and Inflammatory Markers. Biol Trace Elem Res. 2020;198:16–24. doi: https://doi.org/10.1007/s12011-020-02046-6.
Armstrong E, Boonekamp J. Does oxidative stress shorten telomeres in vivo? A meta-analysis. Ageing Res Rev. 2023;85:101854. doi: https://doi.org/10.1016/j.arr.2023.101854.
Yang Q, Yang J, Liu X, et al. Crosstalk Between the Mitochondrial Dynamics and Oxidative Stress in Zinc-induced Cytotoxicity. Biol Trace Elem Res. 2023;201(9): 4419–4428. doi: https://doi.org/10.1007/s12011-022-03504-z. Epub 2022 Nov 29.
Prasad AS, Bao B. Molecular Mechanisms of Zinc as a Pro-Antioxidant Mediator: Clinical Therapeutic Implications. Antioxidants (Basel). 2019;8(6):164. doi: https://doi.org/10.3390/antiox8060164.
Bie YN, Gu P, Chen YT, et al. TZAP plays an inhibitory role in the self-renewal of porcine mesenchymal stromal cells and is implicated the regulation of premature senescence via the p53 pathway. J Transl Med. 2019 Mar 7;17(1):72. doi: https://doi.org/10.1186/s12967-019-1820-8.
Smith S, de Lange T. Tankyrase promotes telomere elongation in human cells. Curr Biol. 2000;10(20):1299–302. doi: https://doi.org/10.1016/s0960-9822(00)00752-1.
Sharif R, Thomas P, Zalewski P, et al. The role of zinc in genomic stability. Mutat Res. 2012;733(1–2):111–21. doi: https://doi.org/10.1016/j.mrfmmm.2011.08.009. Epub 2011 Sep 16.
Trumbo P, Yates AA, Schlicker S, Poos M. Dietary reference intakes: vitamin A, vitamin K, arsenic, boron, chromium, copper, iodine, iron, manganese, molybdenum, nickel, silicon, vanadium, and zinc. J Am Diet Assoc. 2001;101: 294–301. doi: https://doi.org/10.1016/S0002-8223(01)00078-5.
Wang Y, Sun X, Ma H, et al. Iron, Zinc and Copper from Cereal Food Sources and Cognitive Performance in Older Adults in China. Iran J Public Health. 2021; 50(12): 2546–2554. doi: https://doi.org/10.18502/ijph.v50i12.7937.
de Oliveira Otto MC, Alonso A, Lee DH, et al. Dietary intakes of zinc and heme iron from red meat, but not from other sources, are associated with greater risk of metabolic syndrome and cardiovascular disease. J Nutr. 2012;142:526–33. doi:https://doi.org/10.3945/jn.111.149781.
Acknowledgements
We are grateful to all of the participants in this study. The authors’ responsibilities were as follows–BD Xing, HB Zhang, YX Li: designed the research; BD Xing, J Yu, YW Liu, XY Chen, ZY Li, LY He, N Yang: helped to organize the data; SLH: calculated the nutrient related data; BD Xing, J Yu, HB Zhang analyzed the data; BD Xing wrote the paper; LL Xu, F Ping, W Li gave instruction to data analyses: W Li and YX Li reviewed and edited the manuscript. All authors read and approved the final manuscript.
Funding
Funding: This research is supported by National High Level Hospital Clinical Research Funding (2022-PUMCH-B-015) and the Chinese Academy of Medical Sciences Innovation Fund for Medical Sciences (CIFMS,2021-I2M-1-002).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interests: All authors declare that they have no conflict of interests.
Ethical standards: The study was complied with the Declaration of Helsinki and approved by the Ethics Committee of Peking Union Medical College Hospital.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Xing, B., Yu, J., Liu, Y. et al. High Dietary Zinc Intake Is Associated with Shorter Leukocyte Telomere Length, Mediated by Tumor Necrosis Factor-α: A Study of China Adults. J Nutr Health Aging 27, 904–910 (2023). https://doi.org/10.1007/s12603-023-1992-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12603-023-1992-z