Abstract
Objectives
Coffee and tea intake might be associated with psychiatry diseases. However, it is unclear whether the effect of coffee/tea on anxiety and depression depending on the different types of proteins.
Design
This was a cross-sectional study.
Setting
Our datasets were downloaded from online.
Participants
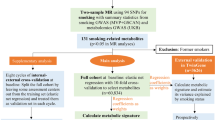
Phenotypic and genotypic data for coffee intake(N=376,196) and tea intake (N=376,078) were derived from UK Biobank. GWAS data of proteins (N=1,537) from neurologically relevant tissues (brain, cerebrospinal fluid (CSF) and plasma) were obtained from a recently published study.
Measurements
Multivariate linear analysis was then used to evaluate the potential interaction effect between coffee/tea intake and proteins polygenetic risk score (PRS) on the risks of anxiety and depression controlling for age, sex, Townsend deprivation index (TDI), smoke, drinking and education level.
Results
34 coffee intake-proteins interactions and 15 tea intake-proteins interactions were observed in anxiety individuals, such as coffee intake-c-Jun interaction (β=0.0169, P=4.131×10−3), coffee intake-Fas interaction (β=−0.0190, P=8.132×10−4), tea intake-sL-Selectin interaction (β=0.0112, P=5.412×10−3) and tea intake-IL-1F6 (β=0.0083, P=4.471×10−2). 25 coffee intake-proteins and 14 tea intake-proteins interactions were observed in depression individuals, including coffee intake- IL-1 sRI (β=0.0171, P=4.888×10−3) and coffee intake-NXPH1 interaction (β=0.0156, P=9.819×10−3), tea intake-COLEC12 interaction (β=0.0127, P=3.280×10−3), and tea intake-Layilin interaction (β=0.0117, P=7.926×10−3).
Conclusions
Our results suggested the important role of multiple proteins in neurologically relevant tissues in the associations between coffee/tea intake and psychiatry diseases, providing entry points to explore the mechanisms underlying anxiety and depression.


Similar content being viewed by others
Availability of data and material: The data that support the findings of this study are available from the corresponding author upon reasonable request.
Abbreviations
- PRS :
-
Polygenetic risk score
- GWAS :
-
Genome-wide association study
- TDI :
-
Townsend deprivation index
- PDEs :
-
phosphodiesterase
- BBB :
-
Blood brain barrier
- AD :
-
Alzheimer’s disease
- RFU :
-
Relative fluorescent units
- pQTLs :
-
Protein quantitative trait loci
- CSF :
-
Cerebrospinal fluid
- QC :
-
Quality control
- GAD-7 :
-
General anxiety disorder
- PHQ-9 :
-
Patient Health Questionnaire
- AP-1 :
-
Activator protein 1
- MAPK :
-
p38 mitogen-activated protein kinase
- EGCG :
-
epigallocatechin-3-gallate
- EGC :
-
epigallocatechin
- ECG :
-
epicatechin-3-gallate
- CGAs :
-
chlorogenic acids
References
Vigo D, Thornicroft G, Atun R. Estimating the true global burden of mental illness. Lancet Psychiatry 2016;3:171–178.doi:https://doi.org/10.1016/S2215-0366(15)00505-2
Zhou Y, Cao Z, Yang M, Xi X, Guo Y, Fang M, Cheng L, Du Y. Comorbid generalized anxiety disorder and its association with quality of life in patients with major depressive disorder. Sci Rep 2017;7:40511.doi:https://doi.org/10.1038/srep40511
Jamal M, Willem Van der Does AJ, Cuijpers P, Penninx BW. Association of smoking and nicotine dependence with severity and course of symptoms in patients with depressive or anxiety disorder. Drug and alcohol dependence 2012;126:138–146. doi:https://doi.org/10.1016/j.drugalcdep.2012.05.001
Harvey SB, Overland S, Hatch SL, Wessely S, Mykletun A, Hotopf M. Exercise and the Prevention of Depression: Results of the HUNT Cohort Study. The American journal of psychiatry 2018;175:28–36.doi:https://doi.org/10.1176/appi.ajp.2017.16111223
Reyes CM, Cornelis MC. Caffeine in the Diet: Country-Level Consumption and Guidelines. Nutrients 2018;10.doi:https://doi.org/10.3390/nu10111772
Grosso G, Micek A, Castellano S, Pajak A, Galvano F. Coffee, tea, caffeine and risk of depression: A systematic review and dose-response meta-analysis of observational studies. Molecular nutrition & food research 2016;60:223–234.doi:https://doi.org/10.1002/mnfr.201500620
Asil E, Yilmaz MV, Yardimci H. Effects of black tea consumption and caffeine intake on depression risk in black tea consumers. African health sciences 2021;21:858–865. doi:https://doi.org/10.4314/ahs.v21i2.47
Kasimay Cakir O, Ellek N, Salehin N, Hamamci R, Keles H, Kayali DG, Akakin D, Yuksel M, Ozbeyli D. Protective effect of low dose caffeine on psychological stress and cognitive function. Physiol Behav 2017;168:1–10.doi:https://doi.org/10.1016/j.physbeh.2016.10.010
A ML, B JAC, C HRLJN, Reviews B. A review of caffeine’s effects on cognitive, physical and occupational performance. 2016;71:294–312. doi: https://doi.org/10.1016/j.neubiorev.2016.09.001
Yao Y, Chen H, Chen L, Ju SY, Yang H, Zeng Y, Gu D, Ng TP. Type of tea consumption and depressive symptoms in Chinese older adults. BMC Geriatr 2021;21:331.doi:https://doi.org/10.1186/s12877-021-02203-z
Bertasi RAO, Humeda Y, Bertasi TGO, Zins Z, Kimsey J, Pujalte G. Caffeine Intake and Mental Health in College Students. Cureus 2021;13:e14313.doi:https://doi.org/10.7759/cureus.14313
Pervin M, Unno K, Takagaki A, Isemura M, Nakamura Y. Function of Green Tea Catechins in the Brain: Epigallocatechin Gallate and its Metabolites. Int J Mol Sci 2019;20:3630.doi:https://doi.org/10.3390/ijms20153630
Zhang C, Lueptow LM, Zhang HT, O’Donnell JM, Xu Y. The Role of Phosphodiesterase-2 in Psychiatric and Neurodegenerative Disorders. Advances in neurobiology 2017;17:307–347.doi:https://doi.org/10.1007/978-3-319-58811-7_12
Ribeiro JA, Sebastiao AM. Caffeine and adenosine. Journal of Alzheimer’s disease: JAD 2010;20 Suppl 1:S3–15.doi:https://doi.org/10.3233/JAD-2010-1379
Cao C, Cirrito JR, Lin X, Wang L, Verges DK, Dickson A, Mamcarz M, Zhang C, Mori T, Arendash GW, Holtzman DM, Potter H. Caffeine suppresses amyloid-beta levels in plasma and brain of Alzheimer’s disease transgenic mice. Journal of Alzheimer’s disease: JAD 2009;17:681–697.doi:https://doi.org/10.3233/JAD-2009-1071
Londzin P, Zamora M, Kakol B, Taborek A, Folwarczna J. Potential of Caffeine in Alzheimer’s Disease-A Review of Experimental Studies. Nutrients 2021;13:537. doi:https://doi.org/10.3390/nu13020537
Chen X, Gawryluk JW, Wagener JF, Ghribi O, Geiger JD. Caffeine blocks disruption of blood brain barrier in a rabbit model of Alzheimer’s disease. J Neuroinflammation 2008;5:12.doi:https://doi.org/10.1186/1742-2094-5-12
Sudlow C, Gallacher J, Allen N, Beral V, Burton P, Danesh J, Downey P, Elliott P, Green J, Landray M, Liu B, Matthews P, Ong G, Pell J, Silman A, Young A, Sprosen T, Peakman T, Collins R. UK biobank: an open access resource for identifying the causes of a wide range of complex diseases of middle and old age. PLoS medicine 2015;12:e1001779.doi:https://doi.org/10.1371/journal.pmed.1001779
Yang C, Farias FHG, Ibanez L, Suhy A, Sadler B, Fernandez MV, Wang F, Bradley JL, Eiffert B, Bahena JA, Budde JP, Li Z, Dube U, Sung YJ, Mihindukulasuriya KA, Morris JC, Fagan AM, Perrin RJ, Benitez BA, Rhinn H, Harari O, Cruchaga C. Genomic atlas of the proteome from brain, CSF and plasma prioritizes proteins implicated in neurological disorders. Nat Neurosci 2021;24:1302–1312.doi:https://doi.org/10.1038/s41593-021-00886-6
Gold L, Ayers D, Bertino J, Bock C, Bock A, Brody EN, Carter J, Dalby AB, Eaton BE, Fitzwater T, Flather D, Forbes A, Foreman T, Fowler C, Gawande B, Goss M, Gunn M, Gupta S, Halladay D, Heil J, Heilig J, Hicke B, Husar G, Janjic N, Jarvis T, Jennings S, Katilius E, Keeney TR, Kim N, Koch TH, Kraemer S, Kroiss L, Le N, Levine D, Lindsey W, Lollo B, Mayfield W, Mehan M, Mehler R, Nelson SK, Nelson M, Nieuwlandt D, Nikrad M, Ochsner U, Ostroff RM, Otis M, Parker T, Pietrasiewicz S, Resnicow DI, Rohloff J, Sanders G, Sattin S, Schneider D, Singer B, Stanton M, Sterkel A, Stewart A, Stratford S, Vaught JD, Vrkljan M, Walker JJ, Watrobka M, Waugh S, Weiss A, Wilcox SK, Wolfson A, Wolk SK, Zhang C, Zichi D. Aptamer-based multiplexed proteomic technology for biomarker discovery. PloS one 2010;5:e15004.doi:https://doi.org/10.1371/journal.pone.0015004
Yang C, Farias FHG, Ibanez L, Suhy A, Sadler B, Fernandez MV, Wang F, Bradley JL, Eiffert B, Bahena JA, Budde JP, Li Z, Dube U, Sung YJ, Mihindukulasuriya KA, Morris JC, Fagan AM, Perrin RJ, Benitez BA, Rhinn H, Harari O, Cruchaga C. Genomic atlas of the proteome from brain, CSF and plasma prioritizes proteins implicated in neurological disorders. Nature Neuroscience 2021;24:1302–1312. doi:https://doi.org/10.1038/s41593-021-00886-6
Kroenke K, Spitzer RL, Williams J, L?We BJGHP. The Patient Health Questionnaire Somatic, Anxiety, and Depressive Symptom Scales: a systematic review. 2010;32:345–359
Davis K, Cullen B, Adams M, Brailean A, Hotopf MJIJoMiPR. Indicators of mental disorders in UK Biobank—A comparison of approaches. 2019;28. doi: https://doi.org/10.1002/mpr.1796
Gonçalves PB, Sodero ACR, Cordeiro Y. Green Tea Epigallocatechin-3-gallate (EGCG) Targeting Protein Misfolding in Drug Discovery for Neurodegenerative Diseases. 2021;11:767.doi: https://doi.org/10.3390/biom11050767
Lee B, Shim I, Lee H, Hahm DH. Effects of Epigallocatechin Gallate on Behavioral and Cognitive Impairments, Hypothalamic-Pituitary-Adrenal Axis Dysfunction, and Alternations in Hippocampal BDNF Expression Under Single Prolonged Stress. Journal of medicinal food 2018;21:979–989.doi:https://doi.org/10.1089/jmf.2017.4161
Wang J, Li P, Qin T, Sun D, Zhao X, Zhang B. Protective effect of epigallocatechin-3-gallate against neuroinflammation and anxiety-like behavior in a rat model of myocardial infarction. Brain and behavior 2020;10:e01633.doi:https://doi.org/10.1002/brb3.1633
Wakabayashi C, Numakawa T, Ninomiya M, Chiba S, Kunugi H. Behavioral and molecular evidence for psychotropic effects in L-theanine. Psychopharmacology (Berl) 2012;219:1099–1109.doi:https://doi.org/10.1007/s00213-011-2440-z
Shen M, Yang Y, Wu Y, Zhang B, Wu H, Wang L, Tang H, Chen J. L-theanine ameliorate depressive-like behavior in a chronic unpredictable mild stress rat model via modulating the monoamine levels in limbic-cortical-striatal-pallidal-thalamic-circuit related brain regions. Phytotherapy research: PTR 2019;33:412–421. doi:https://doi.org/10.1002/ptr.6237
Arendash GW, Schleif W, Rezai-Zadeh K, Jackson EK, Zacharia LC, Cracchiolo JR, Shippy D, Tan J. Caffeine protects Alzheimer’s mice against cognitive impairment and reduces brain beta-amyloid production. Neuroscience 2006;142:941–952.doi:https://doi.org/10.1016/j.neuroscience.2006.07.021
Cho ES, Jang YJ, Hwang MK, Kang NJ, Lee KW, Lee HJ. Attenuation of oxidative neuronal cell death by coffee phenolic phytochemicals. Mutat Res 2009;661:18–24. doi:https://doi.org/10.1016/j.mrfmmm.2008.10.021
Liang N, Kitts DD. Role of Chlorogenic Acids in Controlling Oxidative and Inflammatory Stress Conditions. Nutrients 2015;8.doi:https://doi.org/10.3390/nu8010016
Stefanello N, Schmatz R, Pereira LB, Rubin MA, da Rocha JB, Facco G, Pereira ME, Mazzanti CM, Passamonti S, Rodrigues MV, Carvalho FB, da Rosa MM, Gutierres JM, Cardoso AM, Morsch VM, Schetinger MR. Effects of chlorogenic acid, caffeine, and coffee on behavioral and biochemical parameters of diabetic rats. Molecular and cellular biochemistry 2014;388:277–286.doi:https://doi.org/10.1007/s11010-013-1919-9
Goshen I, Kreisel T, Ben-Menachem-Zidon O, Licht T, Weidenfeld J, Ben-Hur T, Yirmiya R. Brain interleukin-1 mediates chronic stress-induced depression in mice via adrenocortical activation and hippocampal neurogenesis suppression. Mol Psychiatry 2008;13:717–728.doi:https://doi.org/10.1038/sj.mp.4002055
Bouayed J, Rammal H, Soulimani R. Oxidative stress and anxiety: relationship and cellular pathways. Oxid Med Cell Longev 2009;2:63–67.doi:https://doi.org/10.4161/oxim.2.2.7944
Felger JC. Imaging the Role of Inflammation in Mood and Anxiety-related Disorders. Curr Neuropharmacol 2018;16:533–558.doi:https://doi.org/10.2174/1570159X15666171123201142
Felger JC, Lotrich FE. Inflammatory cytokines in depression: neurobiological mechanisms and therapeutic implications. Neuroscience 2013;246:199–229. doi:https://doi.org/10.1016/j.neuroscience.2013.04.060
Muqaku B, Tahir A, Klepeisz P, Bileck A, Kreutz D, Mayer RL, Meier SM, Gerner M, Schmetterer K, Gerner C. Coffee consumption modulates inflammatory processes in an individual fashion. Molecular nutrition & food research 2016;60:2529–2541. doi:https://doi.org/10.1002/mnfr.201600328
Lagha AB, Grenier D. Tea polyphenols inhibit the activation of NF-kappaB and the secretion of cytokines and matrix metalloproteinases by macrophages stimulated with Fusobacterium nucleatum. Sci Rep 2016;6:34520.doi:https://doi.org/10.1038/srep34520
Liu Y, Ho RC, Mak A. The role of interleukin (IL)-17 in anxiety and depression of patients with rheumatoid arthritis. International journal of rheumatic diseases 2012;15:183–187.doi:https://doi.org/10.1111/j.1756-185X.2011.01673.x
O’Donovan A, Hughes BM, Slavich GM, Lynch L, Cronin MT, O’Farrelly C, Malone KM. Clinical anxiety, cortisol and interleukin-6: evidence for specificity in emotion-biology relationships. Brain Behav Immun 2010;24:1074–1077.doi:https://doi.org/10.1016/j.bbi.2010.03.003
Milaneschi Y, Kappelmann N, Ye Z, Lamers F, Moser S, Jones PB, Burgess S, Penninx B, Khandaker GM. Association of inflammation with depression and anxiety: evidence for symptom-specificity and potential causality from UK Biobank and NESDA cohorts. Mol Psychiatry 2021;26:7393–7402.doi:https://doi.org/10.1038/s41380-021-01188-w
McKim DB, Weber MD, Niraula A, Sawicki CM, Liu X, Jarrett BL, Ramirez-Chan K, Wang Y, Roeth RM, Sucaldito AD, Sobol CG, Quan N, Sheridan JF, Godbout JP. Microglial recruitment of IL-1beta-producing monocytes to brain endothelium causes stress-induced anxiety. Mol Psychiatry 2018;23:1421–1431.doi:https://doi.org/10.1038/mp.2017.64
Yu WR, Fehlings MG. Fas/FasL-mediated apoptosis and inflammation are key features of acute human spinal cord injury: implications for translational, clinical application. Acta neuropathology 2011;122:747–761.doi:https://doi.org/10.1007/s00401-011-0882-3
Park DR, Thomsen AR, Frevert CW, Pham U, Skerrett SJ, Kiener PA, Liles WC. Fas (CD95) induces proinflammatory cytokine responses by human monocytes and monocyte-derived macrophages. Journal of immunology (Baltimore, Md: 1950) 2003;170:6209–6216.doi:https://doi.org/10.4049/jimmunol.170.12.6209
Martínez M, Fernández-Vivancos E, Frank A, De la Fuente M, Hernanz A. Increased cerebrospinal fluid Fas (Apo-1) levels in Alzheimer’s disease. Brain Research 2000;869:216–219.doi:https://doi.org/10.1016/s0006-8993(00)02363-5
Hess J, Angel P, Schorpp-Kistner M. AP-1 subunits: quarrel and harmony among siblings. J Cell Sci 2004;117:5965–5973.doi:https://doi.org/10.1242/jcs.01589
Raivich G, Behrens A. Role of the AP-1 transcription factor c-Jun in developing, adult and injured brain. Prog Neurobiol 2006;78:347–363.doi:https://doi.org/10.1016/j.pneurobio.2006.03.006
Ma HP. Relationship Between c-Jun N-terminal Kinase and Depression. 2020 INTERNATIONAL CONFERENCE ON ENERGY, ENVIRONMENT AND BIOENGINEERING (ICEEB 2020)2020.
Marcus DL, Strafaci JA, Miller DC, Masia S, Thomas CG, Rosman J, Hussain S, Freedman ML. Quantitative neuronal c-fos and c-jun expression in Alzheimer’s disease. Neurobiol Aging 1998;19:393–400.doi:https://doi.org/10.1016/s0197-4580(98)00077-3
de Melo Pereira GV, de Carvalho Neto DP, Magalhaes Junior AI, do Prado FG, Pagnoncelli MGB, Karp SG, Soccol CR. Chemical composition and health properties of coffee and coffee by-products. Advances in food and nutrition research 2020;91:65–96.doi:https://doi.org/10.1016/bs.afnr.2019.10.002
Lim DW, Han T, Jung J, Song Y, Um MY, Yoon M, Kim YT, Cho S, Kim IH, Han D, Lee C, Lee J. Chlorogenic Acid from Hawthorn Berry (Crataegus pinnatifida Fruit) Prevents Stress Hormone-Induced Depressive Behavior, through Monoamine Oxidase B-Reactive Oxygen Species Signaling in Hippocampal Astrocytes of Mice. Molecular nutrition & food research 2018:e1800029.doi:https://doi.org/10.1002/mnfr.201800029
Ruusunen A, Lehto SM, Tolmunen T, Mursu J, Kaplan GA, Voutilainen S. Coffee, tea and caffeine intake and the risk of severe depression in middle-aged Finnish men: the Kuopio Ischaemic Heart Disease Risk Factor Study. Public health nutrition 2010;13:1215–1220.doi:https://doi.org/10.1017/S1368980010000509
Chei CL, Loh JK, Soh A, Yuan JM, Koh WP. Coffee, tea, caffeine, and risk of hypertension: The Singapore Chinese Health Study. Eur J Nutr 2018;57:1333–1342. doi:https://doi.org/10.1007/s00394-017-1412-4
da Silva Pinto M. Tea: A new perspective on health benefits. Food Research International 2013;53:558–567.doi:https://doi.org/10.1016/j.foodres.2013.01.038
Leung LK, Su Y, Chen R, Zhang Z, Huang Y, Chen ZY. Theaflavins in black tea and catechins in green tea are equally effective antioxidants. The Journal of nutrition 2001;131:2248–2251.doi:https://doi.org/10.1093/jn/131.9.2248
Bastianetto S, Yao ZX, Papadopoulos V, Quirion R. Neuroprotective effects of green and black teas and their catechin gallate esters against beta-amyloid-induced toxicity. The European journal of neuroscience 2006;23:55–64.doi:https://doi.org/10.1111/j.1460-9568.2005.04532.x
Lorenz M, Urban J, Engelhardt U, Baumann G, Stangl K, Stangl V. Green and black tea are equally potent stimuli of NO production and vasodilation: new insights into tea ingredients involved. Basic Res Cardiol 2009;104:100–110.doi:https://doi.org/10.1007/s00395-008-0759-3
Lee KW, Lee HJ, Lee CY. Antioxidant activity of black tea vs. green tea. The Journal of nutrition 2002;132:785; author reply 786.doi:https://doi.org/10.1093/jn/132.4.785
Ng TP, Feng L, Niti M, Kua EH, Yap KB. Tea consumption and cognitive impairment and decline in older Chinese adults. Am J Clin Nutr 2008;88:224–231.doi:https://doi.org/10.1093/ajcn/88.1.224
Noguchi-Shinohara M, Yuki S, Dohmoto C, Ikeda Y, Samuraki M, Iwasa K, Yokogawa M, Asai K, Komai K, Nakamura H, Yamada M. Consumption of green tea, but not black tea or coffee, is associated with reduced risk of cognitive decline. PloS one 2014;9:e96013.doi:https://doi.org/10.1371/journal.pone.0096013
Acknowledgements
None.
Funding
Funding: This work was supported by the National Natural Scientific Foundation of China [81922059]; the Natural Science Basic Research Plan in Shaanxi Province of China [2021JCW-08].
Author information
Authors and Affiliations
Contributions
Authors’ contributions: Conceptualization, Jing Ye, Shiqiang Cheng and Yan Wen; Data curation, Feng Zhang; Funding acquisition, Feng Zhang; Methodology, Bolun Cheng, Yan Wen, Yumeng Jia, Peilin Meng, Xuena Yang, Chuyu Pan, Yujing Chen; Writing — original draft, Li Liu, Xin Qi; Writing - review & editing, Huijie Zhang, Zhen Zhang, Jingxi Zhang, Chune Li, Bolun Cheng, Yao Yao.
Corresponding author
Ethics declarations
Ethics approval and consent to participate Ethical approval of UK Biobank was granted by the National Health Service National Research Ethics Service (reference 11/ NW/0382).
Ethical standrads Our experiments comply with the current laws of the China. And Our data was downloaded from online.
Additional information
Consent for publication: Not applicable. Our data was downloaded from online.
Competing interests: The authors report no financial interests or potential conflicts of interest.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Liu, L., Cheng, B., Ye, J. et al. Understanding the Complex Interactions between Coffee, Tea Intake and Neurologically Relevant Tissues Proteins in the Development of Anxiety and Depression. J Nutr Health Aging 26, 1070–1077 (2022). https://doi.org/10.1007/s12603-022-1869-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12603-022-1869-6