Abstract
Objectives
The study aimed to evaluate the association of dietary iron intake with incident dementia and brain iron deposition.
Design/Setting/Participants
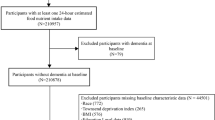
We included dementia-free participants from the UK Biobank who completed at least one 24-hour dietary recall at study baseline (2009–2012) and were followed up to 2021. Incident dementia was determined through linkage to medical records and death registries. Brain MRI was conducted in a subgroup of participants since 2014, with T2* measurements being used as indicators of brain iron deposition.
Measurements
Cox proportional hazard models were used to assess the associations of high (top quintile) and low (bottom quintile) versus medium (quintile 2 to 4) level of dietary iron intake with incident dementia, respectively. Linear regression was applied to assess the relations between dietary iron intake and brain T2* measurements.
Results
During follow-up (mean = 9.5 years), a total of 1,454 participants (650 women and 804 men) developed dementia among 191,694 participants (55.0% female; mean age, 56.2 years). When adjusted for sociodemographic, lifestyle, and other dietary factors, participants with low dietary iron intake (< 10.05 mg/day) had a significantly higher dementia risk (hazard ratio [HR], 1.50, 95% confidence interval [CI], 1.19–1.89), while the relation for high intake (>16.92 mg/day) was non-significant (HR, 1.16, 95% CI, 0.92–1.46). A significant gender difference (P-interaction < 0.001) was observed, with a U-shaped association in male participants (HR for low vs. medium, 1.56, 95% CI, 1.14–2.13; HR for high vs. medium, 1.39, 95% CI, 1.03–1.88; P-nonlinearity < 0.001) and no significant association in females, regardless of their menopause status. In general, dietary iron intake was not related to T2* measurements of iron deposition in most brain regions.
Conclusion
Our findings suggested a U-shape relationship between dietary iron intake and risk of dementia among males, but not females.

Similar content being viewed by others
Data Availability Statement
The UK Biobank is an open-access resource that all bona fide researchers can use for approved research by registering and applying at http://www.ukbiobank.ac.uk/register-apply/. Data used in this study are accessible by request to the UK Biobank. Application ID was 55005.
References
[Global trends in the prevalence of dementia]. Nihon Ronen Igakkai Zasshi. 2018;55(4):547–552. doi:https://doi.org/10.3143/geriatrics.55.547
Baumgart M, Snyder HM, Carrillo MC, Fazio S, Kim H, Johns H. Summary of the evidence on modifiable risk factors for cognitive decline and dementia: A population-based perspective. Alzheimers Dement. Jun 2015;11(6):718–26. doi:https://doi.org/10.1016/j.jalz.2015.05.016
World Health Organization Risk Reduction of Cognitive Decline and Dementia. [(accessed on 14 March 2022)];. Available online: https://wwww.hoint/publications/i/item/risk-reduction-of-cognitive-decline-and-dementia.
Norton S, Matthews FE, Barnes DE, Yaffe K, Brayne C. Potential for primary prevention of Alzheimer’s disease: an analysis of population-based data. Lancet Neurol. Aug 2014;13(8):788–94. doi:https://doi.org/10.1016/S1474-4422(14)70136-X
Gil Martinez V, Avedillo Salas A, Santander Ballestin S. Vitamin Supplementation and Dementia: A Systematic Review. Nutrients. Feb 28 2022;14(5)doi:https://doi.org/10.3390/nu14051033
Prado EL, Dewey KG. Nutrition and brain development in early life. Nutr Rev. Apr 2014;72(4):267–84. doi:https://doi.org/10.1111/nure.12102
Georgieff MK. The role of iron in neurodevelopment: fetal iron deficiency and the developing hippocampus. Biochem Soc Trans. Dec 2008;36(Pt 6):1267–71. doi:https://doi.org/10.1042/BST0361267
McCann S, Perapoch Amado M, Moore SE. The Role of Iron in Brain Development: A Systematic Review. Nutrients. Jul 5 2020;12(7)doi:https://doi.org/10.3390/nu12072001
Cheng R, Dhorajia VV, Kim J, Kim Y. Mitochondrial iron metabolism and neurodegenerative diseases. Neurotoxicology. Jan 2022;88:88–101. doi:https://doi.org/10.1016/j.neuro.2021.11.003
Salvador GA. Iron in neuronal function and dysfunction. Biofactors. Mar–Apr 2010;36(2):103–10. doi:https://doi.org/10.1002/biof.80
Ozawa M, Shipley M, Kivimaki M, Singh-Manoux A, Brunner EJ. Dietary pattern, inflammation and cognitive decline: The Whitehall II prospective cohort study. Clinical nutrition (Edinburgh, Scotland). Apr 2017;36(2):506–512. doi:https://doi.org/10.1016/j.clnu.2016.01.013
Takeuchi H, Kawashima R. Diet and Dementia: A Prospective Study. Nutrients. Dec 16 2021;13(12)doi:https://doi.org/10.3390/nu13124500
Shi Z, Li M, Wang Y, Liu J, El-Obeid T. High iron intake is associated with poor cognition among Chinese old adults and varied by weight status-a 15-y longitudinal study in 4852 adults. Am J Clin Nutr. Jan 1 2019;109(1):109–116. doi:https://doi.org/10.1093/ajcn/nqy254
Cherbuin N, Kumar R, Sachdev PS, Anstey KJ. Dietary Mineral Intake and Risk of Mild Cognitive Impairment: The PATH through Life Project. Front Aging Neurosci. 2014;6:4. doi:https://doi.org/10.3389/fnagi.2014.00004
Hernandez Mdel C, Allan J, Glatz A, et al. Exploratory analysis of dietary intake and brain iron accumulation detected using magnetic resonance imaging in older individuals: the Lothian Birth Cohort 1936. J Nutr Health Aging. Jan 2015;19(1):64–9. doi:https://doi.org/10.1007/s12603-014-0523-3
Hagemeier J, Tong O, Dwyer MG, Schweser F, Ramanathan M, Zivadinov R. Effects of diet on brain iron levels among healthy individuals: an MRI pilot study. Neurobiol Aging. Apr 2015;36(4):1678–1685. doi:https://doi.org/10.1016/j.neurobiolaging.2015.01.010
Bycroft C, Freeman C, Petkova D, et al. The UK Biobank resource with deep phenotyping and genomic data. Nature. Oct 2018;562(7726):203–209. doi:https://doi.org/10.1038/s41586-018-0579-z
Anderson JJ, Gray SR, Welsh P, et al. The associations of sugar-sweetened, artificially sweetened and naturally sweet juices with all-cause mortality in 198,285 UK Biobank participants: a prospective cohort study. BMC Med. Apr 24 2020;18(1):97. doi:https://doi.org/10.1186/s12916-020-01554-5
Greenwood DC, Hardie LJ, Frost GS, et al. Validation of the Oxford WebQ Online 24-Hour Dietary Questionnaire Using Biomarkers. Am J Epidemiol. Oct 1 2019;188(10):1858–1867. doi:https://doi.org/10.1093/aje/kwz165
Galante J, Adamska L, Young A, et al. The acceptability of repeat Internet-based hybrid diet assessment of previous 24-h dietary intake: administration of the Oxford WebQ in UK Biobank. The British journal of nutrition. Feb 28 2016;115(4):681–6. doi:https://doi.org/10.1017/S0007114515004821
Carter JL, Lewington S, Piernas C, et al. Reproducibility of dietary intakes of macronutrients, specific food groups, and dietary patterns in 211 050 adults in the UK Biobank study. J Nutr Sci. 2019;8:e34. doi:https://doi.org/10.1017/jns.2019.31
Alfaro-Almagro F, Jenkinson M, Bangerter NK, et al. Image processing and Quality Control for the first 10,000 brain imaging datasets from UK Biobank. Neuroimage. Feb 1 2018;166:400–424. doi:https://doi.org/10.1016/j.neuroimage.2017.10.034
Atkins JL, Pilling LC, Heales CJ, et al. Hemochromatosis Mutations, Brain Iron Imaging, and Dementia in the UK Biobank Cohort. J Alzheimers Dis. 2021;79(3):1203–1211. doi:https://doi.org/10.3233/JAD-201080
Etemadi A, Sinha R, Ward MH, et al. Mortality from different causes associated with meat, heme iron, nitrates, and nitrites in the NIH-AARP Diet and Health Study: population based cohort study. BMJ. May 9 2017;357:j1957. doi:https://doi.org/10.1136/bmj.j1957
Lee DH, Folsom AR, Jacobs DR, Jr. Iron, zinc, and alcohol consumption and mortality from cardiovascular diseases: the Iowa Women’s Health Study. Am J Clin Nutr. Apr 2005;81(4):787–91. doi:https://doi.org/10.1093/ajcn/81.4.787
Zaccardi F, Davies MJ, Khunti K, Yates T. Comparative Relevance of Physical Fitness and Adiposity on Life Expectancy: A UK Biobank Observational Study. Mayo Clin Proc. Jun 2019;94(6):985–994. doi:https://doi.org/10.1016/j.mayocp.2018.10.029
Liu B, Young H, Crowe FL, et al. Development and evaluation of the Oxford WebQ, a low-cost, web-based method for assessment of previous 24 h dietary intakes in large-scale prospective studies. Public Health Nutr. Nov 2011;14(11):1998–2005. doi:https://doi.org/10.1017/S1368980011000942
Chiuve SE, Fung TT, Rimm EB, et al. Alternative dietary indices both strongly predict risk of chronic disease. The Journal of nutrition. Jun 2012;142(6):1009–18. doi:https://doi.org/10.3945/jn.111.157222
Harshfield EL, Pennells L, Schwartz JE, et al. Association Between Depressive Symptoms and Incident Cardiovascular Diseases. Jama. Dec 15 2020;324(23):2396–2405. doi:https://doi.org/10.1001/jama.2020.23068
Remontet L, Uhry Z, Bossard N, et al. Flexible and structured survival model for a simultaneous estimation of non-linear and non-proportional effects and complex interactions between continuous variables: Performance of this multidimensional penalized spline approach in net survival trend analysis. Stat Methods Med Res. Aug 2019;28(8):2368–2384. doi:https://doi.org/10.1177/0962280218779408
Zhang H, Greenwood DC, Risch HA, Bunce D, Hardie LJ, Cade JE. Meat consumption and risk of incident dementia: cohort study of 493,888 UK Biobank participants. Am J Clin Nutr. Jul 1 2021;114(1):175–184. doi:https://doi.org/10.1093/ajcn/nqab028
Hosking DE, Ayton S, Beckett N, Booth A, Peters R. More evidence is needed. Iron, incident cognitive decline and dementia: a systematic review. Ther Adv Chronic Dis. 2018;9(12):241–256. doi:https://doi.org/10.1177/2040622318788485
Scarmeas N, Anastasiou CA, Yannakoulia M. Nutrition and prevention of cognitive impairment. Lancet Neurol. Nov 2018;17(11):1006–1015. doi:https://doi.org/10.1016/S1474-4422(18)30338-7
McGrattan AM, McGuinness B, McKinley MC, et al. Diet and Inflammation in Cognitive Ageing and Alzheimer’s Disease. Curr Nutr Rep. Jun 2019;8(2):53–65. doi:https://doi.org/10.1007/s13668-019-0271-4
Shi Z, El-Obeid T, Li M, Xu X, Liu J. Iron-related dietary pattern increases the risk of poor cognition. Nutr J. Aug 29 2019;18(1):48. doi:https://doi.org/10.1186/s12937-019-0476-9
Hong CT, Hsieh YC, Liu HY, Chiou HY, Chien LN. Association Between Anemia and Dementia: A Nationwide, Populationbased Cohort Study in Taiwan. Curr Alzheimer Res. 2020;17(2):196–204. doi:https://doi.org/10.2174/1567205017666200317101516
Castellani RJ, Moreira PI, Perry G, Zhu X. The role of iron as a mediator of oxidative stress in Alzheimer disease. Biofactors. Mar-Apr 2012;38(2):133–8. doi:https://doi.org/10.1002/biof.1010
Sobotka TJ, Whittaker P, Sobotka JM, et al. Neurobehavioral dysfunctions associated with dietary iron overload. Physiol Behav. Feb 1996;59(2):213–9. doi:https://doi.org/10.1016/0031-9384(95)02030-6
Ceylan H, Budak H, Kocpinar EF, Baltaci NG, Erdogan O. Examining the link between dose-dependent dietary iron intake and Alzheimer’s disease through oxidative stress in the rat cortex. J Trace Elem Med Biol. Dec 2019;56:198–206. doi:https://doi.org/10.1016/j.jtemb.2019.09.002
Chen M, Xu E, Zeng C, Zhu W, Zheng J, Chen H. High Dietary Iron Has a Greater Impact on Brain Iron Homeostasis and Cognitive Function in Old Compared with Young C57BL/6J Male Mice. The Journal of nutrition. Sep 4 2021;151(9):2835–2842. doi:https://doi.org/10.1093/jn/nxab189
Grubic Kezele T, Curko-Cofek B. Age-Related Changes and Sex-Related Differences in Brain Iron Metabolism. Nutrients. Aug 27 2020;12(9)doi:https://doi.org/10.3390/nu12092601
Kung WM, Yuan SP, Lin MS, et al. Anemia and the Risk of Cognitive Impairment: An Updated Systematic Review and Meta-Analysis. Brain Sci. Jun 11 2021;11(6) doi:https://doi.org/10.3390/brainsci11060777
Rizzo MR, Fasano R, Paolisso G. Adiponectin and Cognitive Decline. Int J Mol Sci. Mar 16 2020;21(6)doi:https://doi.org/10.3390/ijms21062010
Hogervorst E, Yaffe K, Richards M, Huppert FA. Hormone replacement therapy to maintain cognitive function in women with dementia. Cochrane Database Syst Rev. Jan 21 2009;(1):CD003799. doi:https://doi.org/10.1002/14651858.CD003799.pub2
Simpkins JW, Perez E, Wang X, Yang S, Wen Y, Singh M. The potential for estrogens in preventing Alzheimer’s disease and vascular dementia. Ther Adv Neurol Disord. Jan 2009;2(1):31–49. doi:https://doi.org/10.1177/1756285608100427
Savica R, Grossardt BR, Bower JH, Ahlskog JE, Rocca WA. Risk factors for Parkinson’s disease may differ in men and women: an exploratory study. Horm Behav. Feb 2013;63(2):308–14. doi:https://doi.org/10.1016/j.yhbeh.2012.05.013
Chen TY, Lin CL, Wang LF, Tsai KL, Lin JY, Hsu C. Targeting GPER1 to suppress autophagy as a male-specific therapeutic strategy for iron-induced striatal injury. Sci Rep. Apr 30 2019;9(1):6661. doi:https://doi.org/10.1038/s41598-019-43244-0
Burger A, Kotze MJ, Stein DJ, Janse van Rensburg S, Howells FM. The relationship between measurement of in vivo brain glutamate and markers of iron metabolism: A proton magnetic resonance spectroscopy study in healthy adults. Eur J Neurosci. Feb 2020;51(4):984–990. doi:https://doi.org/10.1111/ejn.14583
Beam CR, Kaneshiro C, Jang JY, Reynolds CA, Pedersen NL, Gatz M. Differences Between Women and Men in Incidence Rates of Dementia and Alzheimer’s Disease. J Alzheimers Dis. 2018;64(4):1077–1083. doi:https://doi.org/10.3233/JAD-180141
Hale JM, Schneider DC, Mehta NK, Myrskyla M. Cognitive impairment in the U.S.: Lifetime risk, age at onset, and years impaired. SSM Popul Health. Aug 2020;11:100577. doi:https://doi.org/10.1016/j.ssmph.2020.100577
Ritchie K, Lovestone S. The dementias. Lancet (London, England). Nov 30 2002;360(9347):1759–66. doi:https://doi.org/10.1016/S0140-6736(02)11667-9
Masaldan S, Bush AI, Devos D, Rolland AS, Moreau C. Striking while the iron is hot: Iron metabolism and ferroptosis in neurodegeneration. Free Radic Biol Med. Mar 2019;133:221–233. doi:https://doi.org/10.1016/j.freeradbiomed.2018.09.033
Hocq A, Brouette N, Saussez S, Luhmer M, Gillis P, Gossuin Y. Variable-field relaxometry of iron-containing human tissues: a preliminary study. Contrast Media Mol Imaging. Jul–Aug 2009;4(4):157–64. doi:https://doi.org/10.1002/cmmi.275
Haast RA, Ivanov D, Formisano E, Uludag K. Reproducibility and Reliability of Quantitative and Weighted T1 and T2(*) Mapping for Myelin-Based Cortical Parcellation at 7 Tesla. Front Neuroanat. 2016;10:112. doi:https://doi.org/10.3389/fnana.2016.00112
Fukunaga M, Li TQ, van Gelderen P, et al. Layer-specific variation of iron content in cerebral cortex as a source of MRI contrast. Proc Natl Acad Sci U S A. Feb 23 2010;107(8):3834–9. doi:https://doi.org/10.1073/pnas.0911177107
Cohen-Adad J, Polimeni JR, Helmer KG, et al. T(2)* mapping and B(0) orientation-dependence at 7 T reveal cyto- and myeloarchitecture organization of the human cortex. Neuroimage. Apr 2 2012;60(2):1006–14. doi:https://doi.org/10.1016/j.neuroimage.2012.01.053
Vogt AS, Arsiwala T, Mohsen M, Vogel M, Manolova V, Bachmann MF. On Iron Metabolism and Its Regulation. Int J Mol Sci. Apr 27 2021;22(9)doi:https://doi.org/10.3390/ijms22094591
Wang C, Martins-Bach AB, Alfaro-Almagro F, et al. Phenotypic and genetic associations of quantitative magnetic susceptibility in UK Biobank brain imaging. Nat Neurosci. Jun 2022;25(6):818–831. doi:https://doi.org/10.1038/s41593-022-01074-w
Acknowledgement
This research uses data from the UK Biobank and we appreciate the UK Biobank participants. We thank Anahid Pinchis from Liwen Bianji (Edanz) (www.liwenbianji.cn) for editing the English text of a draft of this manuscript.
Funding
The current study was supported by the Fundamental Research Fund for the Zhejiang Provincial Universities (grant number 2021XZZX029), the Zhejiang University Education Foundation Global Partnership Fund (granted to CY), the National Science Fund for Excellent Young Scholars (81922060), and the Talent Introduction Programme of Chinese Academy of Sciences (granted to GZ). The UK Biobank received the support of the Wellcome Trust, Medical Research Council, Department of Health, Scottish government, and Northwest Regional Development Agency. The research was designed, conducted, analyzed, and interpreted by the authors entirely independently of the funding sources.
Author information
Authors and Affiliations
Contributions
Min Yang and Changzheng Yuan contributed to the conception and design of the study. Hui Chen contributed to the acquisition of data and data analysis. Wenfei Pan, Hui Chen, and Chunxiao Ni wrote the original draft. Min Yang, Geng Zong, and Changzheng Yuan contributed to revising the manuscript. Geng Zong acquired the data and provided the storage and computing resources. All authors reviewed and edited the draft, and approved the final manuscript.
Corresponding authors
Ethics declarations
Ethical approval was obtained from the North West-Haydock Research Ethics Committee (reference: 16/NW/0274).
Additional information
Conflict of Interest Statement
The authors declare that there is no duality of interest related to this manuscript.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Pan, W., Chen, H., Ni, C. et al. Sex-Specific Associations of Dietary Iron Intake with Brain Iron Deposition on Imaging and Incident Dementia: A Prospective Cohort Study. J Nutr Health Aging 26, 954–961 (2022). https://doi.org/10.1007/s12603-022-1852-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12603-022-1852-2