Abstract
Background
The estimation of the risk of poor tolerance and overdose of antineoplastic agents protocols represents a major challenge in oncology, particularly in older patients. We hypothesize that age-related modifications of body composition (i.e. increased fat mass and decreased lean mass) may significantly affect tolerance to chemotherapy.
Method
We conducted a systematic review for the last 25 years (between 1990 and 2015), using US National library of Medicine Medline electronic bibliographic database and Embase database of cohorts or clinical trials exploring (i) the interactions of body composition (assessed by Dual X-ray Absorptiometry, Bioelectrical Impedance Analyses, or Computerized Tomography) with pharmacokinetics parameters, (ii) the tolerance to chemotherapy, and (iii) the consequences of chemotherapies or targeted therapies on body composition.
Results
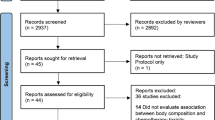
Our search identified 1504 articles. After a selection (using pre-established criteria) on titles and abstract, 24 original articles were selected with 3 domains of interest: impact of body composition on pharmacokinetics (7 articles), relationship between body composition and chemotoxicity (14 articles), and effect of anti-cancer chemotherapy on body composition (11 articles). The selected studies suggested that pharmacokinetic was influenced by lean mass, that lower lean mass could be correlated with toxicity, and that sarcopenic patients experienced more toxicities that non-sarcopenic patients. Regarding fat mass, results were less conclusive. No studies specifically explored the topic of body composition in older cancer patients.
Conclusions
Plausible pathophysiological pathways linking body composition, toxicity, and pharmacokinetics are sustained by the actual review. However, despite the growing number of older cancer patients, our review highlighted the lack of specific studies in the field of anti-neoplastic agents toxicity regarding body composition conducted in elderly.




Similar content being viewed by others
References
Rigal O, Blot E, Druesne L. Epidémiologie: cancer et sujet âgé. Rev Francoph Psycho-Oncologie. 2006;3:141–6.
Lewis JH, Kilgore ML, Goldman DP, Trimble EL, Kaplan R, Montello MJ et al. Participation of patients 65 years of age or older in cancer clinical trials. J Clin Oncol. 2003;21(7):1383–9.
Green JM, Hacker ED. Chemotherapy in the geriatric population. Clin J Oncol Nurs. 2004;8(6):591–7.
Field KM, Kosmider S, Jefford M, Michael M, Jennens R, Green M et al. Chemotherapy dosing strategies in the obese, elderly, and thin patient: results of a nationwide survey. J Oncol Pract. 2008;4(3):108–13.
Griggs JJ, Mangu PB, Anderson H, Balaban EP, Dignam JJ, Hryniuk WM et al. Appropriate chemotherapy dosing for obese adult patients with cancer: American Society of Clinical Oncology clinical practice guideline. J Clin Oncol. 2012;30(13):1553–61.
Prado CM, Maia YL, Ormsbee M, Sawyer MB, Baracos VE. Assessment of nutritional status in cancer—the relationship between body composition and pharmacokinetics. Anticancer Agents Med Chem. 2013;13(8):1197–203.
Prado CM, Antoun S, Sawyer MB, al e. Two faces of drug therapy in cancer: drugrelated lean tissue loss and its adverse consequences to survival and toxicity. Curr Opin Clin Nutr Metab Care. 2011;14(3):250–4.
Amigues I, Schott AM, Amine M, Gelas-Dore B, Veerabudun K, Paillaud E et al. Low skeletal muscle mass and risk of functional decline in elderly communitydwelling women: the prospective EPIDOS study. J Am Med Dir Assoc. 2013;14(5):352–7.
Abellan van Kan G, Rolland Y, Gillette-Guyonnet S, Gardette V, Annweiler C, Beauchet O et al. Gait speed, body composition, and dementia. The EPIDOSToulouse cohort. J Gerontol A Biol Sci Med Sci. 2012;67(4):425–32.
Cruz-Jentoft AJ, Baeyens JP, Bauer JM, al e. Sarcopenia: European consensus on definition and diagnosis: Report of the European Working Group on Sarcopenia in Older People. Age Ageing. 2010;39(4):412–23. doi:10.1093/ageing/afq034.
Gusella M, Toso S, Ferrazzi E, Ferrari M, Padrini R. Relationships between body composition parameters and fluorouracil pharmacokinetics. Br J Clin Pharmacol. 2002;54(2):131–9.
Prado CM, Baracos VE, McCargar LJ, Mourtzakis M, Mulder KE, Reiman T et al. Body composition as an independent determinant of 5-fluorouracil-based chemotherapy toxicity. Clin Cancer Res. 2007;13(11):3264–8.
Stanisavljevic NS, Marisavljevic DZ. Weight and body composition changes during R-CHOP chemotherapy in patients with non-Hodgkin’s lymphoma and their impact on dose intensity and toxicity. J Buon. 2010;15(2):290–6.
Prado CM, Lima IS, Baracos VE, Bies RR, McCargar LJ, Reiman T et al. An exploratory study of body composition as a determinant of epirubicin pharmacokinetics and toxicity. Cancer Chemother Pharmacol. 2011;67(1):93–101.
Mir O, Coriat R, Blanchet B, Durand JP, Boudou-Rouquette P, Michels J et al. Sarcopenia predicts early dose-limiting toxicities and pharmacokinetics of sorafenib in patients with hepatocellular carcinoma. PLoS One. 2012;7(5):e37563.
Massicotte MH, Borget I, Broutin S, Baracos VE, Leboulleux S, Baudin E et al. Body composition variation and impact of low skeletal muscle mass in patients with advanced medullary thyroid carcinoma treated with vandetanib: results from a placebo-controlled study. J Clin Endocrinol Metab. 2013;98(6):2401–8.
Wong AL, Seng KY, Ong EM, Wang LZ, Oscar H, Cordero MT et al. Body fat composition impacts the hematologic toxicities and pharmacokinetics of doxorubicin in Asian breast cancer patients. Breast Cancer Res Treat. 2014;144(1):143–52.
Prado CM, Baracos VE, McCargar LJ, Reiman T, Mourtzakis M, Tonkin K et al. Sarcopenia as a determinant of chemotherapy toxicity and time to tumor progression in metastatic breast cancer patients receiving capecitabine treatment. Clin Cancer Res. 2009;15(8):2920–6.
Barret M, Antoun S, Dalban C, Malka D, Mansourbakht T, Zaanan A et al. Sarcopenia is linked to treatment toxicity in patients with metastatic colorectal cancer. Nutr Cancer. 2014;66(4):583–9.
Cousin S, Hollebecque A, Koscielny S, Mir O, Varga A, Baracos VE et al. Low skeletal muscle is associated with toxicity in patients included in phase I trials. Invest New Drugs. 2014;32(2):382–7.
Prado CM, Baracos VE, Xiao J, Birdsell L, Stuyckens K, Park YC et al. The association between body composition and toxicities from the combination of Doxil and trabectedin in patients with advanced relapsed ovarian cancer. Appl Physiol Nutr Metab. 2014;39(6):693–8.
Tan BH, Brammer K, Randhawa N, Welch NT, Parsons SL, James EJ et al. Sarcopenia is associated with toxicity in patients undergoing neo-adjuvant chemotherapy for oesophago-gastric cancer. Eur J Surg Oncol. 2014;41(3):333–8.
Antoun S, Baracos VE, Birdsell L, Escudier B, Sawyer MB. Low body mass index and sarcopenia associated with dose-limiting toxicity of sorafenib in patients with renal cell carcinoma. Ann Oncol. 2010;21(8):1594–8.
Huillard O, Mir O, Peyromaure M, Tlemsani C, Giroux J, Boudou-Rouquette P et al. Sarcopenia and body mass index predict sunitinib-induced early dose-limiting toxicities in renal cancer patients. Br J Cancer. 2013;108(5):1034–41.
Cushen SJ, Power DG, Teo MY, Maceneaney P, Maher MM, McDermott R et al. Body Composition by Computed Tomography as a Predictor of Toxicity in Patients With Renal Cell Carcinoma Treated With Sunitinib. Am J Clin Oncol. 2014. [Epub ahead of print].
Jebb SA, Osborne RJ, Dixon AK, Bleehen NM, Elia M. Measurements of resting energy expenditure and body composition before and after treatment of small cell lung cancer. Ann Oncol. 1994;5(10):915–9.
Cheney CL, Mahloch J, Freeny P. Computerized tomography assessment of women with weight changes associated with adjuvant treatment for breast cancer. Am J Clin Nutr. 1997;66(1):141–6.
Gil KM, Frasure HE, Hopkins MP, Jenison EL, von Gruenigen VE. Body weight and composition changes in ovarian cancer patients during adjuvant chemotherapy. Gynecol Oncol. 2006;103(1):247–52.
Awad S, Tan BH, Cui H, Bhalla A, Fearon KC, Parsons SL et al. Marked changes in body composition following neoadjuvant chemotherapy for oesophagogastric cancer. Clin Nutr. 2012;31(1):74–7.
Porciuncula Frenzel A, Aberici Pastore C, Gonzalez MC. The influence of body composition on quality of life of patients with breast cancer. Nutr Hosp. 2013;28(5):1475–82.
Yip C, Goh V, Davies A, Gossage J, Mitchell-Hay R, Hynes O et al. Assessment of sarcopenia and changes in body composition after neoadjuvant chemotherapy and associations with clinical outcomes in oesophageal cancer. Eur Radiol. 2014;24(5):998–1005.
Ida S, Watanabe M, Karashima R, Imamura Y, Ishimoto T, Baba Y et al. Changes in body composition secondary to neoadjuvant chemotherapy for advanced esophageal cancer are related to the occurrence of postoperative complications after esophagectomy. Ann Surg Oncol. 2014;21(11):3675–9.
Cooper AB, Slack R, Fogelman D, Holmes HM, Petzel M, Parker N et al. Characterization of Anthropometric Changes that Occur During Neoadjuvant Therapy for Potentially Resectable Pancreatic Cancer. Ann Surg Oncol. 2014. [Epub ahead of print]
Prado CM, Bekaii-Saab T, Doyle LA, Shrestha S, Ghosh S, Baracos VE et al. Skeletal muscle anabolism is a side effect of therapy with the MEK inhibitor: selumetinib in patients with cholangiocarcinoma. Br J Cancer. 2012;106(10):1583–6.
Boker B, Luders H, Grohe C. Prognostic relevance of body mass index and rash for patients with metastatic non-small-cell lung cancer under therapy with erlotinib. Pneumologie. 2012;66(2):89–95.
Schmid SM, Eichholzer M, Bovey F, Myrick ME, Schotzau A, Guth U. Impact of body mass index on compliance and persistence to adjuvant breast cancer therapy. Breast. 2012;21(4):487–92.
Breccia M, Mazzarella L, Bagnardi V, Disalvatore D, Loglisci G, Cimino G et al. Increased BMI correlates with higher risk of disease relapse and differentiation syndrome in patients with acute promyelocytic leukemia treated with the AIDA protocols. Blood. 2012;119(1):49–54.
Simkens LH, Koopman M, Mol L, Veldhuis GJ, Ten Bokkel Huinink D, Muller EW et al. Influence of body mass index on outcome in advanced colorectal cancer patients receiving chemotherapy with or without targeted therapy. Eur J Cancer. 2011;47(17):2560–7.
Kizer NT, Thaker PH, Gao F, Zighelboim I, Powell MA, Rader JS et al. The effects of body mass index on complications and survival outcomes in patients with cervical carcinoma undergoing curative chemoradiation therapy. Cancer. 2011;117(5):948–56.
Jones JA, Fayad LE, Elting LS. Body mass index and outcomes in patients receiving chemotherapy for intermediate-grade B-cell non-Hodgkin lymphoma. Leuk Lymphoma. 2010;51(9):1649–57.
Barpe DR, Rosa DD, Froehlich PE. Pharmacokinetic evaluation of doxorubicin plasma levels in normal and overweight patients with breast cancer and simulation of dose adjustment by different indexes of body mass. Eur J Pharm Sci. 2010;41(3–4):458–63.
Jeddi R, Ghedira H, Mnif S, Gouider E, Fenaux P, Meddeb B. High body mass index is an independent predictor of differentiation syndrome in patients with acute promyelocytic leukemia. Leuk Res. 2010;34(4):545–7.
de Azambuja E, McCaskill-Stevens W, Francis P, Quinaux E, Crown JP, Vicente M et al. The effect of body mass index on overall and disease-free survival in nodepositive breast cancer patients treated with docetaxel and doxorubicin-containing adjuvant chemotherapy: the experience of the BIG 02-98 trial. Breast Cancer Res Treat. 2010;119(1):145–53.
Farhat MH, Shamseddine AI, Tawil AN, Berjawi G, Sidani C, Shamseddeen W et al. Prognostic factors in patients with advanced cholangiocarcinoma: role of surgery, chemotherapy and body mass index. World J Gastroenterol. 2008;14(20):3224–30.
McRackan TR, Watkins JM, Herrin AE, Garrett-Mayer EM, Sharma AK, Day TA et al. Effect of body mass index on chemoradiation outcomes in head and neck cancer. Laryngoscope. 2008;118(7):1180–5.
Barrett SV, Paul J, Hay A, Vasey PA, Kaye SB, Glasspool RM. Does body mass index affect progression-free or overall survival in patients with ovarian cancer? Results from SCOTROC I trial. Ann Oncol. 2008;19(5):898–902.
Sparreboom A, Wolff AC, Mathijssen RH, Chatelut E, Rowinsky EK, Verweij J et al. Evaluation of alternate size descriptors for dose calculation of anticancer drugs in the obese. J Clin Oncol. 2007;25(30):4707–13.
Modesitt SC, Tian C, Kryscio R, Thigpen JT, Randall ME, Gallion HH et al. Impact of body mass index on treatment outcomes in endometrial cancer patients receiving doxorubicin and cisplatin: a Gynecologic Oncology Group study. Gynecol Oncol. 2007;105(1):59–65.
Dignam JJ, Polite BN, Yothers G, Raich P, Colangelo L, O’Connell MJ et al. Body mass index and outcomes in patients who receive adjuvant chemotherapy for colon cancer. J Natl Cancer Inst. 2006;98(22):1647–54.
Gordinier ME, Dizon DS, Fleming EL, Weitzen S, Schwartz J, Parker LP et al. Elevated body mass index does not increase the risk of palmar-plantar erythrodysesthesia in patients receiving pegylated liposomal doxorubicin. Gynecol Oncol. 2006;103(1):72–4.
Ingram C, Brown JK. Patterns of weight and body composition change in premenopausal women with early stage breast cancer: has weight gain been overestimated? Cancer Nurs. 2004;27(6):483–90.
Meyerhardt JA, Catalano PJ, Haller DG, Mayer RJ, Benson AB, 3rd, Macdonald JS et al. Influence of body mass index on outcomes and treatment-related toxicity in patients with colon carcinoma. Cancer. 2003;98(3):484–95.
Miya T, Goya T, Yanagida O, Nogami H, Koshiishi Y, Sasaki Y. The influence of relative body weight on toxicity of combination chemotherapy with cisplatin and etoposide. Cancer Chemother Pharmacol. 1998;42(5):386–90.
Thomas X, Fiere D, Archimbaud E. Influence of increased body mass index on drug toxicity in patients with acute promyelocytic leukemia. Leukemia. 1998;12(9):1503–6.
Gallagher D, Visser M, Sepulveda D, Pierson RN, Harris T, Heymsfield SB. How useful is body mass index for comparison of body fatness across age, sex, and ethnic groups? Am J Epidemiol. 1996;143(3):228–39.
Micozzi MS, Harris TM. Age variations in the relation of body mass indices to estimates of body fat and muscle mass. Am J Phys Anthropol. 1990;81(3):375–9.
Bosy-Westphal A, Muller MJ. Identification of skeletal muscle mass depletion across age and BMI groups in health and disease—there is need for a unified definition. Int J Obes (Lond). 2015;39(3):379–86.
Svendsen OL, Haarbo J, Hassager C, Christiansen C. Accuracy of measurements of body composition by dual-energy x-ray absorptiometry in vivo. Am J Clin Nutr. 1993;57(5):605–8.
Brodie D, Moscrip V, Hutcheon R. Body composition measurement: a review of hydrodensitometry, anthropometry, and impedance methods. Nutrition. 1998;14(3):296–310.
Prado CM, Lieffers JR, McCargar LJ, Reiman T, Sawyer MB, Martin L et al. Prevalence and clinical implications of sarcopenic obesity in patients with solid tumours of the respiratory and gastrointestinal tracts: a population-based study. Lancet Oncol. 2008;9(7):629–35.
Baracos VE, Reiman T, Mourtzakis M, Gioulbasanis I, Antoun S. Body composition in patients with non-small cell lung cancer: a contemporary view of cancer cachexia with the use of computed tomography image analysis. Am J Clin Nutr. 2010;91(4):1133S–7S.
Mourtzakis M, Prado CM, Lieffers JR, Reiman T, McCargar LJ, Baracos VE. A practical and precise approach to quantification of body composition in cancer patients using computed tomography images acquired during routine care. Appl Physiol Nutr Metab. 2008;33(5):997–1006.
Fredrix EW, Saris WH, Soeters PB, Wouters EF, Kester AD, von Meyenfeldt MF et al. Estimation of body composition by bioelectrical impedance in cancer patients. Eur J Clin Nutr. 1990;44(10):749–52.
Thibault R, Pichard C. The evaluation of body composition: a useful tool for clinical practice. Ann Nutr Metab. 2012;60(1):6–16. doi:10.1159/000334879.
Jacquelin-Ravel N, Pichard C. Clinical nutrition, body composition and oncology: a critical literature review of the synergies. Crit Rev Oncol Hematol. 2012;84(1):37–46. doi:10.1016/j.critrevonc.2012.02.001.
John V, Mashru S, Lichtman S. Pharmacological factors influencing anticancer drug selection in the elderly. Drugs Aging. 2003;20(10):737–59.
Lichtman SM, Skirvin JA, Vemulapalli S. Pharmacology of antineoplastic agents in older cancer patients. Crit Rev Oncol Hematol. 2003;46(2):101–14.
Peyrade F, Gastaud L, Re D, Pacquelet-Cheli S, Thyss A. Treatment decisions for elderly patients with haematological malignancies: a dilemma. Lancet Oncol. 2012;13(8):e344–52.
Ma G, Alexander H. Prevalence and pathophysiology of cancer cachexia. Oxford University Press. 1998:91–129.
Ferchichi S, Antoine V. Appropriate drug prescribing in the elderly. Rev Med Interne. 2004;25(8):582–90.
Gurney HP, Ackland S, Gebski V, Farrell G. Factors affecting epirubicin pharmacokinetics and toxicity: evidence against using body-surface area for dose calculation. J Clin Oncol. 1998;16(7):2299–304.
Gurney H. Dose calculation of anticancer drugs: a review of the current practice and introduction of an alternative. J Clin Oncol. 1996;14(9):2590–611.
Miller AA. Body surface area in dosing anticancer agents: scratch the surface! J Natl Cancer Inst. 2002;94(24):1822–3.
Jacquelin-Ravel N, Aapro M, Pichard C. Body composition: decision making support in oncology, current and future practices. Rev Med Suisse. 2012;8(342):1118–23.
Miya T, Goya T, Fujii H, Ohtsu T, Itoh K, Igarashi T et al. Factors affecting the pharmacokinetics of CPT-11: the body mass index, age and sex are independent predictors of pharmacokinetic parameters of CPT-11. Invest New Drugs. 2001;19(1):61–7.
Tan BH, Birdsell LA, Martin L, Baracos VE, Fearon KC. Sarcopenia in an overweight or obese patient is an adverse prognostic factor in pancreatic cancer. Clin Cancer Res. 2009;15(22):6973–9. doi:10.1158/1078-0432.CCR-09-1525.
Parsons HA, Baracos VE, Dhillon N, Hong DS, Kurzrock R. Body composition, symptoms, and survival in advanced cancer patients referred to a phase I service. PLoS One. 2012;7(1):e29330.
Gadea E, Thivat E, Planchat E, Morio B, Durando X. Importance of metabolic changes induced by chemotherapy on prognosis of early-stage breast cancer patients: a review of potential mechanisms. Obes Rev. 2012;13(4):368–80.
Schakman O, Gilson H, Thissen JP. Mechanisms of glucocorticoid-induced myopathy. J Endocrinol. 2008;197(1):1–10. doi:10.1677/JOE-07-0606.
Antoun S, Birdsell L, Sawyer MB, Venner P, Escudier B, Baracos VE. Association of skeletal muscle wasting with treatment with sorafenib in patients with advanced renal cell carcinoma: results from a placebo-controlled study. J Clin Oncol. 2010;28(6):1054–60.
Meador BM, Huey KA. Statin-associated myopathy and its exacerbation with exercise. Muscle Nerve. 2010;42(4):469–79. doi:10.1002/mus.21817.
Galvao DA, Taaffe DR, Spry N, Joseph D, Turner D, Newton RU. Reduced muscle strength and functional performance in men with prostate cancer undergoing androgen suppression: a comprehensive cross-sectional investigation. Prostate Cancer Prostatic Dis. 2009;12(2):198–203. doi:10.1038/pcan.2008.51.
Del Rio G, Zironi S, Valeriani L, Menozzi R, Bondi M, Bertolini M et al. Weight gain in women with breast cancer treated with adjuvant cyclophosphomide, methotrexate and 5-fluorouracil. Analysis of resting energy expenditure and body composition. Breast Cancer Res Treat. 2002;73(3):267–73.
Vance V, Mourtzakis M, McCargar L, Hanning R. Weight gain in breast cancer survivors: prevalence, pattern and health consequences. Obes Rev. 2011;12(4):282–94.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gérard, S., Bréchemier, D., Lefort, A. et al. Body composition and anti-neoplastic treatment in adult and older subjects - A systematic review. J Nutr Health Aging 20, 878–888 (2016). https://doi.org/10.1007/s12603-015-0653-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12603-015-0653-2