Abstract
Objectives
The present study was aimed at evaluating telomere length in blood and in different vascular tissues with or without atheroma, in 3 groups of subjects: a group of atherosclerotic subjects who underwent surgery (Atherosclerosis-Surgery), a second group of subjects with asymptomatic atherosclerotic carotid plaques but who did not undergo cardiovascular surgery (Atherosclerosis-No surgery), and a third group of subjects without atherosclerotic disease (Controls). The main objective was to determine if there is in vivo regulation of telomere length in situ by atherosclerotic lesions.
Methods
A total of 84 subjects (mean age 69±8 years) were studied. Blood and arterial tissue telomere lengths were determined by Southern blotting. Personal medical history (diabetes, hypertension, cardiovascular disease, dyslipidemia), family medical history, drug intake, and lifestyle were evaluated in the entire population through the use of a questionnaire.
Results and Conclusion
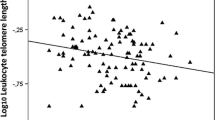
Arterial segments which did not develop atherosclerosis such as the saphenous vein and internal mammary artery, had longer telomere length than aortic segments. On the other hand, telomere length was shorter in aortic tissues which presented atherosclerotic lesions compared to corresponding tissues without atherosclerotic lesions. These results also suggest tissue regulation of telomere size by local factors likely related to oxidative stress responses.
Similar content being viewed by others
References
Kurz DJ, Kloeckener-Gruissem B, Akhmedov A, et al. Degenerative aortic valve stenosis, but not coronary disease, is associated with shorter telomere length in the elderly. Arterioscler Thromb Vasc Biol 2006;26:e114–117.
Nakashima H, Ozono R, Suyama C, Sueda T, Kambe M and Oshima T. Telomere attrition in white blood cell correlating with cardiovascular damage. Hypertens Res 2004;27:319–325.
Samani NJ, Boultby R, Butler R, Thompson JR and Goodall AH. Telomere shortening in atherosclerosis. Lancet 2001;358:472–473.
van der Harst P, van der Steege G, de Boer RA, et al. Telomere length of circulating leukocytes is decreased in patients with chronic heart failure. J Am Coll Cardiol 2007;49:1459–1464.
Benetos A, Gardner JP, Zureik M, et al. Short telomeres are associated with increased carotid atherosclerosis in hypertensive subjects. Hypertension 2004;43:182–185.
Matthews C, Gorenne I, Scott S, et al. Vascular smooth muscle cells undergo telomere-based senescence in human atherosclerosis: effects of telomerase and oxidative stress. Circ Res 2006;99:156–164.
Ogami M, Ikura Y, Ohsawa M, et al. Telomere shortening in human coronary artery diseases. Arterioscler Thromb Vasc Biol 2004;24:546–550.
Basha BJ and Sowers JR. Atherosclerosis: an update. Am Heart J 1996;131:1192–1202.
Viles-Gonzalez JF, Anand SX, Valdiviezo C, et al. Update in atherothrombotic disease. Mt Sinai J Med 2004;71:197–208.
Ben-Porath I and Weinberg RA. The signals and pathways activating cellular senescence. Int J Biochem Cell Biol 2005;37:961–976.
Toussaint O, Medrano EE and von Zglinicki T. Cellular and molecular mechanisms of stress-induced premature senescence (SIPS) of human diploid fibroblasts and melanocytes. Exp Gerontol 2000;35:927–945.
Warnholtz A, Nickenig G, Schulz E, et al. Increased NADH-oxidase-mediated superoxide production in the early stages of atherosclerosis: evidence for involvement of the renin-angiotensin system. Circulation 1999;99:2027–2033.
Petersen S, Saretzki G and von Zglinicki T. Preferential accumulation of single-stranded regions in telomeres of human fibroblasts. Exp Cell Res 1998;239:152–160.
von Zglinicki T, Saretzki G, Docke W and Lotze C. Mild hyperoxia shortens telomeres and inhibits proliferation of fibroblasts: a model for senescence? Exp Cell Res 1995;220:186–193.
Burney S, Niles JC, Dedon PC and Tannenbaum SR. DNA damage in deoxynucleosides and oligonucleotides treated with peroxynitrite. Chem Res Toxicol 1999;12:513–520.
Okuda K, Bardeguez A, Gardner JP, et al. Telomere length in the newborn. Pediatr Res 2002;52:377–381.
Cattan V, Mercier N, Gardner JP, et al. Chronic oxidative stress induces a tissue-specific reduction in telomere length in CAST/Ei mice. Free Radic Biol Med 2008;44:1592–1598.
Voghel G, Thorin-Trescases N, Farhat N, et al. Cellular senescence in endothelial cells from atherosclerotic patients is accelerated by oxidative stress associated with cardiovascular risk factors. Mech Ageing Dev 2007;128:662–671.
Minamino T, Miyauchi H, Yoshida T, Ishida Y, Yoshida H and Komuro I. Endothelial cell senescence in human atherosclerosis: role of telomere in endothelial dysfunction. Circulation 2002;105:1541–1544.
Okuda K, Khan MY, Skurnick J, Kimura M, Aviv H and Aviv A. Telomere attrition of the human abdominal aorta: relationships with age and atherosclerosis. Atherosclerosis 2000;152:391–398.
Chang E and Harley CB. Telomere length and replicative aging in human vascular tissues. Proc Natl Acad Sci U S A 1995;92:11190–11194.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nzietchueng, R., Elfarra, M., Nloga, J. et al. Telomere length in vascular tissues from patients with atherosclerotic disease. J Nutr Health Aging 15, 153–156 (2011). https://doi.org/10.1007/s12603-011-0029-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12603-011-0029-1