Abstract
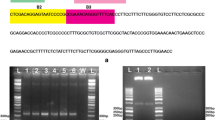
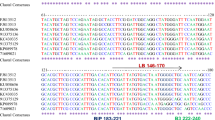
SYBR green based realtime RT-PCR assay coupled with melt curve analysis was developed for the detection of Potato spindle tuber viroid (PSTVd) along with and without internal control from potato. The amplification of the specific targets was identified by their melting points viz., 85.93±0.22, 85.62±0.34 and 82.07±0.23 for primer pairs PSTVd-1F/PSTVd-1R, PSTVd-NFP/PSTVd-NRP and PSTVd-QFP1/PSTVd-QRP1, respectively. The realtime RT-PCR assay was 1×104 times more sensitive than RT-PCR assay and the assay could detect up to 20 copies of the target using serially diluted plasmid and up to 0.025 fg of total RNA from infected tissues diluted in healthy plant RNA. Duplex realtime RT-PCR assay was also standardized to detect PSTVd along with elongation factor 1- α (ef-1α) gene, a stable housekeeping gene of potato. Duplex realtime RT-PCR assay showed two melt peaks indicating the successful amplification both the PSTVd RNA and internal control RNA. The developed assays could consistently detect PSTVd and proved to be highly sensitive and rapid for the detection of PSTVd in post entry quarantine testing.



Similar content being viewed by others
References
Boonham, N., Pérez, G. L., Mendez, M. S., Lilia Peralta, E., Blockly, A., Walsh, K., Barker, I., & Mumford, R. A. (2004). Development of a real-time RT-PCR assay for the detection of Potato spindle tuber viroid. Journal of Virological Methods, 116, 139–146.
Botermans, M., van de Vossenberg, B. T. L. H., Verhoeven, J. T. J., Roenhorst, J. W., Hooftman, M., Dekter, R., & Meekes, E. T. M. (2013). Development and validation of a real-time RT-PCR assay for generic detection of pospiviroids. Journal of Virological Methods, 187, 43–50.
Diener, T. O. (1979). Viroids and Viroid Diseases (p. 252). New York, USA: Wiley- Interscience.
Gast, F. U., Kempe, D., Spieker, R. L., & Sanger, H. L. (1996). Secondary structure probing of Potato spindle tuber viroid (PSTVd) and sequence comparison with other small pathogenic RNA replicons provides evidence for central non-canonical base-pairs, large A-rich loops, and a terminal branch. Journal of Molecular Biology, 262, 652–670.
Hataya, T. (2009). Duplex reverse transcription-polymerase chain reaction system to detect Potato spindle tuber viroid using an internal control mRNA and a non-infectious positive control RNA. Journal of General Virology, 75, 167–172.
Hui, M., Xiao-liang, X., Jiang, Y., Chun-xiu, W., & Zhi-ming, W. (2007). Duplex RT-PCR Detection of Potato spindle tuber viroid with an Internal Control of Potato ND2 mRNA. Acta Horticulturae Sinica, 34, 1213–1216.
Lenarcic, R., Morisset, D., Mehle, N., & Ravnikar, M. (2012). Fast real-time detection of Potato spindle tuber viroid by RT-LAMP. Plant Pathology. doi:10.1111/ppa.12017.
Matsushita, Y., Usugi, T., & Tsuda, S. (2010). Development of a multiplex RT-PCR detection and identification system for Potato spindle tuber viroid and Tomato chlorotic dwarf viroid. European Journal of Plant Pathology, 128, 165–170.
Monger, W., Tomlinson, J., Boonham, N., Marn, M. V., Plesko, I. M., Molinero-Demilly, V., Tassus, X., Meekes, E., Toonen, M., Papayiannis, L., Perez-Egusquiza, Z., Mehle, N., Jansen, C., & Nielsen, S. L. (2010). Development and inter-laboratory evaluation of real-time PCR assays for the detection of pospiviroids. Journal of Virological Methods, 169, 207–210.
Nicot, N., Hausman, J. F., Hoffmann, L., & Evers, D. (2005). Housekeeping gene selection for real-time RT-PCR normalization in potato during biotic and abiotic stress. Journal of Experimental Botany, 56, 2907–2914.
Olivier, T., Demonty, E., Fauche, F., & Steyer, S. (2014). Generic detection and identification of pospiviroids. Archives of Virology, 159, 2097–2102.
Owens, R. A., Paul Khurana, S. M., Smith, D. R., Singh, M. N., & Garg, I. D. (1992). A new mild strain of potato spindle tuber viroid isolated from wild Solanum spp. in India. Plant Disease, 76, 527–529.
Pfannenstiel, M. A., & Slack, S. A. (1980). Response of potato cultivars to infection by the potato spindle tuber viroid. Phytopathology, 70, 922–926.
Roenhorst, J. W., Jansen, C. C. C., Kox, L. F. F., deHaan, E. G., vanden Bovenkamp, G. W., Boonham, N., Fisher, T., & Mumford, R. A. (2005). Application of real-time RT-PCR for large-scale testing of potato for Potato spindle tuber pospiviroid. European Plant Protection Organization Bulletin, 35, 133–140.
Seigner, L., Kappen, M., Huber, C., Kistler, M., & Köhler, D. (2008). First trials for transmission of Potato spindle tuber viroid from ornamental Solanaceae to tomato using RT-PCR and an mRNA based internal positive control for detection. Journal of Plant Diseases and Protection, 115, 97–101.
Verma, Y., Shukla, A., Paul Khurana, S. M., & Garg, I. D. (2006). Detection of potato spindle tuber viroid (pstvd) using fluorescein labeled probe. Potato Journal, 33, 122–125.
Welnicki, M., & Hiruki, C. (1992). Highly sensitive digoxigenin labeled DNA probe for the detection of potato spindle tuber viroid. Journal of Virological Methods, 39, 91–99.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jeevalatha, A., Kumar, R., Raigond, B. et al. Duplex realtime RT-PCR assay for the detection of Potato spindle tuber viroid (PSTVd) along with ef 1-α gene of potato. Phytoparasitica 43, 317–325 (2015). https://doi.org/10.1007/s12600-014-0452-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12600-014-0452-z