Abstract
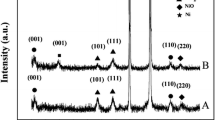
Samples of lanthanum-doped nickel hydroxide were prepared by electrodeposition method. The structure and electrochemical properties of the samples were studied by X-ray diffraction and a home-made open three-electrode cell system, respectively. The results show that the deposition process of Ni(OH)2 and La(OH)3 is mainly controlled by electrochemical polarization, which makes it easy to form uniform fine crystals. In addition, La(OH)3 is not a separate phase and lanthanum ions are doped into Ni(OH)2 crystal lattices. When V(0.5 mol/L Ni(NO3)2)/V(0.25 mol/L La(NO3)3) was 9:1, the lanthanum-doped nickel hydroxide reached the highest discharge capability of 840 F/g with a good cyclic reversibility. The capability still retains 670 F/g when the discharge current reaches 1000 mA/g.
Similar content being viewed by others
References
Yu W.P., Yang X.P., Meng L.K., and Liu Z.Z., Character of Co-doped Ni(OH)2 electrode materials prepared by electrodeposition, Trans. Mater. Heat Treat. (in Chinese), 2005, 26(6): 30.
Liu Z., Wu F., Wang F., Zhang W.Q., Zhang C.Z., and Chen S., Research on electrodeposition preparation and performance of NiO thin-film electrode, Mater. Rev. (in Chinese), 2006, 20(6): 137.
Wu M.Q., Gao J.H., Zhang S.R., and Chen A., Comparative studies of nickel oxide films on different substrates for electrochemical supercapacitors, J. Power Sources, 2006, 159:365.
Zhu W.H., Ke J.J., Yu H.M., and Zhang D.J., A study of the electrochemistry of nickel hydroxide electrodes with various additives, J. Power Sources, 1995, 56: 75.
Meng L.K. and Yu W.P., Study on Ni(OH)2 supercapacitor positive electrode material prepared by electrochemical deposition in the ethanol and water system, Trans. Mater. Heat Treat. (in Chinese), 2006, 27(5): 17.
Shao G.J. and Qin X.J., Influence of rare earth on bath performance of Ni-P alloy electrodeposition, J. Chin. Rare Earth Soc. (in Chinese), 2002, 20(1): 88.
Yang Y.P., Zhang P.M., Zhang Y.B., Tang Y.G., Liu K.Y., and Shang S.B., Preparation and electrochemical performance of lanthanum doping α-Ni(OH)2, J. Cent. South Univ. Sci. Technol. (in Chinese), 2005, 36(6): 988.
Guo Y. and Lu G.Z., Current status and some perspectives of rare earth catalytic materials, J. Chin. Rare Earth Soc. (in Chinese), 2007, 25(1): 1.
Escudero M.J., Mendoza L., Cassir M., Gonzalez T., and Daza L., Porous nickel MCFC cathode coated by potentiostatically deposited cobalt oxide II: structural and morphological behavior in molten carbonate, J. Power Sources, 2006, 160: 775.
Ganesh V., Pitchumani S., and Lakshminarayanan V., New symmetric and asymmetric supercapacitors based on high surface area porous nickel and activated carbon, J. Power Sources, 2006, 158: 1523.
Deabate S., Fourgeot F., and Henn F., Electrochemical behaviour of the β(II)-Ni(OH)2/β(III)-NiOOH redox couple upon potentiodynamic cycling conditions, Electrochim. Acta, 2006, 51: 5430.
Salimi A., Sharifi E., Noorbakhsh A., and Soltanian S., Direct voltammetry and electrocatalytic properties of hemoglobin immobilized on a glassy carbon electrode modified with nickel oxide nanoparticles, Electrochem. Commun., 2006, 8:1499.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Shao, G., Yao, Y., Zhang, S. et al. Supercapacitor characteristic of La-doped Ni(OH)2 prepared by electrode-position. Rare Metals 28, 132–136 (2009). https://doi.org/10.1007/s12598-009-0026-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12598-009-0026-2