Abstract
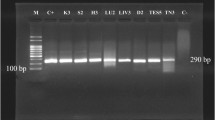
Diagnosis of mycobacterial infection is paramount important from the public health perspective since treatment and control measures are very significant, particularly in captive animals. In this diagnostic study of Mycobacterium bovis infection in sloth bears (Melursus ursinus), polymerase chain reaction (PCR) had been used with the primer sequence of pncA-8 (5′-GGTTGGGTGGCCGCCGGTCAG-3′) and pncA-11 (5′-GCTTTGCGGCGAGCGCTCCA-3′) that were specific for M. bovis pncA gene. Forty-two fresh faecal samples were collected randomly from the apparently healthy sloth bears maintained in captive conditions. The DNA extraction procedure was done as per the manufacturer’s protocol and further subjected to amplification. The amplification profile includes respectively: initial heating of the samples for 5 min at 94 °C, annealing at 55 °C for 1 min, primer extension at 72 °C for 1 min and final elongation step for 10 min at 72 °C. Out of 57 samples, 5 samples were yielded on expected amplified PCR product size of 744 bp when electrophoresed in 1.5 % agarose gel. A positive control of M. bovis DNA procured from Tuberculosis Research Centre and a negative control from a healthy bovine sample were used. These results demonstrated that PCR test will increase the effectiveness of laboratory diagnosis to detect and identifying the M. bovis in captive wild animals.

Similar content being viewed by others
References
Arora, B.M. 1994. Wildlife diseases in India. Infectious and parasitic diseases of mammals, reptiles and amphibian. New Delhi: Periodical expert book agency.
Balamurugan, R., S. Venkataraman, K.R. John, and B.S. Ramakrishna. 2006. PCR Amplification of the IS6110 Insertion element of Mycobacterium tuberculosis in faecal samples from patients with intestinal tuberculosis. Journal of Clinical Microbiology 44(5): 1884–1886.
Berland, J.C. 1982. No five fingers are alike, 98–104. Cambridge: Harvard University Press.
Cousins, D.V., S.D. Wilton, and B.R. Francis. 1991. Use of DNA amplification for the rapid identification of Mycobacterium bovis. Veterinary Microbiology 27: 187–195.
Das, A.M., and B.M. Jayarao. 1986. Tuberculosis in a lioness (Panthera leo persica). Indian Journal of Comparative Microbiology Immunology, Infectious disease 7(2&3): 116.
Hannan, M.M., E.P. Desmond, G.P. Morlock, G.H. Mazurek, and J.T. Crawford. 2001. Pyrazinamide—Monoresistant Mycobacterium tuberculosis in the United States. Journal of Clinical Microbiology 39(2): 647–650.
Hawkey, P.M., D.M. Gascoyne-Binzi, and P. Kite. 1996. Molecular diagnosis and epidemiology of tuberculosis. In Tuberculosis into the rent century, ed. Hart, C.A., M.J. Beeching, and B.I. Duarden. Journal of Medical Microbiology 44: 1–34.
Muralimanohar, M., A.V. Korandi, S. Vairamuthu, M.G. Jayathangaraj, and K. Senthilkumar. 2006. Tuberculosis in a bear. Indian Veterinary Journal 83: 684–685.
Rathore, B.S., and S.S. Kher. 1982. Tuberculosis in wild mammals and birds in captivity and free living state. Indian Veterinary Journal 6: 665–668.
Scorpio, A., and Y. Zhang. 1996. Mutations in pncA, a gene encoding pyrazinamidase/nicotinamidase, cause resistance to the antituberculosis drug pyrazinamide in tubercle bacillus. Natural Medicine 2: 662–667.
Sen Gupta, M.R. 1974. A preliminary report on diseases and parasites of zoo animals, birds, reptiles. Indian Journal and Animal Health 13: 15–24.
Stetter, M.D., S.K. Mikota, A.F. Gutter, E.R. Monterroso, J.R. Dalvisio, C. Degraw, and T. Farley. 1995. Epizootic of Mycobacterium bovis in a zoologic park. Journal of American Veterinary Medical Association 207(12): 1618–1621.
Thakuria, D. 1996. Prevalence, transmission, diagnosis and control of tuberculosis in Assam State zoo: A review. Zoos’ Print 11(3): 6–15.
Thoen, C.O. 1988. Tuberculosis. Journal of American Veterinary Medical Association 193(9): 1045–1048.
Thoen, C.O., J.H. Steele, and M.J. Gilsdorf. 2006. Mycobacterium bovis infection in animals and humans, 2nd ed. Boston: Blackwell Publishers.
Winkler, W.G., and N.B. Gale. 1970. Tuberculosis. In Infectious disease and wild mammals, 1st ed, ed. J.W. Davis, L.H. Karstad, and D.O. Trainer, 248–256. Ames: The Iowa state University Press.
Acknowledgments
The authors are thankful to PCCF and Chief Wildlife Warden, Bangalore and Secretary Wildlife SOS® for granting permission to carryout this research work. Facilities provided by Department of Animal Biotechnology, Madras veterinary College is also duly acknowledged.
Conflict of interest
We declare that we have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Veeraselvam, M., Sridhar, R., Senthilkumar, T.M.A. et al. Diagnosis of Mycobacterium bovis in Captive Sloth Bears (Melursus ursinus) by Polymerase Chain reaction. Proc Zool Soc 68, 109–111 (2015). https://doi.org/10.1007/s12595-014-0102-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12595-014-0102-y