Abstract
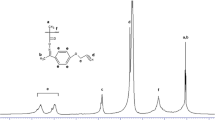
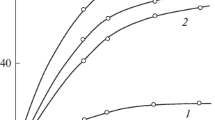
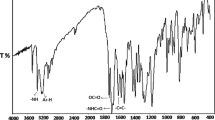
A recently developed successful approach to achieve the balanced properties involves the copolymerization of film forming monomers like acrylate and methacrylates with crosslinkable monomers. The crosslinker used in the copolymerization of acrylates are basically compound with at least two non-conjugated ethylenic double bonds in their molecule. The kinetics of esterification of multifunctional monomers were investigated using poly(propylene glycol) diacrylates. The effect of methyl group on reaction kinetics was investigated. The diacrylate / dimethacrylate based on polypropylene glycol (molecular weight 400) were synthesized by reacting with acrylic / methacrylic acid in presence of a catalyst. The decrease in acid value and the presence of prominent peak, –C = O stretching, at 1,731 cm−1 showed the formation of diacrylate and dimethacrylate. The value of rate constant for PPGDA at all temperatures were higher than that for PPGDA, indicating that the reactivity of PPG towards acrylic acid is more than methacrylic acid.








Similar content being viewed by others
References
Spevacek J, Schneider B (1987) Aggregation of stereoregular poly(methyl methacrylates). Adv Colloid Interface Sci 27:81
Wolff EM (1962) The effect of cross-linking agents on acrylic resins. Aust Dent J 7:439
Anseth KS, Kline LM, Walker TA, Anderson KJ, Bowman CN (1995) Reaction kinetics and volume relaxation during polymerizations of multiethylene glycol dimethacrylates. Macromolecules 28(7):2491
Takezawa Y, Tanno S, Taketani N, Ohara S, Asano H (1991) Analysis of thermal degradation for plastic optical fibers. J Appl Polym Sci 42(10):2811
Ma CCM, Chen CH (1991) Pultruded fiber reinforced thermoplastic poly(methyl methacrylate) composites. Part I. Correlation of processing parameters for optimizing the process. Polym Eng Sci 31:1086
Acrylic Acid and Derivatives (1987) Ullmann’s Encyclopedia, fourth edn., vol. A1 Wiley, pp. 16
Acrylic Acid and Derivatives (1987) Kirk–Othmer Encyclopedia of chemical technology, fourth edn., vol. 1, Wiley:287
Chen X, Xu Z, Okuhara T (1999) Liquid phase esterification of acrylic acid with 1-butanol catalyzed by solid acid catalysts. Appl Catal A Gen 180:261
Van Eikeren A, Plaumann MT (2010) Dental masking product for teeth and gum. US Pat 7789662
Steller KE, Vanderberg EJ (1975) Polyethers. ACS, Washington, DC
Bajpai S, Rai JSP, Nigam I (2008) Synthesis and characterization of methyl methacrylate – n – butylacrylate latexes. Int J Plast Technol 12:1039
Simons DM, Verbanc JJ (1960) The polymerization of propylene oxide. J Polym Sci 44:303
Lilja J, Aumo J, Salmi T, Murzin DY, Arvela PM, Sundell M, Ekman K, Peltonen R, Vainio H (2002) Appl Catal A Gen 228:223
Akbay E, Altıokka MR (2011) Performance and regeneration studies of Pd–Ag/Al2O3 catalysts for the selective hydrogenation of acetylene. Appl Catal A Gen 396:14
Ali SH, Tarakmah A, Merchant SQ, Al-Sahhaf T (2007) Synthesis of esters: development of the rate expression for the Dowex 50 Wx8-400 catalyzed esterification of propionic acid with 1-propanol. Chem Eng Sci 62:3197
Delgado P, Sanz M, Beltran S (2007) Kinetic study for esterification of lactic acid with ethanol and hydrolysis of ethyl lactate using an ion-exchange resin catalyst. Chem Eng J 126:111
Shanmugam S, Viswanathan B, Varadarajan TK (2004) Esterification by solid acid catalysts a comparison. J Mol Catal A Chem 223:143
Altiokka MR, Odes E (2009) Reaction kinetics of the catalytic esterification of acrylic acid with propylene glycol. Appl Catal A Gen 362:115
Korshak VV, Vinogradova (1965) In: Burdon (ed) Polyesters. Pergamon, Oxford, p 191
Odian G (1974) Principles of polymerization. Wiley Interscience, New York, p 46
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Shukla, S., Rai, J.S.P. Synthesis and kinetic study of diacrylate and dimethacrylate. Int J Plast Technol 17, 182–193 (2013). https://doi.org/10.1007/s12588-013-9058-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12588-013-9058-4