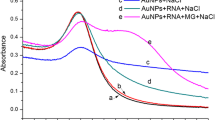
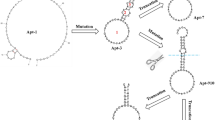
Abstract
Leucomalachite green (LMG), a derivative antimicrobial agent, can accumulate in animal tissues for long periods and is categorized as a carcinogen affecting consumer health. Therefore, determining the contamination of aquatic products by LMG is necessary to prevent residual contamination. In the work described herein, the detection of LMG based on a colorimetric method using gold nanoparticles (AuNPs) was developed. The specificity of isolated single-stranded DNA aptamers to LMG was proven by using LMG and other antibiotics. The binding affinity of the LMG-aptamer showed a Kd value of 1.94 pM. Furthermore, a colorimetric detection module was successfully developed, showing a limit of detection (LOD) value of LMG at 250 nM using standard LMG substance. Taken together, this research demonstrates a rapid and sensitive method to detect chemical residues, reducing the possibility of contamination in fishery products. Considering these findings, the aptamer-based colorimetric approach might be feasible to utilize as a biosensing module, providing a rapid, inexpensive analysis method for detecting deposited chemical residues.





Similar content being viewed by others
References
Abro K, Mahesar SA, Iqbal S, Perveen S (2014) Quantification of malachite green in fish feed utilising liquid chromatography-tandem mass spectrometry with a monolithic column. Food Addit Contam Part A 31(5):827–832. https://doi.org/10.1080/19440049.2014.893398
Antczak M, Popenda M, Zok T, Sarzynska J, Ratajczak T, Tomczyk K, Adamiak RW, Szachniuk M (2016) New functionality of RNAComposer: an application to shape the axis of miR160 precursor structure. Acta Biochim Pol 63(4):737–744. https://doi.org/10.18388/abp.2016_1329
Asefa T, Duncan CT, Sharma KK (2009) Recent advances in nanostructured chemosensors and biosensors. Analyst 134(10):1980–1990
Bhasikuttan AC, Mohanty J, Pal H (2007) Interaction of malachite green with guanine-rich single-stranded DNA: preferential binding to a G-quadruplex. Angew Chem Int Ed 46(48):9305–9307. https://doi.org/10.1002/anie.200703251
Conti G, Copat C, Wang Z, D’Agati P, Cristaldi A, Ferrante M (2015) Determination of illegal antimicrobials in aquaculture feed and fish: an ELISA study. Food Control 50:937–941
Culp SJ, Beland FA (1996) Malachite green: a toxicological review. Int J Toxicol 15:219–238. https://doi.org/10.3109/10915819609008715
Department of Food Safety of Japan, Pharmaceutical and Food Safety Bureau, Ministry of Health (2005) The Director Notice (Syoku-An No.1129001 dated November 29, 2005) Detection Limits of the Analytical Methods Stipulated in Items 5, 6, and 7 in the Revised General Requirements. Labour and Welfare, Japan
EFSA Contam Panel (EFSA Panel on Contaminants in the Food Chain) (2016) Scientific opinion on malachite green in food. EFSA J 14(7):4530. https://doi.org/10.2903/j.efsa.2016.4530
Famulok M, Mayer G (2011) Aptamer modules as sensors and detectors. Acc Chem Res 44(12):1349–1358. https://doi.org/10.1021/ar2000293
FAO/WHO (2009) Principles and methods for the risk assessment of chemicals in food. Environmental Health Criteria. 240. World Health Organization, Geneva, Switzerland. http://www.who.int/foodsafety/chem/principles/en/index1.html Accessed 7 Sep 2022
Gruber AR, Lorenz R, Bernhart SH, Neuböck R, Hofacker IL (2008) The Vienna RNA websuite. Nucleic Acids Res 36:W70–W74. https://doi.org/10.1093/nar/gkn188
Han K, Liang Z, Zhou N (2010) Design strategies for aptamer-based biosensors. Sensors 10(5):4541–4557. https://doi.org/10.3390/s100504541
Hu X, Chang K, Wang S, Sun X, Hu J, Jiang M (2018) Aptamer-functionalized AuNPs for the high-sensitivity colorimetric detection of melamine in milk samples. PLoS ONE 13(8):e0201626. https://doi.org/10.1371/journal.pone.0201626
Huntington T, Hasan M (2009) Fish as feed inputs for aquaculture-practices, sustainability and implications: a global synthesis. FAO Fish Tech Pap 518:1–61
Ilgu M, Nilsen-Hamilton M (2016) Aptamers in analytics. Anlst 141(5):1551–1568. https://doi.org/10.1039/C5AN01824B
Iliuk AB, Hu L, Tao WA (2011) Aptamer in bioanalytical applications. Anal Chem 83(12):4440–4452. https://doi.org/10.1021/ac201057w
Jia J, Yan S, Lai X, Xu Y, Liu T, Xiang Y (2018) Colorimetric aptasensor for detection of malachite green in fish sample based on RNA and gold nanoparticles. Food Anal Methods 11(6):1668–1676. https://doi.org/10.1007/s12161-017-1144-3
Kalra P, Dhiman A, Cho WC, Bruno JG, Sharma TK (2018) Simple methods and rational design for enhancing aptamer sensitivity and specificity. Front Mol Biosci 5:41. https://doi.org/10.3389/fmolb.2018.00041
Khoshfetrat SM, Mehrgardi MA (2017) Amplified detection of leukemia cancer cells using an aptamer-conjugated gold-coated magnetic nanoparticles on a nitrogen-doped graphene modified electrode. Bioelectrochemistry 114:24–32. https://doi.org/10.1016/j.bioelechem.2016.12.001
Kim YS, Kim JH, Kim IA, Lee SJ, Jurng J, Gu MB (2010) A novel colorimetric aptasensor using gold nanoparticle for a highly sensitive and specific detection of oxytetracycline. Biosens Bioelectron 26(4):1644–1649. https://doi.org/10.1016/j.bios.2010.08.046
Kohlberger M, Gadermaier G (2021) SELEX: Critical factors and optimization strategies for successful aptamer selection. Biotechnol Appl Biochem. https://doi.org/10.1002/bab.2244
Komarova N, Andrianova M, Glukhov S, Kuznetsov A (2018) Selection, characterization, and application of ssDNA aptamer against furaneol. Molecules 23:3159. https://doi.org/10.3390/molecules23123159
Li RLJ (2004) Label-free colorimetric detection of specific sequences in genomic DNA amplified by the polymerase chain reaction. J Am Chem Soc 126(35):10958–10961. https://doi.org/10.1021/ja048749n
López-Gutiérrez N, Romero-González R, Martínez Vidal JL, Frenich AG (2013) Analysis of triphenylmethane dyes in seafood products: a review of extraction methods and determination by liquid chromatography coupled to mass spectrometry. Anal Methods. https://doi.org/10.1039/c3ay40485d
Mathews DH (2004) Using an RNA secondary structure partition function to determine confidence in base pairs predicted by free energy minimization. RNA 10(8):1178–1190. https://doi.org/10.1261/rna.7650904
Morris GM, Huey R, Lindstrom W, Sanner MF, Belew RK, Goodsell DS, Olson AJ (2009) Autodock4 and AutoDockTools4: automated docking with selective receptor flexibility. J Comput Chem 30:2785–2791
Nebot C, Iglesias A, Barreiro R, Miranda JM, Vázquez B, Franco CM, Cepeda A (2013) A simple and rapid method for the identification and quantification of malachite green and its metabolite in hake by HPLC-MS/MS. Food Control 31(1):102–107. https://doi.org/10.1016/j.foodcont.2012.09.020
Nguyen DK, Jang CH (2021) A simple and ultrasensitive colorimetric biosensor for anatoxin-a based on aptamer and gold nanoparticles. Micromachines 12(12):1526. https://doi.org/10.3390/mi12121526
Nohmi T (2018) Thresholds of genotoxic and non-genotoxic carcinogens. Toxicol Res. https://doi.org/10.5487/TR.2018.34.4.281
Official Journal of the European Union (2004) Commission Decision 2004/25/EC of 22 December 2003 amending Decision 2002/657/EC as regards the setting of minimum required performance limits (MRPLs) for certain residues in food of animal origin. 6:38–39
Popenda M, Szachniuk M, Antczak M, Purzycka KJ, Lukasiak P, Bartol N, Blazewicz J, Adamiak RW (2012) Automated 3D structure composition for large RNAs. Nucleic Acids Res 40(14):e112. https://doi.org/10.1093/nar/gks339
Saha K, Agasti SS, Kim C, Li X, Rotello VM (2012) Gold nanoparticles in chemical and biological sensing. Chem Rev 112(5):2739–2779. https://doi.org/10.1021/cr2001178
Schuetze A, Heberer T, Juergensen S (2008) Occurrence of residues of the veterinary drug malachite green in eels caught downstream from municipal sewage treatment plants. Chemosphere 72(11):1664–1670. https://doi.org/10.1016/j.chemosphere.2008.05.036
Shipway AN, Katz E, Willner I (2000) Nanoparticle arrays on surfaces for electronic, optical, and sensor applications. ChemPhysChem 1(1):18–52. https://doi.org/10.1002/1439-7641(20000804)1:1%3c18::AID-CPHC18%3e3.0.CO;2-L
Singh G, Koerner T, Gelinas JM, Abbott M, Brady B, Huet AC, Charlier C, Delahaut P, Benrejeb Godefroy S (2011) Design and characterization of a direct ELISA for the detection and quantification of leucomalachite green. Food Addit Contam Part A 28(6):731–739. https://doi.org/10.1080/19440049.2011.567360
Spahn CM, Prescott CD (1996) Throwing a spanner in the works: antibiotics and the translation apparatus. J Mol Med 74(8):423–439. https://doi.org/10.1007/BF00217518
Srivastava S, Sinha R, Roy D (2004) Toxicological effects of malachite green. Aquat Toxicol 66(3):319–329. https://doi.org/10.1016/j.aquatox.2003.09.008
Stead SL, Ashwin H, Johnston BH, Dallas A, Kazakov SA, Tarbin JA, Sharman M, Kay J, Keely BJ (2010) An RNA-aptamer-based assay for the detection and analysis of malachite green and leucomalachite green residues in fish tissue. Anal Chem 82(7):2652–2660. https://doi.org/10.1021/ac902226v
Stoltenburg R, Nikolaus N, Strehlitz B (2012) Capture-SELEX: selection of DNA aptamers for aminoglycoside antibiotics. J Anal Chem. https://doi.org/10.1155/2012/415697
Sudova E, Machova J, Svobodova Z, Vesely T (2007) Negative effects of malachite green and possibilities of its replacement in the treatment of fish eggs and fish. Vet Med 52:527–539. https://doi.org/10.17221/2027-VETMED
Takagi M, Yamatani S, Sugiura K, Ministry of Agriculture, Forestry and Fisheries (2011) Guidelines used in Japan to prevent the contamination of feed products with undesirable substances. Vet 47(1):49–52
Tuševljak N, Dutil L, Rajić A, Uhland FC, McClure C, St-Hilaire S, Reid-Smith RJ, McEwen SA (2013) Antimicrobial use and resistance in aquaculture: findings of a globally administered survey of aquaculture-allied professionals. Zoonoses Public Health 60(6):426–436. https://doi.org/10.1111/zph.12017
US Food and Drug Administration (2020b) Compliance Program Guidance Manual. Program 7304.018. Chapter 04-Pesticides and Chemical Contaminants. Chemotherapeutics in Aquaculture Se Compliance Program. C. Target Testing Level (TTL)/Regulatory Action Level (RAL). 25–26
Wandtke T, Woźniak J, Kopiński P (2015) Aptamers in diagnostics and treatment of viral infections. Viruses 7(2):751–780. https://doi.org/10.3390/v7020751
Wang L, Liu X, Hu X, Song S, Fan C (2006) Unmodified gold nanoparticles as a colorimetric probe for potassium DNA aptamers. Chem Commun 36:3780–3782. https://doi.org/10.1039/B607448K
Wang H, Wang J, Sun N, Cheng H, Chen H, Pei R (2016) Selection and characterization of malachite green aptamers for the development of light–up probes. ChemistrySelect 1:1571
Wu W, Sun Q, Li T, Liu K, Jiang Y, Wang Y, Yang Y (2022) Selection and characterization of bispecific aptamers against malachite green and leucomalachite green. Anal Biochem 658:114849. https://doi.org/10.1016/j.ab.2022.114849
Xie M, Chen Z, Zhao F, Lin Y, Zheng S, Han S (2022) Selection and application of ssDNA aptamers for fluorescence biosensing detection of malachite green. Foods 11:801. https://doi.org/10.3390/foods11060801
Yan C, Zhang J, Yao L, Xue F, Lu J, Li B, Chen W (2018) Aptamer-mediated colorimetric method for rapid and sensitive detection of chloramphenicol in food. Food Chem 260:208–212. https://doi.org/10.1016/j.foodchem.2018.04.014
Zayats M, Baron R, Popov I, Willner I (2005) Biocatalytic growth of au nanoparticles: from mechanistic aspects to biosensors design. Nano Lett 5(1):21–25
Zhang J, Liu B, Liu H, Zhang X, Tan W (2013) Aptamer-conjugated gold nanoparticles for bioanalysis. Nanomedicine 8(6):983–993. https://doi.org/10.2217/nnm.13.80
Zhang Y, Lai BS, Juhas M (2019) Recent advances in aptamer discovery and applications. Molecules 24(5):941. https://doi.org/10.3390/molecules24050941
Zhou J, Rossi J (2017) Aptamers as targeted therapeutics: current potential and challenges. Nat Rev Drug Discov 16:181–202. https://doi.org/10.1038/nrd.2016.199
Acknowledgements
This work was also supported by the Japan Science and Technology Agency/Japan International Cooperation Agency, Science and Technology Research Partnership for Sustainable Development (JST/JICA, SATREPS) and Kasetsart University Research and Development Institute under grant number KURDI FF(KU) 17.64. CJ is supported in part by the Graduate Program Scholarship from The Graduate School, Kasetsart University.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Jaengphop, C., Phurahong, T., Hirono, I. et al. Novel, rapid, and sensitive colorimetric detection of leucomalachite green using a specific aptamer. Fish Sci 89, 809–821 (2023). https://doi.org/10.1007/s12562-023-01723-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12562-023-01723-4