Abstract
We isolated Burkholderia sp. AIU M5M02, which accumulates poly(3-hydroxybutyrate) [P(3HB)] homopolymer in a nitrogen-limiting mineral salt medium containing mannitol as a sole carbon source, from a marine environment. When the isolated strain was cultured in a nitrogen-limiting mineral salt medium (pH 5.0) containing 10% (v/v) mannitol as a sole carbon source for 4 days, P(3HB) content and P(3HB) productivity reached 40 weight percent and 1.5 g/l, respectively. Thus, we demonstrated that Burkholderia sp. AIU M5M02 can be utilized for P(3HB) production from mannitol, which is a major sugar in seaweed. Furthermore, the isolate utilized not only mannitol but also gluconic acid, glycerol, starch, and sugars such as fructose, glucose, maltose, sucrose, and xylose to produce P(3HB). Based on these results, we expect to be able to apply Burkholderia sp. AIU M5M02 to the production of P(3HB) from other raw materials with low environmental loads.

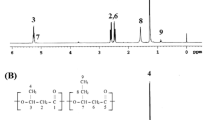
Reproduced with permission from Yamada et al.

Reproduced with permission from Yamada et al.

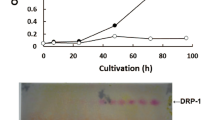
Reproduced with permission from Yamada et al.

Reproduced with permission from Yamada et al.

Reproduced with permission from Yamada et al.

Reproduced with permission from Yamada et al.
Similar content being viewed by others
References
Altschul SF, Madden TL, Schaffer AA, Zhang J, Zhang Z, Miller W, Lipman DJ (1997) Gapped BLAST and PSI-BLAST: a new generation of protein database search programs. Nucleic Acids Res 25:3389–3402
Arafiles KH, Iwasaka H, Eramoto Y, Okamura Y, Tajima T, Matsumura Y, Nakashimada Y, Aki T (2014) Value-added lipid production from brown seaweed biomass by two-stage fermentation using acetic acid bacterium and thraustochytrid. Appl Microbiol Biotechnol 98:9207–9216
Cerrone F, Davis R, Kenny ST, Woods T, O’Donovan A, Gupta VK, Tuohy M, Babu RP, O’Kiely P, O’Connor K (2015) Use of a mannitol rich ensiled grass press juice (EGPJ) as a sole carbon source for polyhydroxyalkanoates (PHAs) production through high cell density cultivation. Bioresour Technol 191:45–52
Chee JY, Tan Y, Samian MR, Sudesh K (2010) Isolation and characterization of a Burkholderia sp. USM (JCM15050) capable of producing polyhydroxyalkanoate (PHA) from triglycerides, fatty acids and glycerols. J Poly Environ 18:584–592
Chenyu D, Julia S, Wim S, Carol Lin SK (2012) Polyhydroxyalkanoates production from low-cost sustainable raw materials. Curr Chem Biol 6:14–25
Coenye T, Laevens S, Willems A, Ohlen M, Hannant W, Govan JR, Gillis M, Falsen E, Vandamme P (2001) Burkholderia fungorum sp. nov. and Burkholderia caledonica sp. nov., two new species isolated from the environment, animals and human clinical samples. Int J Syst Evol Microbiol 51:1099–1107
Doi Y (1990) Microbial polyesters. VCH, New York
Doi Y, Steinbüchel A (2001) Biopolymers. Wiley-VCH, Weinheim
Hang X, Zhang G, Wang G, Zhao X, Chen GQ (2002) PCR cloning of polyhydroxyalkanoate biosynthesis genes from Burkholderia caryophylli and their functional expression in recombinant Escherichia coli. FEMS Microbiol Lett 210:49–54
Hansson G (1983) Methane production from marine, green macro-algae. Resour Conserv 8:185–194
Hou X, From N, Angelidaki I, Huijgen WJJ, Bjerre AB (2017) Butanol fermentation of the brown seaweed Laminaria digitata by Clostridium beijerinckii DSM-6422. Bioresour Technol 238:16–21
Ito K, Hori K (1989) Seaweed: chemical composition and potential food uses. Food Rev Int 5:101–144
Jambunathan P, Zhang K (2016) Engineered biosynthesis of biodegradable polymers. J Ind Microbiol Biotechnol 43:1037–1058
Jiang G, Hill DJ, Kowalczuk M, Johnston B, Adamus G, Irorere V, Radecka I (2016) Carbon sources for polyhydroxyalkanoates and an integrated biorefinery. Int J Mol Sci 17:1157–1178
Kahar P, Tsuge T, Taguchi K, Doi Y (2004) High yield production of polyhydroxyalkanoates from soybean oil by Ralstonia eutropha and its recombinant strain. Polym Degrad Stab 83:79–86
Keenan TM, Nakas JP, Tanenbaum SW (2006) Polyhydroxyalkanoate copolymers from forest biomass. J Ind Microbiol Biotechnol 33:616–626
Lopes MS, Gomez JG, Taciro MK, Mendonca TT, Silva LF (2014) Polyhydroxyalkanoate biosynthesis and simultaneous remotion of organic inhibitors from sugarcane bagasse hydrolysate by Burkholderia sp. J Ind Microbiol Biotechnol 41:1353–1363
Mendonca TT, Gomez JG, Buffoni E, Sanchez Rodriguez RJ, Schripsema J, Lopes MS, Silva LF (2014) Exploring the potential of Burkholderia sacchari to produce polyhydroxyalkanoates. J Appl Microbiol 116:815–829
Nomura CT, Taguchi S (2007) PHA synthase engineering toward superbiocatalysts for custom-made biopolymers. Appl Microbiol Biotechnol 73:969–979
Park JI, Lee J, Sim SJ, Lee JH (2009) Production of hydrogen from marine macro-algae biomass using anaerobic sewage sludge microflora. Biotech Bioprod Eng 14:307–315
Richardson GH (1985) Standard methods for the examination of dairy products, 15th edn. American Public Health Association, Washington, DC
Rodrigues MFA, da Silva LF, Gomez JGC, Valentin HE, Steinbüchel A (1995) Biosynthesis of poly (3-hydroxybutyric acid-co-3-hydroxy-4-pentenoic acid) from unrelated substrates by Burkholderia sp. Appl Microbiol Biotechnol 43:880–886
Simon-Colin C, Raguenes G, Crassous P, Moppert X, Guezennec J (2008) A novel mcl-PHA produced on coprah oil by Pseudomonas guezennei biovar. tikehau, isolated from a “kopara” mat of French Polynesia. Int J Biol Macromol 43:176–181
Spiekermann P, Rehm BH, Kalscheuer R, Baumeister D, Steinbüchel A (1999) A sensitive, viable-colony staining method using Nile red for direct screening of bacteria that accumulate polyhydroxyalkanoic acids and other lipid storage compounds. Arch Microbiol 171:73–80
Sudesh K, Abe H, Doi Y (2000) Synthesis, structure and properties of polyhydroxyalkanoates: biological polyesters. Prog Polym Sci 25:1503–1555
Sudesh K, Bhubalan K, Chuah JA, Kek YK, Kamilah H, Sridewi N, Lee YF (2011) Synthesis of polyhydroxyalkanoate from palm oil and some new applications. Appl Microbiol Biotechnol 89:1373–1386
Taguchi S, Doi Y (2004) Evolution of polyhydroxyalkanoate (PHA) production system by “enzyme evolution”: successful case studies of directed evolution. Macromol Biosci 4:146–156
Uchida M, Miyoshi T (2013) Algal fermentation—the seed for a new fermentation industry of foods and related products. JARQ 47:53–63
Uchida M, Murata M (2004) Isolation of a lactic acid bacterium and yeast consortium from a fermented material of Ulva spp. (Chlorophyta). J Appl Microbiol 97:1297–1310
Uchida M, Amasaku H, Satoh Y, Murata M (2004) Combinations of lactic acid bacteria and yeast suitable for preparation of marine silage. Fish Sci 70:507–517
Uchida M, Murata M, Ishikawa F (2007) Lactic acid bacteria effective for regulating the growth of contaminant bacteria during the fermentation of Undaria pinnatifida (Phaeophyta). Fish Sci 73:694–704
Valentin HE, Berger PA, Gruys KJ, Rodrigues MFA, Steinbüchel A, Tran M, Asrar J (1999) Biosynthesis and characterization of poly(3-hydroxy-4-pentenoic acid). Macromolecules 32:7389–7395
Verlinden RA, Hill DJ, Kenward MA, Williams CD, Radecka I (2007) Bacterial synthesis of biodegradable polyhydroxyalkanoates. J Appl Microbiol 102:1437–1449
Weisburg WG, Barns SM, Pelletier DA, Lane DJ (1991) 16S ribosomal DNA amplification for phylogenetic study. J Bacteriol 173:697–703
Whitman WB (2015) Bergey’s manual of systematics of archaea and bacteria. Wiley Online Library, Weinheim
Wise DL, Augenstein DC, Ryther JH (1979) Methane fermentation of aquatic biomass. Resour Recovery Conserv 4:217–237
Xia A, Jacob A, Herrmann C, Tabassum MR, Murphy JD (2015) Production of hydrogen, ethanol and volatile fatty acids from the seaweed carbohydrate mannitol. Bioresour Technol 193:488–497
Yanagisawa M, Kawai S, Murata K (2013) Strategies for the production of high concentrations of bioethanol from seaweeds: production of high concentrations of bioethanol from seaweeds. Bioengineered 4:224–235
Yokoyama S, Jonouchi K, Imou K (2007) Energy production from marine biomass: fuel cell power generation driven by methane produced from seaweed. Int Schol Sci Res In 1:24–27
Zhu C, Nomura CT, Perrotta JA, Stipanovic AJ, Nakas JP (2010) Production and characterization of poly-3-hydroxybutyrate from biodiesel-glycerol by Burkholderia cepacia ATCC 17759. Biotechnol Prog 26:424–430
Acknowledgements
We thank Mr. Kimiaki Naiki (Iwate Fisheries Technology Center) for sampling of the shallow sea mud from Ofunato Bay. The work described here was supported by the Regional Innovation R&D Program (to M. Yamada) of Iwate Prefecture, Japan.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yamada, M., Yukita, A., Hanazumi, Y. et al. Poly(3-hydroxybutyrate) production using mannitol as a sole carbon source by Burkholderia sp. AIU M5M02 isolated from a marine environment. Fish Sci 84, 405–412 (2018). https://doi.org/10.1007/s12562-017-1164-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12562-017-1164-3