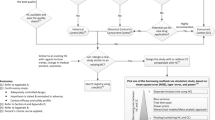
Abstract
With rising costs and prolonged timelines for drug development, more innovative trial designs are critical to improve efficiency. Use of available historical control data in a new trial can reduce the number of control patients and accordingly reduce costs and timelines. A major limitation of historical data borrowing is potential prior-data conflict. This difference can increase false decision rates and confound the outcome interpretation. The potential inflation in both type I error rate and type II error rate should be clearly characterized and controlled during the trial design stage. In this paper, we develop a simple approach to incorporating historical control data in clinical trial design and analysis. First we provide a simple statistical approach to evaluating design properties when using a Bayesian approach to incorporating historical data. We then propose a six-step process for trial design including selection and summarization of historical control data, sample size determination with and without borrowing, design property evaluation, and how much historical data should be borrowed to control false positive/negative rate inflation based on trial variability. A detailed procedure to select historical control data is also provided. Finally, we use an example to illustrate our approach. The simplicity of methodology (no simulation required), the streamlined processes for data selection, and explicit evaluation of the impact of prior-data conflict on type I error rate and power make the proposed approach statistically rigorous and easy to understand and implement.


Similar content being viewed by others
References
Viele K, Berry S, Neuenschwander B et al (2014) Use of historical control data for assessing treatment effects in clinical trials. Pharm Stat 13(1):41–54
Ibrahim JG, Chen MH (2000) Power prior distributions for regression models. Stat Sci 15:46–60
Schmidli H, Gsteiger S, Roychoudhury S, O’Hagan A, Spiegelhalter D, Neuenschwander B (2014) Robust meta-analytic-predictive priors in clinical trials with historical control information. Biometrics 70(4):1023–1032
Psioda MA, Soukup M, Ibrahim JG (2018) A practical Bayesian adaptive design incorporating data from historical controls. Stat Med. https://doi.org/10.1002/sim.7897
Duan Y, Ye K, Smith EP (2006) Evaluating water quality using power priors to incorporate historical information. Environmetrics 17(1):95–106
Ibrahim JG, Chen M-H, Gwon Y, Chen F (2015) The power prior: theory and applications. Stat Med 34(28):3724–3749
Hobbs BP, Carlin BP, Mandrekar SJ, Sargent DJ (2011) Hierarchical commensurate and power prior models for adaptive incorporation of historical information in clinical trials. Biometrics 67(3):1047–1056
Hobbs BP, Sargent DJ, Carlin BP (2012) Commensurate priors for incorporating historical information in clinical trials using general and generalized linear models. Bayesian Anal 7(3):639–674
FDA (2019) Interacting with the FDA on complex innovative trial designs for drugs and biological products. www.fda.gov/media/130897/download
Huber W, Sands BE et al (2012) Secukinumab, a human anti-IL-17A monocolonal antibody, for moderate to severe Crohn’s Disease: unexpected results of a randomized, double-blind placebo-controlled trial. Gut 61:1693–1700
Psioda MA, Ibrahim JG (2018) Bayesian clinical trial design using historical data that inform the treatment effect. Biostatistics. https://doi.org/10.1093/biostatistics/kxy009
Pocock S (1976) The combination of randomized and historical controls in clinical trials. J Chronic Dis 29:175–188
Lim J, Wang L, Best N, Liu J, Yuan J, Yong F, Zhang L, Walley R, Gosselin A, Roebling R et al (2019) Reducing patient burden in clinical trials through the use of historical controls: appropriate selection of historical data to minimize risk of bias. Ther Innov Regul Sci. https://doi.org/10.1007/s43441-019-00014-4
Neuenschwander B, Capkun-Niggli G, Branson M, Spiegelhalter DJ (2010) Summarizing historical information on controls in clinical trials. Clin Trials 7(1):5–18
Genovese MC et al (2016) Baricitinib in patients with refractory rheumatoid arthritis. N Engl J Med 374(13):1243–1252
Burmester GR et al (2013) Tofacitinib (CP-690,550) in combination with methotrexate in patients with active rheumatoid arthritis with an inadequate response to tumour necrosis factor inhibitors: a randomised phase 3 trial. Lancet 2013(381):451–460
Burmester GR et al (2013) Supplement to: Burmester GR, Blanco R, Charles-Schoeman C, et al., on behalf of the ORAL Step investigators. Tofacitinib (CP-690,550) in combination with methotrexate in patients with active rheumatoid arthritis with an inadequate response to tumour necrosis factor inhibitors: a randomised phase 3 trial. Lancet 381:451
Fleischmann R et al (2012) Placebo-controlled trial of tofacitinib monotherapy in rheumatoid arthritis. N Engl J Med 2012(367):495–507
Fleischmann R et al (2012) Supplement to: Fleischmann R, Kremer J, Cush J, et al. Placebo-controlled trial of tofacitinib monotherapy in rheumatoid arthritis. N Engl J Med 367:495–507. https://doi.org/10.1056/NEJMoa1109071
DerSimonian R, Laird N (1986) Meta-analysis in clinical trials. Control Clin Trials 7:177–188
Sidik K, Jonkman JN (2005) Simple heterogeneity variance estimation for meta-analysis. J R Stat Soc Ser C 54:367–384
Sidik K, Jonkman JN (2007) A comparison of heterogeneity variance estimators in combining results of studies. Stat Med 26:1964–1981
Lan KKG, Hu P, Proschan M (2009) A condiction power approach to the evaluation of predictive power. Stat Biopharm Res 2:131–136
Mann H, Andersohn F et al (2018) Adjusted indirect comparison using propensity score matching of Osimertinib to platinum-based doublet chemotherapy in patients with EGFRm T790M NSCLC who have progressed after EGFR-TKI. Clin Drug Investig 38(4):319–331
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
This manuscript was supported by AbbVie. AbbVie participated in the review and approval of the content. Lanju Zhang is a former AbbVie employee and is currently employed by Vertex Pharmaceuticals. Zailong Wang and Li Wang are employees of AbbVie Inc. Lu Cui is a former AbbVie employee and is currently employed by UCB Biosciences Inc. Jeremy Sokolove is a former AbbVie employee and is currently employed by GlaxoSmithKline. Ivan Chan is a former AbbVie employee and currently employed by Bristol Myers Squibb. All authors may own AbbVie stock.
Appendices
Appendix 1
which is equal to (5) with C = \({{\Phi }^{-1}\left({p}_{0}\right)=\Phi }^{-1}\left(1-\alpha \right), var\left({\widehat{\mu }}_{0,h}\right)=\frac{{\sigma }^{2}}{{n}_{0,h}}, var\left({\widehat{\mu }}_{0,c}\right)=\frac{{\sigma }^{2}}{{n}_{0,c}}, var\left({\widehat{\mu }}_{1, f}\right) =\frac{{\sigma }^{2}}{{n}_{1}}\).
Appendix 2 Select relevant historical control data
Concerns abound about using historical control data to supplement or replace a control arm in a randomized trial (Neunschwander et al., 2010; [4]. Pocock [12] proposed six criteria to evaluate whether there is adequate similarity between historical controls and in-study controls. The criteria are comprehensive and still very relevant today, and therefore are included in the following discussion.
-
i.
Patient population To ensure patient populations are similar, enrollment criteria must be compared among all selected historical trials and the new trial. Baseline characteristics including demographics and disease status, if available, should also be compared among selected historical trials.
-
ii.
Prior and concomitant therapy In many trials control patients have received/failed or are on some background therapy that may affect the response level. For example, in rheumatoid arthritis trials, patients with inadequate response to biologics may have much lower response level compared to biologics naïve patients. All prior therapy and concomitant therapies must be compared among historical trials and to the proposed allowable therapies for the control patients in the new trial.
-
iii.
Control treatment If the control is not a placebo, then the control treatment (including dosage and regimen) must be the same for the historical trials and the new trial.
-
iv.
Endpoints The patients have been evaluated similarly and the endpoint to measure the patient response must be the same. This means that they are the same measurement defined in the same way, at the same follow-up time point, and evaluated in the same fashion. If the endpoint includes some lab component, central lab reading is recommended for all trials.
-
v.
Regions Ideally the historical trials and new trials should be conducted in the same regions. If it is known that in a particular region the control response rate is significantly higher, this region should be removed or accounted for to align the response rates.
-
vi.
Contemporaneity Because of advancement of general health care, there might be a drift over time in the response rate even for the same patients. Technology may also affect patient diagnosis and endpoint assessment. So it is cautioned to include very old historical trials.
-
vii.
Any other factors For example, as mentioned in Pocock [12], significantly different enrollment rates between trials may lead to subtle difference in patient response. Another example is that there is some plausible possibility that a smaller chance to receive control in multiple-arm trials may lead to higher placebo response rate.
It should be emphasized that the relevance of the historical data or similarity among historical control and in-study control patients is not determined by the similarity between historical control and current control data. If a historical trial had the same control response as the in-study control response, but the patient populations are very different, the historical control data should not be considered relevant and thus should not be used. On the other hand, if historical control patients are deemed similar but between-trial variability is large, there might be little advantage to borrowing historical control patients.
It is acknowledged that one can never find completely identical historical control patients to match the new in-study control patients. The differences should be carefully evaluated jointly by, at minimum, statisticians and medical doctors, to make sure they will not lead to difference in response rate larger than usual variation.
If significant differences in some of the above aspects are found between historical trials, borrowing using the approach in this paper may not be applicable. This does not mean that historical control cannot be used. If patient level data are available, one can borrow the data after accounting for these different factors with covariate adjustment (Ibrahim, 2018) or propensity score matching or regression [24]. Of course, in this situation, it is hard to incorporate borrowing in the design stage since new in-trial data are not yet available. Historical trials should be selected during trial design stage and documented appropriately in the protocol or statistical analysis plan.
Rights and permissions
About this article
Cite this article
Zhang, L., Wang, Z., Wang, L. et al. A Simple Approach to Incorporating Historical Control Data in Clinical Trial Design and Analysis. Stat Biosci 14, 216–236 (2022). https://doi.org/10.1007/s12561-022-09342-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12561-022-09342-w