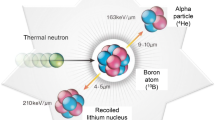
Abstract
Purpose
Several hundreds of patients have been so far treated in clinical trials with boron neutron capture therapy (BNCT).
Methods
This is a non-systematic review of clinical trials with BNCT, with special emphasis on the more recent trials.
Results
The conducted trials have been relatively small single-arm studies and included mostly the patients with head and neck carcinomas resistant to traditional treatment modalities and glioblastomas. In general, the efficacy results have been promising and BNCT has been relatively well tolerated, even in the patients who have already been treated with conventional radiotherapy or chemoradiation. The most frequent adverse events have been similar to those associated with the conventional radiotherapy. At present, there is no evidence how the efficacy of BNCT would compare to the standard treatment modalities in earlier treatment lines.
Conclusions
Most of the existing studies have been performed with reactor-based facilities, but there is now a rapidly increasing number of linear accelerator-based BNCT sites, and the clinical research is apparently activating again. This, combined with the increased knowledge on cancer biology and novel types of oncological therapies, opens possibilities to study innovative boron carriers and to combine BNCT with modern oncological therapies in the future clinical trials. To conduct larger phase III trials, multicenter approaches are encouraged to be applied, keeping in mind the importance of joint instructions and quality control measurements.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 From reactors to linear accelerators
The principle of BNCT was first presented by astrophysicist Gordon Locher in 1936, but the first clinical BNCT trials enrolling glioblastoma patients after debulking craniotomy were published not until in mid-50’s [1,2,3,4]. In these studies, no patients survived beyond one year and substantial toxicities, such oedema, necrosis, and refractory shock were recorded in all patients. Clinical BNCT trials were mostly halted until late 1960’s when a new generation of especially Japanese groups using sodium borocaptate (BSH) as a boron carrier reported trial results, again using mostly patients with glioblastoma [5,6,7,8]. The field continued as active until the millennium, after which several BNCT nuclear reactor facilities were closed. Now, there is a rapidly increasing number of linear accelerator-based BNCT sites and the clinical research is apparently activating again.
2 Overview of the efficacy and toxicity in clinical BNCT trials
So far, several hundreds of patients have been treated with BNCT using neutrons obtained from a nuclear facility and with boronophenylalanine (L-BPA) and/or BSH as the boron carrier, but the trials have been almost solely single-site studies without a comparison arm. By far most of the current evidence comes from head and neck carcinomas and glioblastomas and due to the radiobiological principles of BNCT, also other BNCT-treated tumors have been mostly superficially located. As usually in oncological trials, the development of a new method has started from the tumor types mostly refractory to standard treatments. In all conducted studies, the efficacy results have been compared to the historical controls if anything, which may potentially cause misleading interpretations of toxicity in tumor types such as metastases and melanomas, where the evidence of re-irradiation efficacy is scarce.
Although the multidisciplinary approach is especially emphasized in the treatment of head and neck cancers, external radiotherapy is one of the mainstays in the treatment of early, locally advanced and metastatic head and neck cancers. Of all tumor types, BNCT has been under the most active clinical research in head and neck carcinomas, with some exceptions specifically in tumors with squamous cell carcinoma histology. The number of patients in each trial has been low, up to 30 patients. In the reported modern phase I/II trials the efficacy has still been encouraging with objective response rates more than 70% in heavily pre-treated patients and even complete responses in more than half of the patients have been reported in several trials [9,10,11,12,13,14,15,16,17,18]. In a trial with locally recurred head and cancer and previous photon irradiation to a cumulative dose of 50 to 98 Gy, the 2-year locoregional recurrence-free survival was 27%, 2-year progression-free survival 20%, and 2-year overall survival 30% [9]. The most frequently reported adverse events across the different head and neck cancer trials have included mucositis, oral pain and fatigue, but the rate of grade 3 adverse events have been relatively low. In sum, the available data suggest BNCT as an effective and moderately well tolerated treatment in locally recurrent, inoperable head and neck cancer with relatively high response rates.
Glioblastoma is a malignancy with an aggressive behavior and with extremely rare long-term survivors. External radiotherapy with temozolomide after debulking surgery has been the major first-line treatment modality for over decade [19]. In the BNCT context, trials enrolling glioblastoma patients have recruited a larger number of patients per trial compared with e.g. the trials with head and neck cancer patients, and notably also patients without previous radiotherapy have been included in the glioblastoma trials. Although the latest published BNCT clinical trial including specifically glioblastoma patients dates back to 2011, the results have been promising and without no alarming rates of adverse events in the trials conducted in the modern BNCT era. For example, Joensuu et al. and reported 1-year overall survival rate of 61% and Hideghety et al. 50%, with study sizes of 72 and 36 patients, respectively [14, 20]. The results are consistent with more recent studies, also [21, 22]. There is also preliminary data regarding BNCT efficacy in patients with high-grade meningiomas. In a retrospective series of 44 recurrent and refractory high-grade meningiomas treated with a reactor-based BNCT, median overall survival of 29.6 months was observed [23]. Promisingly, also grade 3 recurrent and refractory meningiomas had an overall survival of 21.6 months, while in grade 2 recurrent and refractory meningiomas median overall survival was 44.0 months. These results are far better than reported from e.g. octreotide analogue and tyrosine kinase inhibitor studies in this patient population [24,25,26]. One patient in the meningioma BNCT series died of disseminated intravascular coagulation syndrome, otherwise no unexpected toxicity signals were reported either in the meningioma trials.
The treatment landscape of cutaneous melanomas has been reformed during the last decade after the successful introduction of immuno-oncological compounds, mainly checkpoint inhibitors, and BRAF/MEK inhibitors [27]. Cutaneous melanomas are considered as radioresistant tumors, but in early phase clinical trials BNCT has demonstrated substantial efficacy. The published response rates have been even tremendous with complete responses observed in 70–75% of the patients [28, 29]. Some preliminary results suggest that BNCT could be effective especially in melanoma subtypes of non-nodular histology [30]. Although skin ulcers and necrosis have been reported, long-term safety overall seems very reasonable [31].
In addition to glioblastomas, head and neck cancers and cutaneous melanomas, there have been single clinical BNCT trials e.g. in the patients with liver metastases, hepatocellular carcinomas, sarcomas and extramammary Paget’s disease in the modern BNCT era [11, 32,33,34,35,36,37]. Still, these studies have included only very few patients, which makes the interpretation of efficacy and toxicity of BNCT in these tumor types currently impossible.
3 Special issues when conducting clinical BNCT trials
As discussed above, BNCT has been demonstrated as a safe method to treat several tumor types with promising efficacy during the last 25 years in investigator-initiated trials. As new linear accelerator-based BNCT facilities are continuously built and several commercial companies are also involved in the research, larger phase II, even phase III trials are expected to be launched in the near future, which will provide BNCT a possibility to become as one of the standard treatment options in several clinical situations.
Still, there are several unsolved issues in the clinical development of BNCT. As there are still few BNCT sites globally and the slow patient recruitment has been a bottleneck in many BNCT trials, all efforts should be undertaken to develop multicenter trial approaches. This would also enhance the generalizability of the obtained results, not mentioning the unification and dissemination of good BNCT practices created when designing the trials. To conduct such multicenter studies, it is obvious that both the unified quality control and operating instructions are in the key role for their success. These include, but are not limited to boron dosing and boron measurements, joint response evaluation and the standardization of the neutron beam characteristics. The radiobiological perspectives of BNCT are not in the scope of this review, but it is obvious that complex BNCT dose distribution with low and high LET components requires special knowledge, and normalization. Similarly, outside of the primary focus of this review, the studies on the toxicity and efficacy of novel boron carriers have special considerations. The binary nature of BNCT means that novel boron carriers, without their own therapeutic effect, have to be tested at first in cancer patients without irradiation, yielding to a rare occasion in the development of oncological treatments with no potential benefit for a patient.
4 Potential combinational targets in BNCT clinical trials
In the future, in phase II-III trials BNCT could be studied to combine it with several intriguing targets. Optimally, these strategies could help to overcome the resistance mechanisms of both BNCT and the combined treatment and increase synergy without an increased toxicity. Boronated epidermal growth factor receptor (EGFR) binding antibodies have shown promising results in preclinical studies, but also very preliminary, successful clinical experiences for combining BNCT with chemoradiotherapy including EGFR antibody cetuximab have been reported [38,39,40,41]. As BNCT is effective only locally, the combination with e.g. checkpoint inhibitors may complement BNCT by eliminating the micrometastatic disease. There is no any literature available on combining BNCT with checkpoint inhibitors, although these drugs have revolutionized the treatment of BNCT-sensitive melanomas and are approved also for the treatment of locally advanced and metastatic head and neck cancers [27, 33]. Despite the lack of any clinical evidence, it is not foreseen that checkpoint inhibitors would affect to resistance mechanisms for BNCT. Instead, destroying tumor cells with BNCT could in theory lead to the increased efficacy of checkpoint inhibitors by the increased release of neoantigens [42].
In contrast to BNCT, chemotherapy is effective only in cancer cells in the proliferative phases of the cell cycle, but could also complement the efficacy of BNCT by destroying subclinical metastases. Adding BNCT to the standard of care, external radiotherapy and temozolomide, in patients with glioblastoma yielded encouraging median survival time of 21.1 months and 2-year overall survival of 45.5%, although the final results are still unpublished (NCT00974987) [21]. Several tumor-targeted boron delivery systems with chemotherapeutic agents such as doxorubicin and paclitaxel are in the early phases of the development [43, 44].
Conventional photon radiotherapy is dependent from oxygen, while BNCT needs especially sufficient and even tumor to normal tissue boron intake ratio to be effective and predictable. Hypothetically, photon radiotherapy could provide means to ameliorate dose shortages in poorly boron-intaking tumors. Still, poorly vascularized tumors are likely to have both deoxygenation and reduced boron intake. These two radiotherapy modalities have little expected cross-resistance and have been successfully combined in preclinical models [45]. In a non-randomized controlled clinical trial of 21 patients with newly diagnosed glioblastoma, BNCT followed by external radiotherapy led to a substantial improvement of median survival time compared with BNCT alone, without no worrying toxicity signals [46]. If BNCT could be beneficial to combine e.g. with hadrontherapies or FLASH radiotherapy, will likely remain as an unsolved issue for long.
5 Conclusions
The binary nature of BNCT requires special solutions for clinical BNCT research. So far, data from hundreds of patients, mainly with head and neck carcinomas, is available from single-site, controlled phase I-II clinical trials with promising results and acceptable toxicity. There is no at present evidence if BNCT would be most effective in earlier treatment lines. Currently there are only two registered BNCT trials in ClinicalTrials.gov enrolling patients (NCT05737212, NCT05601232), but the number of clinical BNCT studies is expected to be a rapidly growing field later in this decade [47]. In the nearest future, it is likely that BNCT will be tested in basket trials with several tumor types. To conduct still lacking comparative phase III trials, multicenter approaches are encouraged to be applied, keeping in mind the importance of joint instructions and quality control measurements.
Data availability
As a review article, there is no original data in this manuscript.
References
Locher G. Biological Effects and Therapeutic Possibilities of Neutrons. Am J Roentgenol Radium Ther. 1936;36:1–13.
Farr LE, Sweet WH, Locksley HB, Robertson JS. Neutron capture therapy of gliomas using boron. Trans Am Neurol Assoc. 1954;13:110–3.
Farr LE, Sweet WH, Robertson JS, Foster CG, Locksley HB, Sutherland DL, Mendelsohn ML, Stickley EE. Neutron capture therapy with boron in the treatment of glioblastoma multiforme. Am J Roentgenol Radium Ther Nucl Med. 1954;71:279–93.
Goodwin JT, Farr LE, Sweet WH, Robertson JS. Pathological study of eight patients with glioblastoma multiforme treated by neutron-capture therapy using boron 10. Cancer. 1955;8:601–15.
Asbury AK, Ojemann RG, Nielsen SL, Sweet WH. Neuropathologic study of fourteen cases of malignant brain tumor treated by boron-10 slow neutron capture radiation. J Neuropathol Exp Neurol. 1972;31:278–303.
Soloway AH, Hatanaka H, Davis MA. Penetration of brain and brain tumor. VII. Tumor-binding sulfhydryl boron compounds. J Med Chem. 1967;10:714–7.
Hatanaka H. Clinical results of boron neutron capture therapy. Basic Life Sci. 1990;54:15–21.
Hatanaka H, Nakagawa Y. Clinical-results of long-surviving brain-tumor patients who underwent boron neutron-capture therapy. Int J Radiat Oncol. 1994;28:1061–6.
Kankaanranta L, Seppala T, Koivunoro H, Saarilahti K, Atula T, Collan J, et al. Boron neutron capture therapy in the treatment of locally recurred head and-neck cancer: final analysis of a phase I/II trial. Int J Radiat Oncol Biol Phys. 2012;82:e67-75.
Kankaanranta L, Seppala T, Koivunoro H, Saarilahti K, Atula T, Collan J, et al. Boron neutron capture therapy in the treatment of locally recurred head and neck cancer. Int J Radiat Oncol. 2007;69:475–82.
Wittig A, Malago M, Collette L, Huiskamp R, Buhrmann S, Nievaart V, et al. Uptake of two 10B-compounds in liver metastases of colorectal cdenocarcinoma for extracorporeal irradiation with boron neutron capture therapy (EORTC Trial 11001). Int J Cancer. 2008;122:1164–71.
Wittig A, Collette L, Moss R, Sauerwein WA. Early clinical trial concept for boron neutron capture therapy: a critical assessment of the EORTC trial 11001. Appl Radiat Isotopes. 2009;67:S59-62.
Kouri M, Kankaanranta L, Seppala T, Tervo L, Rasilainen M, Minn H, et al. Undifferentiated sinonasal carcinoma may respond to single-fraction boron neutron capture therapy. Radiother Oncol. 2004;72:83–5.
Joensuu H, Kankaanranta L, Seppala T, Auterinen I, Kallio M, Kulvik M, et al. Boron neutron capture therapy of brain tumors: clinical trials at the finnish facility using boronophenylalanine. J Neuro-Oncol. 2003;62:123–34.
Wang LW, Wang SJ, Chu PY, Ho CY, Jiang SH, Liu YW, et al. BNCT for locally recurrent head and neck cancer: preliminary clinical experience from a phase I/II trial at tsing hua open-pool reactor. Appl Radiat Isot. 2011;69:1803–6.
Wang LW, Chen YW, Ho CY, Hsueh Liu YW, Chou FI, Liu YH, et al. Fractionated boron neutron capture therapy in locally recurrent head and neck cancer: A prospective phase I/II trial. Int J Radiat Oncol Biol Phys. 2016;95:396–403.
Hirose K, Konno A, Hiratsuka J, Yoshimoto S, Kato T, Ono K, et al. Boron neutron capture therapy using cyclotron-based epithermal neutron source and borofalan ((10)B) for recurrent or locally advanced head and neck cancer (JHN002): An open-label phase II trial. Radiother Oncol. 2021;155:182–7.
Lan TL, Chang FC, Wang CW, Igawa K, Wu SH, Lo WL, et al. Prevention and early management of carotid blowout syndrome for patients receiving head and neck salvage boron neutron capture therapy (BNCT). J Dent Sci. 2021;16:854–60.
Stupp R, Hegi ME, Mason WP, van den Bent MJ, Taphoorn MJ, Janzer RC, Ludwin SK, Allgeier A, Fisher B, Belanger K, Hau P, Brandes AA, Gijtenbeek J, Marosi C, Vecht CJ, Mokhtari K, Wesseling P, Villa S, Eisenhauer E, Gorlia T, Weller M, Lacombe D, Cairncross JG, Mirimanoff RO, European Organisation for Research and Treatment of Cancer Brain Tumour and Radiation Oncology Groups, National Cancer Institute of Canada Clinical Trials Group. Effects of radiotherapy with concomitant and adjuvant temozolomide versus radiotherapy alone on survival in glioblastoma in a randomised phase III study: 5-year analysis of the EORTC-NCIC trial. Lancet Oncol. 2009;10(5):459–66.
Hideghety K, Sauerwein W, Haselsberger K, Grochulla F, Fankhauser H, Moss R, et al. Postoperative treatment of glioblastoma with BNCT at the petten irradiation facility (EORTC Protocol 11961). Strahlenther Onkol. 1999;175:111–4.
Kawabata S, Miyatake S, Hiramatsu R, Hirota Y, Miyata S, Takekita Y, Kuroiwa T, Kirihata M, Sakurai Y, Maruhashi A, Ono K. Phase II clinical study of boron neutron capture therapy combined with X-ray radiotherapy/temozolomide in patients with newly diagnosed glioblastoma multiforme–study design and current status report. Appl Radiat Isot. 2011;69(12):1796–9.
Kageji T, Mizobuchi Y, Nagahiro S, Nakagawa Y, Kumada H. Clinical results of boron neutron capture therapy (BNCT) for glioblastoma. Appl Radiat Isot. 2011;69:1823–5.
Takai S, Wanibuchi M, Kawabata S, Takeuchi K, Sakurai Y, Suzuki M, Ono K, Miyatake SI. Reactor-based boron neutron capture therapy for 44 cases of recurrent and refractory high-grade meningiomas with long-term follow-up. Neuro Oncol. 2022;24(1):90–8.
Norden AD, Ligon KL, Hammond SN, et al. Phase II study of monthly pasireotide LAR (SOM230C) for recurrent or progressive meningioma. Neurology. 2015;84(3):280–6.
Raizer JJ, Grimm SA, Rademaker A, et al. A phase II trial of PTK787/ZK 222584 in recurrent or progressive radiation and surgery refractory meningiomas. J Neurooncol. 2014;117(1):93–101.
Kaley TJ, Wen P, Schiff D, et al. Phase II trial of sunitinib for recurrent and progressive atypical and anaplastic meningioma. Neuro Oncol. 2015;17(1):116–21.
Switzer B, Puzanov I, Skitzki JJ, Hamad L, Ernstoff MS. Managing metastatic melanoma in 2022: A clinical review. JCO Oncol Pract. 2022;18(5):335–51.
Gonzalez SJ, Bonomi MR, Cruz GAS, Blaumann HR, Larrieu OAC, Menendez P, et al. First BNCT treatment of a skin melanoma in Argentina: dosimetric analysis and clinical outcome. Appl Radiat Isotopes. 2004;61:1101–5.
Ke G, Sun Z, Shen F, Liu T, Li Y, Zhou Y. The study of physics and thermal characteristics for in-Hospital Neutron Irradiator (IHNI). Appl Radiat Isot. 2009;67:S234-237.
Fukuda H, Hiratsuka J, Kobayashi T, Sakurai Y, Yoshino K, Karashima H, Turu K, Araki K, Mishima Y, Ichihashi M. Boron neutron capture therapy (BNCT) for malignant melanoma with special reference to absorbed doses to the normal skin and tumor. Australas Phys Eng Sci Med. 2003;26(3):97–103.
Hiratsuka J, Kamitani N, Tanaka R, Tokiya R, Yoden E, Sakurai Y, Suzuki M. Long-term outcome of cutaneous melanoma patients treated with boron neutron capture therapy (BNCT). J Radiat Res. 2020;61(6):945–51.
Yanagie H, Higashi S, Seguchi K, Ikushima I, Fujihara M, Nonaka Y, et al. Pilot clinical study of boron neutron capture therapy for recurrent hepatic cancer involving the intra-arterial injection of a (BSH)-B-10-containing WOW emulsion. Appl Radiat Isotopes. 2014;88:32–7.
Hiratsuka J, Kamitani N, Tanaka R, Yoden E, Tokiya R, Suzuki M, et al. Boron neutron capture therapy for vulvar melanoma and genital extramammary paget’s disease with curative responses. Cancer Commun (Lond). 2018;38:38.
Futamura G, Kawabata S, Siba H, Kuroiwa T, Suzuki M, Kondo N, Ono K, Sakurai Y, Tanaka M, Todo T, Miyatake S. A case of radiation-induced osteosarcoma treated effectively by boron neutron capture therapy. Radiat Oncol. 2014;4(9):237.
Fujimoto T, Andoh T, Sudo T, Fujita I, Fukase N, Takeuchi T, et al. Potential of boron neutron capture therapy (BNCT) for malignant peripheral nerve sheath tumors (MPNST). Appl Radiat Isot. 2015;106:220–5.
Fujimoto T, Suzuki M, Kuratsu S, Fujita I, Morishita M, Sudo T, Sakuma T, Nakamatsu Y, Sakurai Y, Takata T, Tamari Y, Tanaka H, Masunaga SI, Kinashi Y, Kondo N, Sakakibara S, Igaki H, Andoh T, Sakamoto S, Kawamoto T, Watabe T, Hara H, Fukase N, Kawakami Y, Matsumoto T, Akisue T, Ono K, Ichikawa H, Kuroda R, Hirose T. BNCT for primary synovial sarcoma. Appl Radiat Isot. 2021;169: 109407.
Inoue M, Lee CM, Ono K, Suzuki M, Tokunaga T, Sawa Y, et al. Clinical effectiveness of boron neutron capture therapy for a recurrent malignant peripheral nerve sheath tumor in the mediastinum. J Thorac Oncol. 2010;5(12):2037–8.
Rondina A, Fossa P, Orro A, Milanesi L, De Palma A, Perico D, Mauri PL, D’Ursi P. A boron delivery antibody (BDA) with boronated specific residues: New perspectives in boron neutron capture therapy from an in silico investigation. Cells. 2021;10(11):3225.
Barth RF, Wu G, Yang W, Binns PJ, Riley KJ, Patel H, Coderre JA, Tjarks W, Bandyopadhyaya AK, Thirumamagal BT, Ciesielski MJ, Fenstermaker RA. Neutron capture therapy of epidermal growth factor (+) gliomas using boronated cetuximab (IMC-C225) as a delivery agent. Appl Radiat Isot. 2004;61(5):899–903.
Yang W, Barth RF, Wu G, Tjarks W, Binns P, Riley K. Boron neutron capture therapy of EGFR or EGFRvIII positive gliomas using either boronated monoclonal antibodies or epidermal growth factor as molecular targeting agents. Appl Radiat Isot. 2009;67(7–8 Suppl):S328–31.
Kankaanranta L, Saarilahti K, Mäkitie A, Välimäki P, Tenhunen M, Joensuu H. Boron neutron capture therapy (BNCT) followed by intensity modulated chemoradiotherapy as primary treatment of large head and neck cancer with intracranial involvement. Radiother Oncol. 2011;99(1):98–9.
Bhatia A, Burtness B. Treating Head and Neck Cancer in the Age of Immunotherapy: A 2023 Update. Drugs. 2023;83(3):217–48.
Yang Q, Dai Q, Bao X, Zhou Y, Lu Y, Zhong H, Wu L, Guo Y, Liu L, Tan X, Xia Y, Han M, Wei Q. Evaluation of a tumor-targeting oligosaccharide nanosystem in BNCT on an orthotopic hepatocellular carcinoma model. Mol Pharm. 2023;20(2):1025–38.
Xiong H, Zhou D, Qi Y, Zhang Z, Xie Z, Chen X, Jing X, Meng F, Huang Y. Doxorubicin-loaded carborane-conjugated polymeric nanoparticles as delivery system for combination cancer therapy. Biomacromol. 2015;16(12):3980–8.
Barth RF, Grecula JC, Yang W, Rotaru JH, Nawrocky M, Gupta N, Albertson BJ, Ferketich AK, Moeschberger ML, Coderre JA, Rofstad EK. Combination of boron neutron capture therapy and external beam radiotherapy for brain tumors. Int J Radiat Oncol Biol Phys. 2004;58(1):267–77.
Yamamoto T, Nakai K, Tsurubuchi T, Matsuda M, Shirakawa M, Zaboronok A, Endo K, Matsumura A. Boron neutron capture therapy for newly diagnosed glioblastoma: a pilot study in Tsukuba. Appl Radiat Isot. 2009;67(7–8 Suppl):S25–6.
ClinicalTrials.gov website. https://clinicaltrials.gov/. Accessed 22 Nov 2023.
Funding
Open Access funding provided by University of Helsinki (including Helsinki University Central Hospital). The author(s) have received no funding for this review.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical approval
No Ethical Approval was required for this review article.
Consent to participate
No Consent to Participate was required for this review article.
Consent to publish
No Consent to Publish was required for this review article.
Conflict of interests
The author(s) have no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Karihtala, P. The current status and future perspectives of clinical boron neutron capture therapy trials. Health Technol. (2024). https://doi.org/10.1007/s12553-024-00862-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12553-024-00862-7