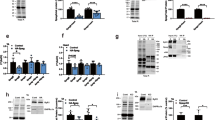
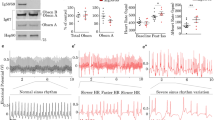
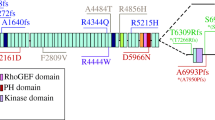
Abstract
Obscurin and its homolog, striated muscle preferentially expressed gene (SPEG), constitute a unique group of proteins abundantly expressed in striated muscles that contain two tandemly arranged MLCK-like kinases. The physiological significance of the dual kinase motifs is largely understudied; however, a collection of recent studies characterizing their binding interactions, putative targets, and disease-linked mutations have begun to shed light on their potential roles in muscle pathophysiology. Specifically, obscurin kinase 1 is proposed to regulate cardiomyocyte adhesion via phosphorylating N-cadherin, whereas SPEG kinases 1 and 2 regulate Ca2+ cycling by phosphorylating junctophilin-2 and the sarcoendoplasmic Ca2+ ATPase 2 (SERCA2). Herein, we review what is currently known regarding the potential substrates, physiological roles, and disease associations of obscurin and SPEG tandem kinase domains and provide future directions that have yet to be investigated.



Similar content being viewed by others
References
Ackermann MA, Shriver M, Perry NA, Hu LY, Kontrogianni-Konstantopoulos A (2014) Obscurins: Goliaths and Davids take over non-muscle tissues. PLoS One 9(2):e88162. https://doi.org/10.1371/journal.pone.0088162
Agrawal PB, Pierson CR, Joshi M, Liu X, Ravenscroft G, Moghadaszadeh B, Talabere T, Viola M, Swanson LC, Haliloglu G, Talim B, Yau KS, Allcock RJ, Laing NG, Perrella MA, Beggs AH (2014) SPEG interacts with myotubularin, and its deficiency causes centronuclear myopathy with dilated cardiomyopathy. Am J Hum Genet 95(2):218–226. https://doi.org/10.1016/j.ajhg.2014.07.004
Bang ML, Centner T, Fornoff F, Geach AJ, Gotthardt M, McNabb M, Witt CC, Labeit D, Gregorio CC, Granzier H, Labeit S (2001) The complete gene sequence of titin, expression of an unusual approximately 700-kDa titin isoform, and its interaction with obscurin identify a novel Z-line to I-band linking system. Circ Res 89(11):1065–1072. https://doi.org/10.1161/hh2301.100981
Benian GM, Tinley TL, Tang X, Borodovsky M (1996) The Caenorhabditis elegans gene unc-89, required fpr muscle M-line assembly, encodes a giant modular protein composed of Ig and signal transduction domains. J Cell Biol 132(5):835–848. https://doi.org/10.1083/jcb.132.5.835
Borisov AB, Raeker MO, Kontrogianni-Konstantopoulos A, Yang K, Kurnit DM, Bloch RJ, Russell MW (2003) Rapid response of cardiac obscurin gene cluster to aortic stenosis: differential activation of Rho-GEF and MLCK and involvement in hypertrophic growth. Biochem Biophys Res Commun 310(3):910–918. https://doi.org/10.1016/j.bbrc.2003.09.035
Borisov AB, Raeker MO, Russell MW (2008) Developmental expression and differential cellular localization of obscurin and obscurin-associated kinase in cardiac muscle cells. J Cell Biochem 103(5):1621–1635. https://doi.org/10.1002/jcb.21551
Catalano J, Paynton B, Kaniper S, Gerhard G, Alvarez R (2018) Identification of a novel obscurin protein variant in nonischemic cardiomyopathy. J Am Coll Cardiol 71(11)
Ferrara TM, Flaherty DB, Benian GM (2005) Titin/connectin-related proteins in C. elegans: a review and new findings. J Muscle Res Cell Motil 26(6-8):435–447. https://doi.org/10.1007/s10974-005-9027-4
Fukuzawa A, Idowu S, Gautel M (2005) Complete human gene structure of obscurin: implications for isoform generation by differential splicing. J Muscle Res Cell Motil 26(6-8):427–434. https://doi.org/10.1007/s10974-005-9025-6
Gautel M (2011) Cytoskeletal protein kinases: titin and its relations in mechanosensing. Pflugers Arch 462(1):119–134. https://doi.org/10.1007/s00424-011-0946-1
Gieseler K, Qadota H, Benian GM (2017) Development, structure, and maintenance of C. elegans body wall muscle. WormBook 2017:1–59. https://doi.org/10.1895/wormbook.1.81.2
Grater F, Shen J, Jiang H, Gautel M, Grubmuller H (2005) Mechanically induced titin kinase activation studied by force-probe molecular dynamics simulations. Biophys J 88(2):790–804. https://doi.org/10.1529/biophysj.104.052423
Grogan A, Kontrogianni-Konstantopoulos A (2019) Unraveling obscurins in heart disease. Pflugers Arch 471(5):735–743. https://doi.org/10.1007/s00424-018-2191-3
Grogan A, Hu L-YR, Ward C, Kontrogianni-Konstantopou A (2019) Loss of binding between giant obscurin and titin results in cardiac maladaptation. Biophys J 116:115a
Hsieh CM, Yet SF, Layne MD, Watanabe M, Hong AM, Perrella MA, Lee ME (1999) Genomic cloning and promoter analysis of aortic preferentially expressed gene-1. Identification of a vascular smooth muscle-specific promoter mediated by an E box motif. J Biol Chem 274(20):14344–14351. https://doi.org/10.1074/jbc.274.20.14344
Hsieh CM, Fukumoto S, Layne MD, Maemura K, Charles H, Patel A, Perrella MA, Lee ME (2000) Striated muscle preferentially expressed genes alpha and beta are two serine/threonine protein kinases derived from the same gene as the aortic preferentially expressed gene-1. J Biol Chem 275(47):36966–36973. https://doi.org/10.1074/jbc.M006028200
Hu LY, Kontrogianni-Konstantopoulos A (2013) The kinase domains of obscurin interact with intercellular adhesion proteins. FASEB J 27(5):2001–2012. https://doi.org/10.1096/fj.12-221317
Hu LR, Ackermann MA, Hecker PA, Prosser BL, King B, O'Connell KA, Grogan A, Meyer LC, Berndsen CE, Wright NT, Jonathan Lederer W, Kontrogianni-Konstantopoulos A (2017) Deregulated Ca(2+) cycling underlies the development of arrhythmia and heart disease due to mutant obscurin. Sci Adv 3(6):e1603081. https://doi.org/10.1126/sciadv.1603081
Kamm KE, Stull JT (2001) Dedicated myosin light chain kinases with diverse cellular functions. J Biol Chem 276(7):4527–4530. https://doi.org/10.1074/jbc.R000028200
Katzemich A, West RJ, Fukuzawa A, Sweeney ST, Gautel M, Sparrow J, Bullard B (2015) Binding partners of the kinase domains in Drosophila obscurin and their effect on the structure of the flight muscle. J Cell Sci 128(18):3386–3397. https://doi.org/10.1242/jcs.170639
Khapchaev AY, Shirinsky VP (2016) Myosin light chain kinase MYLK1: anatomy, interactions, functions, and regulation. Biochemistry (Mosc) 81(13):1676–1697. https://doi.org/10.1134/S000629791613006X
Kontrogianni-Konstantopoulos A, Jones EM, Van Rossum DB, Bloch RJ (2003) Obscurin is a ligand for small ankyrin 1 in skeletal muscle. Mol Biol Cell 14(3):1138–1148. https://doi.org/10.1091/mbc.e02-07-0411
Kontrogianni-Konstantopoulos A, Ackermann MA, Bowman AL, Yap SV, Bloch RJ (2009) Muscle giants: molecular scaffolds in sarcomerogenesis. Physiol Rev 89(4):1217–1267. https://doi.org/10.1152/physrev.00017.2009
Kumar S, Stecher G, Li M, Knyaz C, Tamura K (2018) MEGA X: Molecular evolutionary genetics analysis across computing platforms. Mol Biol Evol 35(6):1547–1549. https://doi.org/10.1093/molbev/msy096
Liu X, Ramjiganesh T, Chen YH, Chung SW, Hall SR, Schissel SL, Padera RF Jr, Liao R, Ackerman KG, Kajstura J, Leri A, Anversa P, Yet SF, Layne MD, Perrella MA (2009) Disruption of striated preferentially expressed gene locus leads to dilated cardiomyopathy in mice. Circulation 119(2):261–268. https://doi.org/10.1161/CIRCULATIONAHA.108.799536
Lornage X, Sabouraud P, Lannes B, Gaillard D, Schneider R, Deleuze JF, Boland A, Thompson J, Bohm J, Biancalana V, Laporte J (2018) Novel SPEG mutations in congenital myopathy without centralized nuclei. J Neuromuscul Dis 5(2):257–260. https://doi.org/10.3233/JND-170265
Mayans O, van der Ven PF, Wilm M, Mues A, Young P, Furst DO, Wilmanns M, Gautel M (1998) Structural basis for activation of the titin kinase domain during myofibrillogenesis. Nature 395(6705):863–869. https://doi.org/10.1038/27603
Puchner EM, Alexandrovich A, Kho AL, Hensen U, Schafer LV, Brandmeier B, Grater F, Grubmuller H, Gaub HE, Gautel M (2008) Mechanoenzymatics of titin kinase. Proc Natl Acad Sci U S A 105(36):13385–13390. https://doi.org/10.1073/pnas.0805034105
Qadota H, McGaha LA, Mercer KB, Stark TJ, Ferrara TM, Benian GM (2008) A novel protein phosphatase is a binding partner for the protein kinase domains of UNC-89 (Obscurin) in Caenorhabditis elegans. Mol Biol Cell 19(6):2424–2432. https://doi.org/10.1091/mbc.E08-01-0053
Qadota H, Moody JC, Lesanpezeshki L, Moncrief T, Kitzler D, Bhat PD, Vanapalli SA, Oberhauser AF, Benian GM (2020) A region of UNC-89 (obscurin) lying between two protein kinase domains is a highly elastic spring required for proper sarcomere assembly. bioRxiv:2020.2004.2003.023374. https://doi.org/10.1101/2020.04.03.023374
Qualls AE, Donkervoort S, Herkert JC, D'Gama AM, Bharucha-Goebel D, Collins J, Chao KR, Foley AR, Schoots MH, Jongbloed JDH, Bonnemann CG, Agrawal PB (2019) Novel SPEG mutations in congenital myopathies: genotype-phenotype correlations. Muscle Nerve 59(3):357–362. https://doi.org/10.1002/mus.26378
Quan C, Li M, Du Q, Chen Q, Wang H, Campbell D, Fang L, Xue B, MacKintosh C, Gao X, Ouyang K, Wang HY, Chen S (2019) SPEG controls calcium reuptake into the sarcoplasmic reticulum through regulating SERCA2a by its second kinase-domain. Circ Res 124(5):712–726. https://doi.org/10.1161/CIRCRESAHA.118.313916
Quick AP, Wang Q, Philippen LE, Barreto-Torres G, Chiang DY, Beavers D, Wang G, Khalid M, Reynolds JO, Campbell HM, Showell J, McCauley MD, Scholten A, Wehrens XH (2017) SPEG (striated muscle preferentially expressed protein kinase) is essential for cardiac function by regulating junctional membrane complex activity. Circ Res 120(1):110–119. https://doi.org/10.1161/CIRCRESAHA.116.309977
Rowland TJ, Graw SL, Sweet ME, Gigli M, Taylor MR, Mestroni L (2016) Obscurin variants in patients with left ventricular noncompaction. J Am Coll Cardiol 68(20):2237–2238. https://doi.org/10.1016/j.jacc.2016.08.052
Russell MW, Raeker MO, Korytkowski KA, Sonneman KJ (2002) Identification, tissue expression and chromosomal localization of human Obscurin-MLCK, a member of the titin and Dbl families of myosin light chain kinases. Gene 282(1-2):237–246. https://doi.org/10.1016/s0378-1119(01)00795-8
Small TM, Gernert KM, Flaherty DB, Mercer KB, Borodovsky M, Benian GM (2004) Three new isoforms of Caenorhabditis elegans UNC-89 containing MLCK-like protein kinase domains. J Mol Biol 342(1):91–108. https://doi.org/10.1016/j.jmb.2004.07.006
Stull JT, Lin PJ, Krueger JK, Trewhella J, Zhi G (1998) Myosin light chain kinase: functional domains and structural motifs. Acta Physiol Scand 164(4):471–482. https://doi.org/10.1111/j.1365-201x.1998.tb10699.x
Sutter SB, Raeker MO, Borisov AB, Russell MW (2004) Orthologous relationship of obscurin and Unc-89: phylogeny of a novel family of tandem myosin light chain kinases. Dev Genes Evol 214(7):352–359. https://doi.org/10.1007/s00427-004-0413-5
Wang H, Castiglioni C, Kacar Bayram A, Fattori F, Pekuz S, Araneda D, Per H, Erazo R, Gumus H, Zorludemir S, Becker K, Ortega X, Bevilacqua JA, Bertini E, Cirak S (2017) Insights from genotype-phenotype correlations by novel SPEG mutations causing centronuclear myopathy. Neuromuscul Disord 27(9):836–842. https://doi.org/10.1016/j.nmd.2017.05.014
Wang H, Schanzer A, Kampschulte B, Daimaguler HS, Logeswaran T, Schlierbach H, Petzinger J, Ehrhardt H, Hahn A, Cirak S (2018a) A novel SPEG mutation causes non-compaction cardiomyopathy and neuropathy in a floppy infant with centronuclear myopathy. Acta Neuropathol Commun 6(1):83. https://doi.org/10.1186/s40478-018-0589-y
Wang L, Geist J, Grogan A, Hu LR, Kontrogianni-Konstantopoulos A (2018b) Thick filament protein network, functions, and disease association. Compr Physiol 8(2):631–709. https://doi.org/10.1002/cphy.c170023
Xiong G, Qadota H, Mercer KB, McGaha LA, Oberhauser AF, Benian GM (2009) A LIM-9 (FHL)/SCPL-1 (SCP) complex interacts with the C-terminal protein kinase regions of UNC-89 (obscurin) in Caenorhabditis elegans muscle. J Mol Biol 386(4):976–988. https://doi.org/10.1016/j.jmb.2009.01.016
Xu J, Li Z, Ren X, Dong M, Li J, Shi X, Zhang Y, Xie W, Sun Z, Liu X, Dai Q (2015) Investigation of pathogenic genes in Chinese sporadic hypertrophic cardiomyopathy patients by whole exome sequencing. Sci Rep 5:16609. https://doi.org/10.1038/srep16609
Young P, Ehler E, Gautel M (2001) Obscurin, a giant sarcomeric Rho guanine nucleotide exchange factor protein involved in sarcomere assembly. J Cell Biol 154(1):123–136. https://doi.org/10.1083/jcb.200102110
Funding
This work was supported by the National Institutes of Health (Training Program in Muscle Biology, T32 AR007592-17 to A.G. and P.T.) and the American Heart Association (Grant In Aid 16GRNT31290010 to A.K.K.).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Grogan, A., Tsakiroglou, P. & Kontrogianni-Konstantopoulos, A. Double the trouble: giant proteins with dual kinase activity in the heart. Biophys Rev 12, 1019–1029 (2020). https://doi.org/10.1007/s12551-020-00715-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12551-020-00715-3