Abstract
Punta de Gavilanes (Mazarrón Bay, Spain) was occupied from third millennium cal BC to the first century BC. Overall, the archaeobotanical remains suggest that agriculture and gathering coexisted in the site since the beginning of the occupation of the site. Depending on the sea level variation, the site passed from island in the middle Holocene to peninsula in the late Holocene. Seeds from this archaeological site associated with radiocarbon dates of ≈ 4200 and ≈ 1500 cal years BP include specimens of eight species of shrubs and trees, of which a winter flowering shrub (Coronilla talaverae Lahora and Sánchez-Gómez), is an endangered species that no longer occurs on Punta de Gavilanes area. The seeds of this endemic Coronilla species are associated with materials dated ≈ 3900 cal years BP in a Bronze Age cultural context. The vanished species was presumably locally exterminated by human alteration of its natural habitat or because of environmental changes. The species of Coronilla identified from Punta de Gavilanes is known from relatively distant seashore areas. This endangered species could be effectively preserved by reintroduction to areas that it occupied prior to human alteration, in particular Punta de los Gavilanes.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The remains of fauna and plants in archaeological contexts have been used as a source of information to reconstruct past areas of different species and probable extinction events (Rivera et al. 2020). It is clear that the Earth’s ecosystems and climate are deteriorating rapidly and that much of this change is due to human activities (Rick et al. 2013). Research, especially in the western Mediterranean, has conclusively demonstrated human involvement not only in the extinction of at least some of the endemic species but also the ability to colonise environments at an early date (Mavridis 2003).
Archaeology, history, and palaeobiology offer important perspectives for modern environmental management by documenting how organisms and ecosystems functioned in the past and responded to a variety of anthropogenic and climatic changes. Returning to pristine “natural” baselines may be impossible, but archaeological records can help define a range of desirable conditions that are key components for restoring and managing ecosystems (Rick et al. 2013).
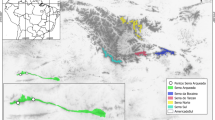
Coronilla talaverae Lahora and Sánchez-Gómez (Loteae, Leguminosae) is an endemism of the Iberian Peninsula. To date, only five localities are known, four of them located in the municipality of Pulpí (province of Almería, region of Andalusia) and the other in the municipality of Mazarrón (province and region of Murcia) in the proximity of Cañada Gallego (Fig. 1). From the biogeographical point of view, the territories where C. talaverae occurs belong to the Murcian-Almeriese chorological province, Almeriese Sector, characterised by the abundance of regional and Ibero-African endemics (Alcaraz et al. 1991; Rivas-Martínez et al. 2014, 2017; Sánchez-Gómez and Guerra 2011). This is an endemic dwarf scrub species (Fig. 2) with an extremely restricted distribution area confined to the seashore of Mazarrón and Pulpí. The plant is not currently in the study area, but grows only 5 km to the west, on the seashore, in fossil dunes (Sánchez-Gómez and Guerra, 2011: 503). The species is accepted in recent works (Charco et al. 2014, 2015) and databases (IPNI 2020; POWO 2020).
The southeast of the Iberian Peninsula possesses a rich archaeological record from Bronze Age populations, with numerous sites in Murcia and Almeria. One of these sites is Punta de los Gavilanes (37º 33′ 31.06″ N; 1º 16′ 46.53″ W) that occupies a strategic coastal promontory of dolomites and limestones entering into the sea, at 4 to 6.30 mamsl, on the coast in the Mazarrón Bay (Murcia, Spain; Figs. 1 and 3). The stratigraphy of the site pointed to four different chrono-cultural phases, later subdivided in different horizons (Ros-Sala 2017). Phase IV is the oldest identified and yielded the Coronilla seeds. It was occupied by Argaric Early Bronze and post-Argaric Late Bronze populations, who inhabited the site from twenty-second to sixteenth centuries BC (García-Martínez and Ros-Sala 2010; García-Martínez et al. 2013; Ros-Sala 2017). Phase IV was further subdivided in four different horizons (IV a, b, c and d). In addition, during Phase IV, and in addition to the exploitation of marine resources, the archaeobotanical results pointed to an abundant presence of seeds and fruits (caryopses of Poaceae comprised) in different areas and structures of the site.
Aerial view of Punta de Los Gavilanes coastal promontory and surroundings. a General view of the Gavilanes site. North and continent are in the left basal corner of the image. b Location of the site (white circle) and modern localities (red dots) of Coronilla talaverae on the coast from Mazarrón to Pulpí. Images: Gavilanes Project Archive
Whereas Argaric Early Bronze Age and Late Bronze Age populations focussed their economy mostly on the exploitation of marine resources, shellfish harvesting and fishing, later Phoenician and Punic populations, responsible for the formation of subsequent Phases III and II, focussed their economy on the metallurgy and the exploitation of the silver-rich minerals of the Mazarrón mines (Ros-Sala 2017; García-Martínez and Ros-Sala 2008, 2010). In particular, the construction of a Punic metallurgical workshop area during Phase II had a destructive impact on some of the Phase IV remains and likely on the whole peninsula and its vicinity (Ros-Sala 2017).
Remains of local endemic species are underrepresented in the archaeobotanical records of the Mediterranean region, notwithstanding the abundance there of such type of taxa. This low frequency could be due to the lack of specialised information and sufficiently complete comparison collections and data banks to allow the identification of remains of endemic species. These resources are rarely available in common identification contexts, leading archaeologists and palaeobotanists to attribute the identity of the remains to similar, widely distributed species. Therefore, this reduces the opportunity of gathering historical information for these species which is crucial for determining long-term conservation strategies. Other basic requirement is the careful absolute, radiocarbon based, and cultural datation of these remains. This will make it easier to assess the factors leading to their presence in a given period. This study offers a valuable example for similar areas in regions with a high proportion of endemics, i.e. Biodiversity Hotspots, where human presence is for a long time documented.
Our objective is to present, describe and document the discovery of seeds belonging to a species of Coronilla endemic to SE Spain, in levels culturally dated Argaric Early Bronze Age at the Punta de Los Gavilanes site and the contribution of this find to our understanding of the history of the species. We also aim to determine the extent to which other similar cases were documented. Along with the above, we intend to present the conservation status of the species and assess to what extent this finding can stimulate conservation actions.
Materials and methods
All samples were collected from different working spaces and structures dedicated to cereal processing and consumption of food within houses 1TM and 1TS and from building 2TM and 1TSM. This includes roasting ovens, milling spaces, domestic hearths, kitchen structures, an esparto basket, sediment from different floors, etc.
Seventy-six samples were recovered from all the phases (IV–I) excavated between 2004 and 2010 using standard flotation techniques. Bulk samples were processed at the Laboratory of Ethnobotany of Murcia University, through a sieve column with decreasing diameters from 2 to 0.1 mm, and then analysed in the laboratory. Optical instruments included Olympus SZ-11 trinocular stereo microscope, mounted on a SZ-STU 1 articulated arm, with a SZ-PT adapter and Leica EC3 camera. Digital images were also obtained with Arcus 1200 Agfa scanner and SEM images with a JEOL Scanning microscope JSM-6.100. The recovered fruit and seed remains were identified using the archaeobotanical comparison collection of the Ethnobotany Laboratory of Murcia University, assuming as a main hypothesis that the species list of the modern Flora of Murcia (late twentieth and twenty-first centuries; Alcaraz and Llimona 1983; Anthos 2020; Charco et al. 2015; Esteve 1973; Sánchez-Gómez and Guerra 2011) includes, among others, those species which were recovered from the site. We consulted De la Fuente et al. (2011, 2016) for Sarcocornia seeds. Nomenclature was actualised according to IPNI (2020), TPL (2020), GBIF (2020) and Anthos (2020). For plant communities, the syntaxonomical checklist of Rivas-Martínez et al. (2001) has been used. Accordingly, the authors’ names and publication dates for the syntaxa are not indicated here. English names of chorological units were adopted from Rivas-Martínez et al. (2014, 2017).
Seeds were measured using a Mitutoyo calliper. Comparison data for Coronilla seeds dimensions and differential characters were obtained from Lahora et al. (2012) and comparison with other archaeological Coronilla samples relied on Rovira (2007).
Radiocarbon dates (Online Resource, Supplementary Table 1) were calibrated with CALIB 7.1 program and version (Stuiver et al. 2018).
Microstructure of carbonised archaeological seeds was investigated by means of field emission scanning electron microscopy (FE-SEM). To obtain semi-quantitative element results, energy-dispersive X-ray spectroscopy (EDX) was conducted at the locations of interest using field emission scanning electron microscopy (SEM) with energy dispersive X-ray spectrometry (EDX).
Specimens were mounted on aluminium stubs and platinum sputter-coated with 5.0 nm thin layer (Leica EM ACE 600). Samples were examined using a FE-SEM (ApreoS Lovac IML Thermofisher) with a selected voltage of 10 kV and 0.20 nA for imaging and coupled with an Octane Plus EDAX microanalyzer (AMETEK, USA) at 20 kV for EDX analysis.
Scientific names of plants are followed along text and tables by the standard abbreviations of their author names as these appear in IPNI (2020) and POWO (2020), according to the provisions of the International Code of Nomenclature for algae, fungi, and plants (Turland et al. 2018).
To establish a comparison with modern seeds under similar conditions, we proceeded to experimentally carbonise two samples of recently collected seeds: three seeds from Fuente Álamo (Murcia) and five from San Juan de los Terreros (Pulpí, Almería) (paratype cited in the protologue). Voucher specimens are kept at the herbarium MUB of the Universidad de Murcia (NYBG 2022). Both samples were previously hydrated overnight, their moisture content was later reduced using a microwave oven at 600 W during 30 min and finally they were carbonised using an alcohol burner. The seeds were within each sample unequal in size. This led to their different behaviour. The five in the Terreros sample were smaller and two of them, the smaller ones, suffered partial or total calcination (reduced to ashes) during carbonisation.
Results and discussion
Modern Coronilla talaverae seeds
The seeds of modern Coronilla talaverae are 2.5–3.7 mm long and 0.6–1 mm of diameter, transverse cylindrical, reddish-brown (Lahora et al. 2012). These present an almost smooth seed coat and a rounded to ellipsoidal hilum with a marked hilar rim and a relatively adnate micropyle (Fig. 5). The linear median hilar fissure was closed on the seeds studied by the funicular remnants (Fig. 5). Other Coronilla species occurring in the area (Coronilla minima s.l. and C. juncea) present thicker seeds (1–1.4 mm) transversely oval, not cylindrical. Furthermore, when analysed with SEM their surfaces present differences: C. juncea shows foveolae with sparsely rugulose surface, C. minima shows deeper foveolae with a folded-rugulose surface. C. talaverae shows shallow foveolae with a striated surface (Lahora et al. 2012).
Archaeological carbonised Coronilla talaverae seeds
A total of 2643 archaeobotanical remains and 500 fragments were recovered and identified from Punta de los Gavilanes. The minimum number of individuals cannot be easily estimated in the case of plants, but in this study, the identifiable fragments of fruits and seeds usually have a volume greater than 50% of a whole fruit or seed, so they would roughly be equivalent to whole remains. Coronilla carbonised seeds were recovered from sample GV-1451, inside an esparto basket corresponding to Phase IVb (Argaric Early Bronze Age period) 3485 ± 30 (1890–1730 cal BC to 2σ/95% probability, Laboratory code KIA-43319; Fig. 4, Online Resource Supplementary Table 1). Two Coronilla talaverae seeds and one seed fragment were identified (Table 1, Fig. 5). Their dimensions (2.4 mm long × 0.7 mm diameter; 3.2 mm long × 0.9 mm diameter) and transverse cylindrical section facilitated their identification. The state of the charred seeds suggests that at the time of charring they were previously hydrated and initiating radicle protrusion (Fig. 5). This radicle was presumably destroyed during deposition, following prehistoric carbonisation, or as a result of modern recovery of archaeological materials.
Coronilla talaverae, modern and carbonised archaeological seeds. a Modern seed, SEM image, scale bar: 2 mm, arrows spot 1 hilum with marked hilar rim, 2 micropyle; b carbonised archaeological seed, optical microscope image, arrows spot 1 hilum, 2 micropyle, 3 point of seed coat rupture and radicle protrusion, scale bar: 2 mm; c carbonised archaeological seeds, optical microscope image, scale bar: 2 mm; d carbonised archaeological seed, the same as b, deteriorated during processing shows the seed coat and a portion of cotyledon, arrows spot 1 hilum, 2 area of seed coat rupture and radicle protrusion; scale bar: 2 mm. Images: a Juan Francisco Jiménez. b Diego Rivera. c and d Javier Valera
FE-SEM images allowed to analyse the microstructure of archaeological Coronilla seeds. Seeds possess a relatively smooth, with shallow foveolae (Fig. 6), 100–150 μm thick, seed coat, and a broadly ellipsoidal to rounded hilum (150 × 122 μm) with a deltoid micropyle (25 × 30 μm) relatively adnate (Fig. 6). The linear median hilar fissure, c. 85 μm long, was open on the seeds studied. The marked hilar rim was still visible, although partially destroyed, in the FE_SEM image (Fig. 6). The micropyle was closed or only slightly open. Seeds present an upper palisade layer (20 μm thick) of macrosclereids that covers the primarily regulate ornamentation sub-superficial due to the large parenchyma cells layer (c. 100 μm thick) that is only clearly visible when the upper layer is broken (Fig. 6). Butler (1989) mentions that thickness of seed coat is inversely proportional to the imbibition ability of the seed and thus related with seed dormancy. The hilum shape and structure is similar to those described by Butler (1996) from Trifolieae (Fabaceae).
Coronilla talaverae, SEM micromorphology of a carbonised archaeological seed. a Detail of seed b and d in Fig. 5, arrows spot 1 hilar zone, 2 seed coat surface and sub-superficial reticule, 3 seed coat transversal section, 4 cotyledons; b detail of hilar area, arrows spot 1 hilum, 2 micropyle, 3 hilar fissure. c Ornamentation of the seed coat surface due to the parenchyma cells layer that is only clearly visible when the upper palisade layer is broken. d Seed coat fragment showing the layers of palisade cells and a thick layer of parenchyma cells, arrow spot 1 shallow foveolae; e parenchyma cell broken showing inside the small spherical, hemispherical or star-like druses of calcium oxalate monohydrate; f seed coat fragment showing the broken raphides of calcium oxalate monohydrate. Images: a–f Maria Teresa Coronado Parra
Calcium oxalate is a common biomineral in plants, occurring as crystals of various shapes. It can be found in any tissue or organ in plants and is often formed in the vacuoles of specialised cells called crystal idioblasts (Franceschi 2001). Crystals of calcium oxalate were present in the archaeological Coronilla seeds between the palisade and parenchyma cells in form of crystal raphides or styloid of crystal in crystal idioblast cells (of different sizes to 15 μm long × 0.5 μm of diameter). Crowther (2009) described a wide range of raphides, 10 to over 300 μm long × 0.5 to 6 μm of diameter in their study on different plant families. In the parenchyma cells, oxalate is present in form of crystal druses (of different sizes to 3 μm in diameter) of monoclinic calcium oxalate monohydrate, similar to but smaller than those described by Hartl et al. (2007) in Cactaceae (Fig. 6).
The EDX spectrum of the crystal druses and nearby areas of the inner surface of the cell wall, shows a great proportion of calcium (Ca) and sodium (Na), arsenic (As) and lead (Pb) in the selected 0.25 μm areas of spot 1 (Online Resource Supplementary Fig. 1). EDS spectrum of the cell wall in the selected 0.25 μm areas of spot 1 shows lesser amounts of these elements that are surpassed by those of carbon (C) and oxygen (O). After correcting the artefacts introduced in the EDX spectrum by the use of a Pt coating to protect the samples (Online Resource Supplementary Figs. 1 and 2), only Ca remains as an element significantly present. But also C, O, As, Na, K, and in smaller quantities lead (Pb) and silver (Ag) are present.
Modern experimentally carbonised Coronilla talaverae seeds
The peculiar characteristics observed in the archaeological seeds studied (Figs. 5 and 6): slight alterations in shape with an increase in the roundness of the cross section, tissue rupture due to the emergence of the radicle and other seed tissues caused by carbonisation and slightly blurred hilum, are also observed in modern seeds subjected to experimental carbonisation (Figs. 7 and 8).
Coronilla talaverae, SEM micromorphology of experimentally carbonised modern seeds from Fuente Álamo (Murcia). a Seed, arrows spot 1 hilar zone, 2 seed coat surface, 3 seed coat transversal section, 4 integument and radicle abruptly protruding through broken seed coat; b detail of hilar area, arrows spot 1 hilum, 2 micropyle, 3 hilar fissure. c Parenchyma cell broken showing inside the small cross shaped druses of tetragonal prisms; d parenchyma cell broken showing inside the small rhomboid and some pinacoid calcium oxalate crystals; e Parenchyma cell broken showing inside the small star-like druses of calcium oxalate monohydrate, f seed coat fragment showing the layers of palisade cells and a thick layer of parenchyma cells, arrow spot 1 shallow foveolae. Images: a–f Maria Teresa Coronado Parra
Coronilla talaverae, SEM micromorphology of experimentally carbonised modern seed from San Juan de los Terreros (Pulpí, Almería). a Seed, protruding material, through the broken area; b detail of hilar area, arrows spot 1 hilum, 2 micropyle, 3 hilar fissure; c parenchyma cell broken showing inside the small horned structures; d glomeruli; e Parenchyma cell broken showing inside the small predominantly rhomboid crystals; f small crossshaped druse of tetragonal prisms and small predominantly rhomboid crystals. Images: a–f Maria Teresa Coronado Parra
FE-SEM images allowed to analyse the microstructure of modern seeds from Coronilla and to compare them with archaeological seeds recovered at Punta de Gavilanes (Table 2). Modern carbonised seeds are very similar to the archaeological ones described above, possessing a relatively smooth seed coat, with shallow foveolae, 100 μm thick (Figs. 7 and 8), and a broadly ellipsoidal to rounded hilum (130–158 × 130–147 μm) with a deltoid micropyle (25 × 30 μm) relatively adnate (Fig. 8), sometimes imperceptible (Fig. 7). The linear median hilar fissure, c. 100 μm long, was open on the seeds studied. The marked hilar rim was visible in several seeds (Fig. 8), although partially destroyed in others (Fig. 7), in the FE-SEM images. The micropyle was closed. Seeds present an upper palisade layer (20–30 μm thick) of macrosclereids (Fig. 8f) that covers the primarily regulate ornamentation sub-superficial due to the large parenchyma cells layer (c. 80 μm thick) that is only clearly visible when the upper layer is broken (Fig. 7f). The hilum shape and structure is similar to those of the archaeological seeds.
Calcium oxalate crystals were present in modern carbonised Coronilla seeds between palisade and parenchyma cells and within parenchyma cells in very diverse forms of isolated crystals, and druses (Table 2 and Figs. 7 and 8), including those previously found in archaeological samples from Punta de Gavilanes.
The EDX spectra of the crystals and other structures analysed in modern carbonised seeds offer a wide range of elements and their combinations. Calcium oxalate druses and crystals are present in modern seeds together with other crystals and structures. Some of the calcium oxalate crystals in the Fuente Alamo sample also show significant amounts of magnesium (Mg), suggesting the presence of magnesium oxalate. Hair-shaped structures (Table 2) contain predominantly carbon (C) and oxygen (O) and are only found in modern seeds. Small chloride (Cl) crystals were identified in Terreros sample. “Horned” structures (Table 2) contain predominantly chloride (Cl) and potassium (K) but also oxygen (O), being compatible with the formula of potassium chlorate. The high proportion of potassium in the druses of tetragonal prisms, accompanied of carbon and oxygen suggests these are of potassium oxalate.
The differences between modern samples and the absence of some of these structures in archaeological seeds may be due to the degree of seed maturity, genetic differences without taxonomic value (mutations), different environmental factors, post-depositional conditions of the remains or simply to changes in the remains over the nearly four thousand years of their preservation.
Oxalate levels vary along the different seed development phases in Fabaceae, in the case of soybean (Glycine max subsp. soja (Siebold & Zucc.) H.Ohashi), the highest percent of oxalate by dry mass was reached in the developing post-fertilisation seeds, and then decreased during late seed maturation (Ilarslan et al. 2001). Mutants within a species or even within a population are another factor to consider. Cellular and biochemical characterisations of the various mutants in Medicago truncatula Gaertn. have revealed mutations affecting crystal nucleation, morphology, distribution, and/or amount (Nakata 2003).
Heavy metals present in the soils of Punta de Gavilanes may have conditioned the formation of crystals, heavy metals stress reduces the deposition of calcium oxalate crystals as it was shown by Jáuregui-Zúñiga et al. (2005) in leaves of Phaseolus vulgaris. In addition, the abundance of calcium oxalate crystals in the plant appears to be directly related to the abundance of available calcium in the soil (Faheed et al. 2013).
Context for carbonised archaeological Coronilla talaverae seeds
Halophytic and subhalophytic species concentrate (185 entire and 40 fragments of seeds or fruits) in Phase IVb TM-TS (1945 cal BC–1885 cal BC to 2σ, 95% probability), where the Coronilla seeds were found. Among the annual crops, Hordeum vulgare is the most abundant species represented (Table 1). It is worth of notice that barley is the cereal crop which shows a higher tolerance to salinity (eHALOPH 2020). The lower presence of caryopses from cereals in comparison to halophytic species suggests that cereal grains had previously been retired from the esparto basket to be processed, and the remains found were indeed the wastes preserved during a subsequent carbonisation event.
Seeds of other Coronilla species have been reported from different archaeological sites and levels of southern Spain: Los Castillejos (Neolithic and Chalcolithic; Rovira 2007), Peñalosa (Bronze Age; Peña-Chocarro 1999), Los Alcores (Chalcolithic; Stika et al. 2017) and Fuente Álamo (Bronze Age; Stika 2004).
Impact of human activities
Argaric and Post-Argaric Bronze populations occupied Punta de los Gavilanes from the twenty-second to sixteenth centuries cal BC. Their economy was based on the exploitation of marine resources, shell fishing and fishing. This period was marked by an increase of population that required a wider use of local resources. In this context, these communities likely exploited the nearby non-favourable saline soils for agricultural purposes. Plants were cultivated, collected and brought to the site where they were processed and prepared for consumption.
The archaeobotanical analysis identified species, which grow in 12 different categories of habitats which can roughly be classified in 3 main groups: anthropic (weeds and crops); saline and subsaline Mediterranean habitats; and scrub and steppe (Anonymous 2013; Bartolomé et al. 2005). Anthropic habitats, as a consequence of cereal and pulse cultivation and harvest, are overrepresented in the samples.
Whereas agriculture seems to be an important activity during the Bronze period, in the following occupation periods, the archaeobotanical record is much smaller. This scarcity of archaeobotanical remains is even higher in Phase II, due to the prevalence of the protoindustrial metallurgical occupation that now covers practically the entire promontory, and to the small number of individuals, that seemingly focussed their work on metals production. Presumably, these activities led to the local extinction of Coronilla talaverae. It should be noted that the detection in the Coronilla archaeological seed of a considerable proportion of tellurium (Te) among the elements identified in the first phase of the EDX analysis should be ruled out as an artefact caused by the presence of a platinum coating. Although tellurium is recovered from, among other sources, calcined copper slag resulting from copper smelting activities (Johnston et al. 2010; Halli et al. 2020) and could have been at the site at a time later than when the seeds were deposited; it is not possible that it was at the period when site activity was predominantly agricultural and fisheries and not metallurgical.
Once established that there were no important changes of any other type, such as those in the seawater level (which could flood and eliminate nearby extensions of salt marshes and scrub), metal processing should have been the activity that most clearly affected the habitat, both for the extraction of minerals and the destruction of vegetation, used as fuel (Ros-Sala 2017). Contemporary contamination of the soils and degradation of the vegetation is suggested by palynological (Carrión et al. 2018) and anthracological results (García-Martínez and Ros-Sala 2008; García-Martínez et al. 2010, 2013). However, this extinction could have occurred later.
Several case studies, focussing on islands, reviewed by Braje et al. (2017) suggest that within a general global pattern of progressive degradation, there was no single trajectory of human impact, but rather complex effects based on variable physiographies, human subsistence strategies, population densities, technologies, sociopolitical organisation and decision-making.
Comparison with other finds of locally extinct species in littoral archaeological sites
There are hardly any cases comparable to the one mentioned here, in reference to the current absence of a wild plant species in the coastal areas where its archaeological remains have been recovered. However, this scarcity could be due to the lack of fine information for the identification of the charred remains of endemic species, which can rarely be available in general identification works (i.e. Cappers and Bekker 2013; Cappers et al. 2006; Schoch et al. 1988), for which archaeologists and palaeobotanists are forced to ascribe the identity of the remains to the closest widely distributed species. A recently published case (Martínez-Varea et al. 2019; Martínez-Varea et al. 2020) is that of Corema album (L.) D. Don ex Steudel, a shrub distributed though the Atlantic coast of continental Spain and Portugal (subsp. album) and Açores archipelago (subsp. azoricum Pinto da Silva), having a unique disjunction in the Mediterranean coast, in Serra Gelada range, near Benidorm (Alicante, region of Valencia, Spain). This site also belongs to the Murcian-Almeriese chorological province, but in a northernmost area, the Alicantese-Murcian sector. The species is currently found there on fossil dunes close to the seashore, where a very small population; last census, made by one of authors of this paper (PPFG) yielded only 11 individuals, which are the sole individuals within the Mediterranean area. The species is listed in the Valencian Catalogue of Endangered Plant Species, in the highest protection category, as Species Endangered of Extinction (Aguilella et al. 2010).
The charred seeds of Corema album have been found in Solutrean and Gravettian, 16,000–29,000 cal BP, in the archaeological site Cova de les Cendres (Teulada, Alicante), more than 25 km away from the present-day population. In this case, the vanishing of C. album, a strict psammophilous plant exclusive to sand dunes, was explained by the strong changes in the shoreline, which during the Last Glacial Maximum, coinciding with the Solutrean period, was placed 15–20 km far afield from the present one, and 120 m below the current sea water level.
Coronilla talaverae in Punta de los Gavilanes was found in much more recent levels, and from that moment to the present, no significant changes in sea level have been recorded that could explain the disappearance of the species. Furthermore, C. talaverae is not a species that strictly needs to live in saline or coastal dune areas.
Coronilla talaverae conservation issues
Coronilla talaverae occurs preferentially on carbonated substrates, especially calcarenites, marls and fossil dunes, although it can also be found in red sandstones and schists, often with slight salinity. It forms part of thickets with moderate nitrification where humidity and salt spray from the sea could play an important role, especially in communities of Thymo-Sideritidion leucanthae and Anthyllido-Salsolion papillosae within the lower thermomediterranean floor, infra-Mediterranean according to Alcaraz et al. (2008), at altitudes between 10 and 290 m. Among the companion species are: Anthyllis terniflora Pau, Coronilla juncea L., Echium humile subsp. pycnanthum (Pomel) Greuter and Burdet, Globularia alypum L., Haplophyllum rosmarinifolium (Pers.) G. Don, Helianthemum almeriense Pau, H. fontqueri Sennen, H. motae Sánchez-Gómez, J.F. Jiménez and J.B. Vera, H. syriacum (Jacq.) Dum.Cours., Periploca angustifolia Labill., Rhamnus oleoides L., Santolina viscosa Lag., Sideritis ibanyezii Pau, Teucrium carolipaui subsp. fontqueri (Sennen) Rivas Mart., T. lanigerum Lag., T. murcicum Sennen, and Thymus hyemalis Lange. It blooms and bears fruit between the months of March and June, although it can occur early or late, depending on the availability of water in the soil that year.
At the present time, five localities are known (Fig. 1; Table 3); however, in the original publication of the taxon (Lahora et al. 2012), only three localities were mentioned, all in the province of Almería. Sánchez Gómez et al. (2011) and Sánchez Gómez and Guerra (2011) mentioned the population of Mazarrón (Murcia). A new locality has recently been detected in Pilar de Jaravía (Almería) by one of us, A. Lahora.
Comparing the 2020 data with those estimated in the years of the discovery of the species (2010), a slight decrease has been observed in all known populations at that time, both in the number of mature individuals and in the quality and size of the habitat. The situation at the locality of Puerto de los Peines (Almería) is especially dramatic, where the works to adapt a viewing platform have caused a drastic decrease in the population (from 20 to 5 individuals) and it is likely at the present time locally extinct. The situation around the Castillo de Terreros and Despeñaperros, main localities, is also worrying, where recreational activities and adaptation of infrastructure related to tourism (hiking, cycling, parking, landscaping, etc.) have caused a decrease in the number of individuals, as well as quality and surface of the habitat. The population of Cañada Gallego (Murcia) presents an additional problem related to the expansion of intensive greenhouse crops; this is the one that has been most affected by the alteration of its habitat, having reduced the population by one half. It is probable that the screen effect of the greenhouses to the humidity and saline spray coming from the sea and the alteration of the soil itself, has had a very negative effect on the survival of the species in that locality.
Therefore, despite the fact that Coronilla talaverae is a species adapted to unfavourable edaphoclimatic conditions, typical of the semi-arid Iberian southeast, given the small known area and low number of individuals, and considering that potential threats and conditions continue, the species must be considered as highly threatened within the Endangered category [EN B1 ab (ii, iii, v) + 2 ab (ii, iii, v]), according to the IUCN (2012) criteria. Threat is similar to those provided by Sánchez-Gómez et al. (2011) and Lahora et al. (2012), later assumed by Gutierrez et al. (2019) and Rivers et al. (2017).
In accordance with the above, we recommend the inclusion of C. talaverae in the National Red List and in the regional, national and European Protection Catalogs. Although the population of Puerto de los Peines is within an area of the Natura 2000 Network (Site of Community Importance “ES6110012 Sierras Almagrera, de los Pinos and El Aguilón”), urgent conservation measures are needed in situ to avoid deterioration and disappearance of the habitat of the species, as well as ex situ measures, through the collection of germplasm that allows future work of population reinforcement, creation of new populations and reintroduction in those places where there are historical or archaeological evidence of its presence, such as the case of the Punta de los Gavilanes archaeological area and neighbouring peninsulas of Mazarrón Bay.
Conclusions
Coronilla talaverae needs urgent conservation measures in situ to avoid deterioration and disappearance of the habitat of the species, as well as ex situ measures, through the collection of germplasm, population reinforcement, creation of new populations and reintroduction in those places where there are historical or archaeological remains of its presence, such as Punta de los Gavilanes archaeological site and area. Overall, the results obtained point to the use of agriculture during the Argaric Early Bronze Age period as one of the main activities together with fishing. Halophytic species, including Coronilla talaverae, are mainly present in samples from earliest horizons of Bronze Age, suggesting a higher dependence on saline and subsaline habitats from 3560 ± 25 to 3425 ± 30 BP.
Our study reveals the opportunities offered by cooperation in multidisciplinary teams of archaeologists, archaeobotanists, plant taxonomists and conservationists and their contribution to the conservation of local endemics. In addition, it will help promote initiatives for the conservation of local endemics not only for their biological but also cultural value. This small peninsula offers an appropriate place for reintroducing Coronilla talaverae within a micro-reserve associated to the musealisation of the site.
It is important in archaeobotanical studies in the Mediterranean area to consider the possibility of the presence of local endemic species and, consequently, to develop comparison and identification tools that can help in this task (including macro and micromorphology). This approach would be useful to better understand the evolutionary history of Mediterranean biodiversity.
A remarkable aspect of the present work is to show the taxonomic and environmental valuable data that can be obtained from FE-SEM studies of the microstructures and crystals of archaeological seeds and fruits, as well as EDX spectra, which can be carried out taking advantage of the good number of identifiable fragments generated during the excavation and the study process in the laboratory, without destroying whole seeds.
References
Aguilella, A., S. Fos, and E. Laguna, eds. 2010. Catalogo Valenciano de Especies de Flora Amenazadas. Valencia: Generalitat Valenciana.
Alcaraz, F., and X. Llimona. 1983. Flora Cormofítica de la Sierra de la Pila (Murcia, SE. de España I). Anales De Ciencias 34: 109–153.
Alcaraz, F., P. Sánchez-Gómez, and A. De La Torre. 1991. Biogeografía de la provincia Murciano–Almeriense hasta el nivel de subsector. Rivasgodaya 6: 77–100.
Alcaraz, F., J.A. Barreña, M. Clemente, J. González- Garnés, J. López- Bernal, D. Rivera, and S. Ríos. 2008. Manual de interpretación de los Hábitats Naturales y Seminaturales de la Región de Murcia. Tomo 1: hábitats y sistemas de hábitats. Murcia: Dirección General del Medio Natural, Consejería de Desarrollo Sostenible y Ordenación del Territorio, Región de Murcia.
Anonymous. 2013. Interpretation manual of European Union Habitats. Version EUR 28. Bruxelles: European Commission. DG Environment. https://ec.europa.eu/environment/nature/legislation/habitatsdirective/docs/Int_Manual_EU28.pdf. Accessed 8 Oct 2020.
Anthos. 2020. Anthos. Sistema de información sobre las plantas de España. http://www.anthos.es/. Accessed 18 Oct 2020.
Bartolomé, C., J. Álvarez, J. Vaquero, M. Costa, M. Casermerio, J. Giraldo, and J. Zamora. 2005. Los tipos de hábitat de interés comunitario en España. Guía básica Ministerio de Medio Ambiente. Madrid: Dirección General para la Biodiversidad.
Braje, T., T. Leppard, S. Fitzpatrick, and J. Erlandson. 2017. Archaeology, historical ecology and anthropogenic island ecosystems. Environmental Conservation 44 (3): 286–297.
Butler, A. 1989. 24 Cryptic anatomical characters as evidence of early cultivation in the grain legumes (pulses). In Foraging and farming: the evolution of plant exploitation, ed. D. Harris and G. Hillman, 390–407. London: Unwin Hyman.
Butler, A. 1996. Trifolieae and related seeds from archaeological contexts: problems in identification. Vegetation History and Archaeobotany 5 (1/2): 157–167.
Cappers, R., and R. Bekker. 2013. A Manual for the identification of plant seeds and fruits. Groeningen Archaeological Studies no. 23. Groningen: Barkhuis Publishing.
Cappers, R., R. Bekker, and J. Jans. 2006. Digitale Zadenatlas van Nederland/Digital seed atlas of the Netherlands. Groningen: Barkhuis Publishing.
Carrion, J., E. Fierro, M. Ros, M. Munuera, S. Fernández, J. Ochando, G. Amorós, F. Navarro, T. Rodríguez-Estrella, S. Manzano, P. González-Sampériz, and A. Moreno. 2018. Ancient Forests in European drylands: holocene palaeoecological record of Mazarrón, south-eastern Spain. Proceedings of the Geologist’s Association 129: 512–525.
Charco, J., M. Becerra, C. Santa-Bárbara, C. Fernández, F. Pérez-García, E. Triano, M. Vizoso, and L. Baena. 2014. Árboles y arbustos autóctonos de Andalucía. Ciudad Real: CIAMED.
Charco, J., F. Alcaraz, F. Carrillo, and D. Rivera. 2015. Árboles y arbustos autóctonos de la Región de Murcia. Murcia: CIAMED.
Crowther, A. 2009. Morphometric analysis of calcium oxalate raphides and assessment of their taxonomic value for archaeological microfossil studies. In Archaeological science under a microscope: studies in residue and ancient DNA analysis in honour of Thomas H. Loy, ed. M. Haslam, G. Robertson, A. Crowther, S. Nugent, and L. Kirkwood, 102–128. Canberra: Australian National University e Press.
De la Fuente, V., L. Nieto, and D. Sánchez-Mata. 2011. Sarcocornia hispanica (Chenopodiaceae), a new species from the Iberian Peninsula. Lazaroa 32: 9–13.
De La Fuente, V., L. Rufo, N. Rodríguez, D. Sánchez-Mata, A. Franco, and R. Amils. 2016. A study of Sarcocornia AJ Scott (Chenopodiaceae) from Western Mediterranean Europe. Plant Biosystems 150 (2): 343–356.
eHALOPH. 2020. eHALOPH—Halophytes Database, Version 3.20. https://www.sussex.ac.uk/affiliates/halophytes/index.php. Accessed 24 Sep 2020.
Esteve, F. 1973. Vegetación y flora de las regiones central y meridional de la provincia de Murcia. Murcia: CEBAS-CSIC.
Faheed, F., A. Mazen, and S. Abd Elmohsen. 2013. Physiological and ultrastructural studies on calcium oxalate crystal formation in some plants. Turkish Journal of Botany 37 (1): 139–152.
Franceschi, V. 2001. Calcium oxalate in plants. Trends in Plant Science 6 (7): 331.
García-Martínez, M., and M. Ros-Sala. 2008. El paisaje vegetal pre- y protohistórico de la costa de Mazarrón (Murcia) según el antracoanálisis de Punta de Los Gavilanes. Cuaternario y Geomorfología 22 (3–4): 107–120.
García-Martínez, M., and M. Ros-Sala. 2010. Gestión del combustible leñoso e impacto medioambiental asociados a la metalurgia protohistórica de Punta de Los Gavilanes (Mazarrón, Murcia). Trabajos De Prehistoria 67–2: 545–559.
García-Martínez, M., E. Grau, and M. Ros-Sala. 2013. Woody plants in the semi-arid South-eastern Iberian during the Bronze Age: charcoal analysis from Punta de Los Gavilanes (Mazarrón, Murcia, Spain). In: Damblon F. (ed.) Proceedings of the Fourth International Meeting of Anthracology 2008, BAR International Series 2486. British Archaeological Reports, Oxford, pp. 103–112.
GBIF. 2020. GBIF, Global Diversity Information Facility. https://www.gbif.org. Accessed 18 Oct 2020.
Gutiérrez, L., J. Fuentes, M. Cueto, and G. Blanca. 2019. Top ten de las plantas más amenazadas de Andalucía Oriental: Taxones endémicos y no endémicos. Acta Botanica Malacitana 44: 5–33.
Halli, P., B.P. Wilson, T. Hailemariam, P. Latostenmaa, K. Yliniemi, and M. Lundström. 2020. Electrochemical recovery of tellurium from metallurgical industrial waste. Journal of Applied Electrochemistry 50 (1): 1–14.
Hartl, W.P., H. Klapper, B. Barbier, H.J. Ensikat, R. Dronskowski, P. Müller, G. Ostendorp, A. Tye, R. Bauer, and W. Barthlott. 2007. Diversity of calcium oxalate crystals in Cactaceae. Botany 85 (5): 501–517.
Ilarslan, H., R.G. Palmer, and H.T. Horner. 2001. Calcium oxalate crystals in developing seeds of soybean. Annals of Botany 88 (2): 243–257.
IPNI. 2020. International Plant Name Index (IPNI). https://www.ipni.org/. Accessed 18 Oct 2020.
IUCN. 2012. IUCN red list categories and criteria: Version 3.1. Gland and Cambridge: IUCN Species Survival Commission.
Jáuregui-Zúñiga, D., M.A. Ferrer, A.A. Calderón, R. Muñoz, and A. Moreno. 2005. Heavy metal stress reduces the deposition of calcium oxalate crystals in leaves of Phaseolus vulgaris. Journal of Plant Physiology 162 (10): 1183–1187.
Johnston, M.D., S. Jahanshahi, L. Zhang, and F.J. Lincoln. 2010. Effect of slag basicity on phase equilibria and selenium and tellurium distribution in magnesia-saturated calcium iron silicate slags. Metallurgical and Materials Transactions B 41 (3): 625–635.
Lahora, A., P. Sánchez-Gómez, and J. Martínez. 2012. A new species of Coronilla (Loteae, Fabaceae) from Southeastern Spain: evidence from morphological and molecular data. Folia Geobotanica 47 (3): 317–335.
Martínez-Varea, C. 2020. Gathering in the dunes Seeds and fruits from the Gravettian levels of Cova de les (Cendres Teulada-Moraira, Alicante Spain). Journal of Archaeological Science Reports. https://doi.org/10.1016/j.jasrep.2020.102540.
Martínez-Varea, C., P. Ferrer-Gallego, M. Raigón, E. Badal, I. Ferrando, E. Laguna, C. Real, D. Román, and V. Villaverde. 2019. Corema album archaeological remains in Western Mediterranean Basin. Assessing fruit consumption during Upper Palaeolithic in Cova de les Cendres (Alicante, Spain). Quaternary Science Reviews 207: 1–12. https://doi.org/10.1016/j.quascirev.2019.01.0044.
Mavridis, F. 2003. Early island archaeology and the extinction of endemic fauna in the eastern Mediterranean: Problems of interpretation and methodology. British School at Athens Studies 9: 65–74.
Nakata, P.A. 2003. Advances in our understanding of calcium oxalate crystal formation and function in plants. Plant Science 164 (6): 901–909.
NYBG. 2022. Index Herbariorum, MUB, Universidad de Murcia. http://sweetgum.nybg.org/science/ih/herbarium-details/?irn=126299. Accessed 8 Feb 2022.
Peña-Chocarro, L. 1999. Prehistoric Agriculture in Southern Spain during the Neolithic and the Bronze Age. The application of ethnographic models, BAR, International Series, 818. Oxford: British Archaeological Reports.
POWO. 2020. Plants of the World Online, Coronilla talaverae Lahora and Sánchez-Gómez. http://powo.science.kew.org/taxon/77122084-1. Accessed 18 Oct 2020.
Rick, T., P. Kirch, J. Erlandson, and S. Fitzpatrick. 2013. Archeology, deep history, and the human transformation of island ecosystems. Anthropocene 4: 33–45.
Rivas-Martínez, S., F. Fernández-González, J. Loidi, M. Lousã, and A. Penas. 2001. Syntaxonomical checklist of vascular plant communities of Spain and Portugal to association level. Itinera Geobotanica 14: 5–341.
Rivas-Martínez, S., A. Penas, T. Díaz, S. Del Río, P. Cantó, L. Herrero, C. Pinto, and J. Costa. 2014. Biogeography of Spain and Portugal, Preliminary typological synopsis. International Journal of Geobotanical Research 4: 1–64.
Rivas-Martínez, S., A. Penas, T. Díaz, P. Cantó, S. del Río, J. Costa, L. Herrero, and J. Molero. 2017. Biogeographic units of the Iberian Peninsula and Balearic Islands to District level. A concise synopsis. In The Vegetation of the Iberian Peninsula, vol. 1, ed. J. Loidi, 131–188. Dordrecht: Springer International Publishing. https://doi.org/10.1007/978-3-319-54784-8_5.
Rivera, D., J. Abellán, J. Palazón, C. Obón, F. Alcaraz, E. Carreño, E. Laguna, A. Ruiz, and D. Johnson. 2020. Modelling ancient areas for date palms (Phoenix species: Arecaceae): Bayesian analysis of biological and cultural evidence. Botanical Journal of the Linnean Society 193 (2): 228–262.
Rivers, M., P. García-Murillo, A. Buira, and P. Fraga-i-Arquimbau. 2017. Coronilla talaverae. The IUCN Red List of Threatened Species 2017: e.T96426740A96427057. https://doi.org/10.2305/IUCN.UK.2017-3.RLTS.T96426740A96427057.en (accessed 25 Sep 2020).
Ros-Sala, M. 2017. Nuevos datos en torno a la presencia fenicia en la bahía de Mazarrón (Sureste Ibérico). In El Oriente de Occidente: Fenicios y púnicos en el área ibérica, ed. F. Sala and F. Prados, 79–104. Alicante: Universidad de Alicante Servicio de Publicaciones.
Rovira, N. 2007. Agricultura y gestión de los recursos vegetales en el SE de la Península Ibérica durante la Prehistoria Reciente. Barcelona: Universitat Pompeu Fabra. PhD Thesis. https://www.tdx.cat/handle/10803/7468. Accessed 16 Jan 2019.
Sánchez-Gómez, P., and J. Guerra, eds. 2011. Nueva Flora de Murcia Plantas Vasculares, 3rd ed. Murcia: Diego Marín.
Sánchez-Gómez, P., J. Jiménez, J. Vera, D. López, A. Lahora, J. Mota, and S. Talavera. 2011. Estado de conservación de tres nuevos edafoendemismos del Sureste Ibérico. In Programa y Libro de Resúmenes V Congreso de Biología de la Conservación de Plantas, 65. Menorca: UIB/SEBICOP.
Schoch, W., B. Pawilk, and F. Schweingruber. 1988. Botanische Makroreste. Ein Atlas zur Bestimmung häufig gefundener und ökologisch wichtiger Pflanzesamen—Botanical macro-remains—Macrorestes botaniques. Bern and Stuttgart: Paul Haupt.
Stika, H. 2004. Resultados arqueobotánicos de la campaña de 1988 en Fuente Álamo. In Fuente Álamo Las excavaciones arqueológicas 1977–1991 en el poblado de la edad del Bronce, ed. H. Schubart, V. Pingel, and O. Arteada, 183–222. Sevilla: Monografías de Arqueología Junta de Andalucía.
Stika, H., M. Berihuete, I. Stoll, and A. Heiss. 2017. Restos vegetales prehistóricos y romanos del área de Porcuna (Jaén, Andalucía). In Miscelánea en homenaje a Lydia Zapata Peña (1965–2015), 246–268. Bilbao: Universidad del País Vasco.
Stuiver, M., P. Reimer, and R. Reimer. 2018. CALIB 7.1 [WWW program] at http://calib.org. Accessed 18 Oct 2018.
TPL. 2020. The Plant List. A working list of all plant species. www.theplantlist.org. Accessed 18 Oct 2018.
Turland, N., J. Wiersema, F. Barrie, W. Greuter, D. Hawksworth, P. Herendeen, S. Knapp, W. Kusber, D. Li, K. Marhold, T. May, J. McNeill, A. Monro, J. Prado, M. Price, and G. Smith, (eds). 2018. International Code of Nomenclature for algae, fungi, and plants (Shenzhen Code) adopted by the Nineteenth International Botanical Congress Shenzhen, China, July 2017. Regnum Vegetabile 159. Glashütten: Koeltz Botanical Books. https://doi.org/10.12705/Code.2018.
Acknowledgements
We deeply acknowledge Juan Bautista Pérez, for the preparation of the map, and Pedro Solano, for having provided us with the location of the Murcia population. We deeply appreciate the meaningful corrections from our reviewers and editors Julian Wiethold, Dieter Uhl, Michael Reich and Ferran Antolin.
Funding
Open Access funding provided thanks to the CRUE-CSIC agreement with Springer Nature. This work received financial support from the Seneca Foundation—Science and Technology Agency of Region of Murcia (Spain) (Project 17998), and the Economy and Competitiveness Minister of Spain (Project HAR2017-85726-C2-1-P).
Author information
Authors and Affiliations
Contributions
All the authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by DR, MR and MLP for archaeological materials and by PS and AL for field data. JV and M-TC were responsible of carbonised seed images. The first draft of the manuscript was written by DR and all the authors commented on previous versions of the manuscript. All the authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors disclose all relationships or interests that could have direct or potential influence or impart bias on the work.
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Handling Editor: Dieter Uhl.
Supplementary Information
Below is the link to the electronic supplementary material.
12542_2022_626_MOESM1_ESM.docx
Supplementary Fig. 1. EDX spectra for different parts of the Coronilla archaeological seed analyzed by FE-SEM/EDX (DOCX 1465 kb)
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Rivera, D., Ros-Sala, M., Obón, C. et al. Seeds of Coronilla talaverae (Fabaceae), an endemic endangered species, in Argaric Early Bronze Age levels of Punta de Gavilanes (Mazarrón, Spain). PalZ 97, 1–17 (2023). https://doi.org/10.1007/s12542-022-00626-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12542-022-00626-6