Abstract
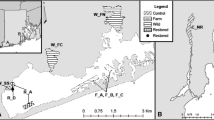
Two native species of Crassostrea cupped oysters, C. rhizophorae and C. gasar, are economically important in Brazil. However, C. gasar genetic resources are poorly characterized. This study assesses and characterizes genetic variation within and between oyster banks along the southeast coast of the state of São Paulo, Brazil, to provide baseline data useful for conservation and management. To correctly distinguish C. gasar from C. rhizophorae, we screened PCR–RFLP markers based on variation of the AluI restriction digest banding pattern. Genetic variation within C. gasar was assessed using a panel of nine microsatellite DNA markers. Intrapopulation analysis revealed low allelic richness (4.50 to 5.31 alleles per locus), deficit of heterozygotes, high rates of inbreeding, and segregation of null alleles in all populations. At one of the five sampling sites, a bottleneck effect was inferred in the only population that exhibited no private alleles. In silico analysis revealed four genetic groups using DAPC and DEST genetic differentiation index, showing moderate to high population genetic structure (DEST 0.05–0.34) with gene flow. The Mantel test did not show a significant correlation (P = 0.27) between geographic and genetic distances. The results indicate that the stocks of C. gasar have low genetic variability with moderate genetic structuring. The establishment of an exotic species (Saccostrea cucullata) may have reduced Crassostrea populations in the oyster banks studied, and loss of genetic diversity may be indicators that further study and timely action are needed to preserve native genetic resources of this essential marine food resource.



Similar content being viewed by others
References
Ajana AM (1980) Fishery of the mangrove oyster, Crassostrea gasar, Adanson (1757), in the Lagos area, Nigeria. Aquaculture 21(2):129–137. https://doi.org/10.1016/0044-8486(80)90021-6
Amaral VS, Simone LRL (2014) Revision of genus Crassostrea (Bivalvia: Ostreidae) of Brazil. J Mar Biol Assoc UK 94(4):811–836. https://doi.org/10.1017/S0025315414000058
Amaral VS, Simone LRL, Tâmega FTS, Barbieri E, Calazans SH, Coutinho R, Spotorno-Oliveira P (2020) New records of the non-indigenous oyster Saccostrea cucullata (Bivalvia: Ostreidae) from the southeast and south Brazilian coast. Reg Stud Mar Sci 33:100924. https://doi.org/10.1016/j.rsma.2019.100924
An HS, Kim WJ, Lim HJ, Byun SG, Hur YB, Park JY, Myeong JI, An CM (2014) Genetic structure and diversity of Crassostrea gigas in Korea revealeed from microsatellite markers. Biochem Syst Ecol 55:283–291. https://doi.org/10.1016/j.bse.2014.03.039
Astanei I, Gosling E, Wilson JIM, Powell E (2005) Genetic variability and phylogeography of the invasive zebra mussel, Dreisseina polymorpha (Pallas). Molec Ecol 14(6):1655–1666. https://doi.org/10.1111/j.1365-294X.2005.02530.x
Avolio ML, Beaulieu JM, Lo EY, Smith MD (2012) Measuring genetic diversity in ecological studies. Plant Ecol 213:1105–1115. https://doi.org/10.1007/s11258-012-0069-6
Baltazar-Soares M, Biastoch A, Harrod C, Hanel R, Marohn L, Prigge E, Evans D, Bodles K, Behrens E, Böning CW, Eizaguirre C (2014) Recruitment collapse and population structure of the European eel shaped by local ocean current dynamics. Curr Biol 24(1):104–108. https://doi.org/10.1016/j.cub.2013.11.031
Bardakci F, Skibinski DOF (1994) Application of the RAPD technique in tilapia fish: species and subspecies identification. Heredity 73:117–123. https://doi.org/10.1038/hdy.1994.110
Barrett RD, Schluter D (2008) Adaptation from standing genetic variation. Trends Ecol Evol 23(1):38–44. https://doi.org/10.1016/j.tree.2007.09.008
Barton NH, Slatkin MA (1986) Quasi-equilibrium theory of the distribution of rare alleles in a subdivided population. Heredity 56:409–415. https://doi.org/10.1038/hdy.1986.63
Beck MW, Brumbaugh RD, Airoldi L, Carranza A, Coen LD, Crawford C, Defeo O, Edgar GJ, Hancock B, Kay MC, Lenilhan HS, Luckenbach MW, Toropova CL, Zhang G, Guo X (2011) Oyster reefs at risk and recommendations for conservation, restoration, and management. Bioscience 61(2):107–116. https://doi.org/10.1525/bio.2011.61.2.5
Bernatchez S, Xuereb A, Laporte M, Benestan L, Steeves R, Laflamme M, Bernatchez L, Mallet MA (2019) Seascape genomics of eastern oyster (Crassostrea virginica) along the Atlantic coast of Canada. Evol Appl 12(3):587–609. https://doi.org/10.1111/eva.12741
Caddy J F, Defeo O (2003) Enhancing or restoring the productivity of natural populations of shellfish and other marine invertebrate resources. FAO Fisheries Technical Paper. No. 448. Rome, FAO
Castric V, Bernatchez L, Belkhir K, Bonhomme F (2002) Heterozygote deficiencies in small lacustrine populations of brook charr Salvelinus fontinalis Mitchill (Pisces, Salmonidae): a test of alternative hypotheses. Heredity 89:27–35. https://doi.org/10.1038/sj.hdy.6800089
Cavaleiro NP, Lazoski C, Tureck CR, Melo CMR, Amaral VS, Lomovasky BJ, Solé-Cava AM (2019) Crassostrea talonata, a new threat to native oyster (Bivalvia: Ostreidae) culture in the Southwest Atlantic. J Exp Mar Biol Ecol 511:91–99. https://doi.org/10.1016/j.jembe.2018.11.011
Cordes JF, Xiao J, Reece KS (2008) Discrimination of nine Crassostrea oyster species based on restriction fragment-length polymorphism (RFLP) analysis of nuclear and mitochondrial DNA markers. J Shellfish Res 27(5):1155–1161. https://doi.org/10.2983/0730-8000-27.5.1155
Dąbrowski MJ, Pilot M, Kruczyk M, Żmihorski M, Umer HM, Gliwicz J (2014) Reliability assessment of null allele detection: inconsistencies between and within different methods. Mol Ecol Resour 14(2):361–373. https://doi.org/10.1111/1755-0998.12177
Dąbrowski MJ, Bornelöv S, Kruczyk M, Baltzer N, Komorowski J (2015) ‘True’null allele detection in microsatellite loci: a comparison of methods, assessment of difficulties and survey of possible improvements. Mol Ecol Resour 15(3):477–488
Darrigran G, Damborenea C (2011) Ecosystem engineering impacts of Limnoperna fortunei in South America. Zoolog Sci 28(1):1–7. https://doi.org/10.2108/zsj.28.1
Dewoody JA, Avise JC (2000) Microsatellite variation in marine, freshwater and anadromous fishes compared with other animals. J Fish Biol 56:461–473. https://doi.org/10.1111/j.1095-8649.2000.tb00748.x
Di Rienzo A, Peterson AC, Garza JC, Valdes AM, Slatkin M, Freimer NB (1994) Mutational processes of simple-sequence repeat loci in human populations. Proc Nat Acad Sci USA 91(8):3166–3170. https://doi.org/10.1073/pnas.91.8.3166
Erse EB, Bernardes MA (2008) Survey of stocks of the oyster Crassostrea sp in natural banks on the Paraná coast. Biotemas 21(2):57–63. https://doi.org/10.5007/2175-7925.2008v21n2p5. (In Portuguese)
Faraco LFD, Andriguetto-Filho JM, Daw T, Lana PDC, Teixeira CF (2016) Vulnerability among fishers in southern Brazil and its relation to marine protected areas in a scenario of declining fisheries. Desenvolv Meio Ambiente 38(1):51–76. https://doi.org/10.5380/dma.v38i0.45850
FAO (Food and Agriculture Organization of the United Nations) (2015) The Second Report on the State of the World’s Animal Genetic Resources for Food and Agriculture. Scherf BD, Pilling D (ed) FAO Commission on Genetic Resources for Food and Agriculture Assessments, FAO, Rome. https://doi.org/10.4060/I4787E
FAO (Food and Agriculture Organization of the United Nations) (2019) The state of the world’s aquatic genetic resources for food and agriculture. FAO Commission on Genetic Resources for Food and Agriculture. FAO, Rome. https://www.fao.org/documents/card/en/c/ca5345en/. Accessed 20 Apr 2022
FAO (Food and Agriculture Organization of the United Nations) (2021) Fisheries and aquaculture. http://www.fao.org/fishery/statistics/global-aquaculture-production/query/en. Acessed 07 Jul 2021
FAO (Food and Agriculture Organization of the United Nations) (2022) Sustainable development goals. https://www.fao.org/sustainable-development-goals/en/. Accessed 23 May 2022
Galvão MSN, Alves PMF, Hilsdorf AWS (2017) First record of the Saccostrea oyster in Bertioga, São Paulo, Brazil. Bol Inst Pesca 43(4):638–645. https://doi.org/10.20950/1678-2305.2017v43n4p638
Galvão MSN, Hilsdorf AWS (2015) Assessing the genetic diversity of the mangrove oyster Crassostrea rhizophorae (Bivalvia, Ostreidae) by microsatellite markers in southeastern Brazil. Mar Biol Res 11(9):944–954. https://doi.org/10.1080/17451000.2015.1047381
Galvão MSN, Pereira OM, Hilsdorf AWS (2013) Molecular identification and distribution of mangrove oysters (Crassostrea) in an estuarine ecosystem in Southeast Brazil: implications for aquaculture and fisheries management. Aquac Res 44(10):1589–1601. https://doi.org/10.1111/j.1365-2109.2012.03166.x
Galvão MSN, Pereira OM, Machado IC, Pimentel CMM, Henriques MB (2009) Desempenho da criação da ostra de mangue Crassostrea sp. A partir da fase juvenil, em sistema suspenso, no estuário de Cananéia e no mar de Ubatuba (SP, Brasil). Bol Inst Pesca 35(3):401–411
Gilg MR, Hilbish TJ (2003) The geography of marine larval dispersal: coupling genetics with fine-scale physical oceanography. Ecology 84(11):2989–2998. https://doi.org/10.1890/02-0498
Guo X, Li Q, Wang QZ, Kong LF (2012) Genetic mapping and QTL analysis of growth-related traits in the Pacific Oyster. Mar Biotechnol 14:218–226. https://doi.org/10.1007/s10126-011-9405-4
Hargreaves AL, Germain RM, Bontrager M, Persi J, Angert AL (2020) Local adaptation to biotic interactions: a meta-analysis across latitudes. Amer Nat 195(3):395–411. https://doi.org/10.1086/707323
Hedgecock D, Gaffney PM, Goulletquer P, Guo X, Reece K, Warr GW (2005) The case for sequencing the Pacific oyster genome. J Shellfish Res 24(2):429–441. https://doi.org/10.2983/0730-8000(2005)24[429:TCFSTP]2.0.CO;2
Holm S (1979) A Simple sequentially rejective multiple test procedure. Scand J Stat 6(2):65–70. http://www.jstor.org/stable/4615733
Hughes AR, Hanley TC, Byers JE, Grabowski JH, Malek JC, Piehler MF, Kimbro DL (2016) Genetic by environmental variation but no local adaptation in oysters (Crassostrea virginica). Ecol Evol 7(2):697–709. https://doi.org/10.1002/ece3.2614
Hurwood DA, Heasman MP, Mather PB (2005) Gene flow, colonization and demographic history of the flat oyster Ostrea angasi. Mar Freshw Res 56(8):1009–1106. https://doi.org/10.1071/MF04261
Ignacio BL, Absher TM, Lazoski C, Solé-Cava AM (2000) Genetic evidence of the presence of two species of Crassostrea (Bivalvia: Ostreidae) on the coast of Brazil. Mar Biol 136(6):987–991. https://doi.org/10.1007/s002270000252
Jombart T, Devillard S, Balloux F (2010) Discriminant analysis of principal components: a new method for the analysis of genetically structured populations. BMC Genet 11(94). https://doi.org/10.1186/1471-2156-11-94
Jost L (2008) GST and its relatives do not measure differentiation. Mol Ecol 17:4015–4026. https://doi.org/10.1111/j.1365-294X.2008.03887.x
Kalinowski ST (2004) Counting allele with rarefaction: private alleles and hierarchical sampling designs. Conserv Genet 5:539–545. https://doi.org/10.1023/B:COGE.0000041021.91777.1a
Kamvar ZN, Brooks JC, Grünwald NJ (2015) Novel R tools for analysis of genome-wide population genetic data with emphasis on clonality. Front Genet 6(208):1–10. https://doi.org/10.3389/fgene.2015.00208
Keenan K, Mcginnity P, Cross TF, Crozier WW, Prodöhl PA (2013) diveRsity: an R package for the estimation of population genetics parameters and their associated errors. Methods Ecol Evol 4(8):782–788. https://doi.org/10.1111/2041-210X.12067
Kimura M, Weiss G (1964) The stepping stone model of population structure and the decrease of genetic correlation with distance. Genetics 49(4):561–576. https://doi.org/10.1093/genetics/49.4.561
Kinlan BP, Gaines SD (2003) Propagule dispersal in marine and terrestrial environments: a community perspective. Ecology 84(8):2007–2020. https://doi.org/10.1890/01-0622
Laing I, Walker P, Areal F (2006) Return of the native – is European oyster (Ostrea edulis) stock restoration in the UK feasible? Aquat Living Res 19(3):283–287. https://doi.org/10.1051/alr:2006029
Lam K, Morton B (2003) Mitochondrial DNA and morphological identification of a new species of Crassostrea (Bivalvia: Ostreidae) cultured for centuries in the Pearl River Delta, Hong Kong. China Aquaculture 228(1–4):1–13. https://doi.org/10.1016/S0044-8486(03)00215-1
Lazoski CV (2004) Sistemática molecular e genética populacional de ostras brasileiras (Crassostrea spp.). 2004. Ph.D. dissertation, Instituto de Biologia, Universidade Federal do Rio de Janeiro
Lazoski C, Gusmão J, Boudry P, Solé-Cava AM (2011) Phylogeny and phylogeography of Atlantic oyster species: evolutionary history, limited genetic connectivity and isolation by distance. Mar Ecol Prog Ser 426:197–212. https://doi.org/10.3354/meps09035
Legat AP, Oliveira JA, Lazoski CVS, Solé-Cava AM, Melo CMR, Galvéz AO (2009) Caracterização genética de ostras nativas do gênero Crassostrea no Brasil: base para o estabelecimento de um programa nacional de melhoramento. Embrapa Meio-Norte, 1st edn, Teresina, Piaui, Documentos, 192
Legat JFA, Puchnick-Legat A, Fogaça FHS, Tureck CR, Suhnel S, Melo CMR (2017) Growth and survival of bottom oyster Crassostrea gasar cultured in the northeast and south of Brazil. Bol Inst Pesca 43(2):72–184. https://doi.org/10.20950/1678-2305.2017v43n2p172
Li A, Dai H, Guo X, Zhang Z, Zhang K, Wang C, Wang X, Wang W, Chen H, Li X, Zheng H, Li L, Zhang G (2021) Genome of the estuarine oyster provides insights into climate impact and adaptive plasticity. Commun Biol 4:1287. https://doi.org/10.1038/s42003-021-02823-6
Li C, Wang H, Guo X (2017) Classification and taxonomic revision of two oyster species from Peru: Ostrea megodon (Hanley, 1846) and Crassostrea talonata (Li & Qi,1994). J Shellfish Res 36(2):359-364. https://doi.org/10.2983/035.036.0208
López-Cortegano E, Pouso R, Labrador A, Pérez-Figueroa A, Fernández J, Caballero A (2019) Optimal management of genetic diversity in subdivided populations. Front Genet 10:843. https://doi.org/10.3389/fgene.2019.00843
Luikart G, Sherwin W, Steele B, Allendorf F (1998) Usefulness of molecular markers for detecting population bottlenecks via monitoring genetic change. Mol Ecol 7:963–974. https://doi.org/10.1046/j.1365-294x.1998.00414.x
Ma H, Li L, Xiao S, Zhang Y, Yu Z (2021) Microsatellite-based study of population genetics of Crassostrea hongkongensis in Southern China. Aquac Rep 19:100591. https://doi.org/10.1016/j.aqrep.2021.100591
Mallet AL, Zouros E, Gartner-Kepkay KE, Freeman KR, Dickie LM (1985) Larval viability and heterozygote deficiency in populations of marine bivalves: Evidence from pair matings of mussels. Mar Biol 87:165–172. https://doi.org/10.1007/BF00539424
Marshall TC, Slate JBKE, Kruuk LEB, Pemberton JM (1998) Statistical confidence for likelihood-based paternity inference in natural populations. Mol Ecol 7:639–655
Martínez L, Méndez J, Insua A, Arias-Pérez A, Freire R (2013) Genetic diversity and population differentiation in the cockle Cerastoderma edule estimated by microsatellite markers. Helgol Mar Res 67(1):179–189. https://doi.org/10.1007/s10152-012-0314-3
McAfee D, Connell SD (2021) The global fall and rise of oyster reefs. Front Ecol Environ 19(2):118–125. https://doi.org/10.1002/fee.2291
Melo AGC, Varela ES, Beasley CR, Schneider H, Sampaio I, Gaffney PM, Reece KS, Tagliaro CH (2010a) Molecular identification, phylogeny and geographic distribution of Brazilian mangrove oysters (Crassostrea). Genet Mol Biol 33(3):564–572. https://doi.org/10.1590/S1415-47572010000300030
Melo CMR, Silva FC, Gomes CHAM, Solé-Cava AM, Lazoski C (2010b) Crassostrea gigas in natural oyster banks in southern Brazil. Biol Invasions 12:441–449. https://doi.org/10.1007/s10530-009-9475-7
Merk V, Colsoul B, Pogoda B (2020) Return of the native: survival, growth and condition of European oysters reintroduced to German offshore waters. Aquat Cons Mar Freshwat Ecosyst 30(11):2180–2190. https://doi.org/10.1002/aqc.3426
Miller PA, Elliott NG, Koutoulis PDK, Vaillancourt RE (2012) Genetic diversity of cultured, naturalized, and native Pacific oysters, Crassostrea gigas, determined from multiplexed microsatellite markers. J Shellfish Res 31(3):611–617. https://doi.org/10.2983/035.031.0303
Palumbi SR (1994) Genetic divergence, reproductive isolation, and marine speciation. Annu Rev Ecol Syst 25(1):547–572. https://doi.org/10.1146/annurev.es.25.110194.002555
Palumbi SR (1996) Nucleic acids II: the polymerase chain reaction. In: Hillis DM, Moritz C, Mable BK Molecular Systematics, 2nd ed., Sinauer Associates, Inc., Sunderland, Massachusetts, pp 205-247
Peakall R, Smouse PE (2012) GenAlEx 6.5: Genetic analysis in excel. Population genetic software for teaching and research-an update. Bioinformatics 28(19):2537–2539. https://doi.org/10.1093/bioinformatics/bts460
Pereira OM, Henriques MB, Machado IC (2003) Estimativa da curva de crescimento da ostra Crassostrea brasiliana em bosques de mangue e proposta para sua extração ordenada no estuário de Cananéia, SP. Brasil Bol Inst Pesca 29(1):19–28
Pie MR, Ribeiro RO, Boeger WA, Ostrensky A, Falleiros RM, Angelo L (2006) A simple PCR-RFLP method for the discrimination of native and introduced oyster species (Crassostrea brasiliana, C. rhizophorae and C. gigas; Bivalvia: Ostreidae) cultured in Southern Brazil. Aquac Res 37(15):1598–1600. https://doi.org/10.1111/j.1365-2109.2006.01591.x
Pilling D, Bélanger J, Diulgheroff S, Koskela J, Leroy G, Mair G, Hoffmann I (2020) Global status of genetic resources for food and agriculture: challenges and research needs. Genet Resour 1(1):4–16. https://doi.org/10.46265/genresj.2020.1.4-16
Piry SG, Luikart G, Cornuet JM (1999) BOTTLENECK: a computer program for detecting recent reductions in the effective population size using allele frequency data. J Hered 90(4):502–503. https://doi.org/10.1093/jhered/90.4.502
R Core Team (2015) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org/.
Ravit B, Cooper K, Buckley B, Comi M, McCandlish E (2014) Improving management support tools for reintroducing bivalve species (Eastern oyster [Crassostrea virginica Gmelin]) in urban estuaries. Integ Environ Assess Manag 10(4):555–565. https://doi.org/10.1002/ieam.1553
Raymond M, Rousset F (1995) GENEPOP (version 1.2): population genetics software for exact tests and ecumenicism. J Hered 86(3):248–249. https://doi.org/10.1093/oxfordjournals.jhered.a111573
Reed DH, Frankham R (2003) Correlation between fitness and genetic diversity. Conserv Biol 17(1):230–237. https://doi.org/10.1046/j.1523-1739.2003.01236.x
Rose CG, Paynter KT, Hare MP (2006) Isolation by distance in the eastern oyster, Crassostrea virginica, in Chesapeake Bay. J Hered 97(2):158–170. https://doi.org/10.1093/jhered/esj019
Rousset F (2008) Genepop’007: a complete reimplementation of the Genepop software for Windows and Linux. Mol Ecol Resour 8(1):103–106. https://doi.org/10.1111/j.1471-8286.2007.01931.x
Russello MA, Amato G (2004) Ex situ population management in the absence of pedigree information. Mol Ecol 13(9):2829–2840. https://doi.org/10.1111/j.1365-294X.2004.02266.x
Sampaio DS, Tagliaro CH, Scheneider H, Beasley CR (2017) Oyster culture on the Amazon mangrove coast: asymmetries and advances in an emerging sector. Revi Aquac 11(1):88–104. https://doi.org/10.1111/raq.12227
Santos NMV, Sousa-Neto APS, Cunha FEA, Fernandes CAF (2016) The extractive production of Crassostrea spp in river delta region Parnaíba, Brazil. Rev Bras Engen Pesca 9(1):1–11. https://doi.org/10.18817/repesca.v9i1.1104. (in Portuguese)
Sato M, Kawamata K, Zaslavskaya N, Nakamura A, Ohta T, Nishikiori T, Brykov V, Nagashima K (2005) Development of microsatellite markers for Japanese scallop (Mizuhopecten yessoensis) and their application to a population genetic study. Mar Biotechnol 7(6):713–728. https://doi.org/10.1007/s10126-004-0127-8
Schuelke M (2000) An economic method for the fluorescent labeling of PCR fragments. Nat Biotechnol 18(2):233–234. https://doi.org/10.1038/72708
Shanks AL (2009) Pelagic larval duration and dispersal distance revisited. Biol Bull 216(3):373–385. https://doi.org/10.1086/BBLv216n3p373
Silliman K (2019) Population structure, genetic connectivity, and adaptation in the Olympia oyster (Ostrea lurida) along the west coast of North America. Evol Appl 12(5):923–939. https://doi.org/10.1111/eva.12766
Silva PM, Scardua MP, Vieira CB, Alves AC, Dungan CF (2015) Survey of pathologies in Crassostrea gasar (Adanson, 1757) oysters from cultures and wild populations in the São Francisco estuary, Sergipe, Northeast Brasil. J Shellfish Res 34:289–296. https://doi.org/10.2983/035.034.0210
Simpson SD, Harrison HB, Claereboudt MR, Planes S (2014) Long-distance dispersal via ocean currents connects Omani clownfish populations throughout entire species range. PLoS One 9(9):107610. https://doi.org/10.1371/journal.pone.0107610
Thorpe JP, Solé-Cava AM (1994) The use of allozyme electrophoresis in invertebrate systematics. Zool Scr 23(1):3–18. https://doi.org/10.1111/j.1463-6409.1994.tb00368.x
Tureck CR, de Melo CMR, Gomes CHAM, Lazoski C, Marenzi AWC, Ferreira JPR, Ferreira JF (2020) Use of artificial collectors to obtain oyster seeds in Babitonga Bay, Santa Catarina, Brazil. Bol Inst Pesca 46(1):541. https://doi.org/10.20950/1678-2305.2020.46.1.541
Vandewoestijne S, Schtickzelle N, Baguette M (2008) Positive correlation between genetic diversity and fitness in a large, well-connected metapopulation. BMC Biol 6:1–11. https://doi.org/10.1186/1741-7007-6-46
Van Oosterhout C, Hutchinson WF, Wills DPM, Shipley P (2004) MICROCHECKER: software for identifying and correcting genotyping errors in microsatellite data. Mol Ecol Notes 4(3):535–538. https://doi.org/10.1111/j.1471-8286.2004.00684.x
Varela ES, Beasley CR, Schneider H, Sampaio I, Marquessilva NS, Tagliaro CH (2007) Molecular phylogeny of mangrove oysters (Crassostrea) from Brazil. J Molluscan Stud 73(3):229–234. https://doi.org/10.1093/mollus/eym018
Vendrami DL, Peck LS, Clark MS, Eldon B, Meredith M, Hoffman JI (2021) Sweepstake reproductive success and collective dispersal produce chaotic genetic patchiness in a broadcast spawner. Sci Adv 7(37):p.eabj4713. https://doi.org/10.1126/sciadv.abj4713
Vera M, Carlsson J, Carlsson JE, Cross T, Lynch S, Kamermans P, Villalba A, Culloty S, Martinez P (2016) Current genetic status, temporal stability and structure of the remnant wild European flat oyster populations: conservation and restoring implications. Mar Biol 163:239. https://doi.org/10.1007/s00227-016-3012-x
Wang J (2011) Coancestry: a program for simulating, estimating and analyzing relatedness and inbreeding coefficients. Mol Ecol Resour 11(1):141–145. https://doi.org/10.1111/j.1755-0998.2010.02885.x
Ward RD, Woodwark M, Skibinski DOF (1994) A comparison of genetic diversity levels in marine, freshwater, and anadromous fishes. J Fish Biol 44(2):213–232. https://doi.org/10.1111/j.1095-8649.1994.tb01200.x
Williams GC (1975) Sex and evolution, 8th edn. Princeton University Press, Princeton, NJ
Wright S (1978) Evolution and the Genetics of Populations, volume 4: variability within and among natural populations (Vol. 4). University of Chicago Press, Chicago.
Xia J, Yu Z, Kong X (2009) Identification of seven Crassostrea oysters from the South China Sea using PCR–RFLP analysis. J Molluscan Stud 75(2):139–147. https://doi.org/10.1093/mollus/eyp001
Xu Q, Zheng J, Yan X, Nie H (2020) Genetic diversity and differentiation of nine populations of the hard clam (Meretrix petechialis) assessed by EST-derived microsatellites. Electron J Biotechnol 48:23–28. https://doi.org/10.1016/j.ejbt.2020.09.003
You FM, Huo N, Gu YQ, Luo MC, Ma Y, Hane D, Lazo GR, Dvorak J, Anderson OD (2008) BatchPrimer3: a high throughput web application for PCR and sequencing primer design. BMC Bioinformatics 9(1):1–13. https://doi.org/10.1186/1471-2105-9-253
Yu Z, Guo X (2003) Genetic linkage map of the eastern oyster Crassostrea virginica Gmelin. Biol Bull 204(3):327–338. https://doi.org/10.2307/1543603
Zerbino DR, Birney E (2008) Velvet: algorithms for de novo short read assembly using de Bruijn graphs. Genome Res 18(5):821–829. https://doi.org/10.1101/gr.074492.107
Zhao X, Shi H, Yu H, Li Q (2015) Characterization of polymorphic microsatellite markers and genetic diversity in the Hong Kong oyster Crassostrea hongkongensis using paired-end Illumina shotgun sequencing. Genes Genom 37(7):615–620. https://doi.org/10.1007/s13258-015-0291-4
Acknowledgements
The authors thank Dr. Helcio Luis de Almeida Marques and all fishers who kindly assisted in field work for sampling and Dr. Ricardo Sartorello for the assistance in the map design. Also, we would like to thank the reviewers for thoughtful comments and efforts towards improving our manuscript. This work was developed as part of the full requirements for the Master of Science thesis of J.R. Monteiro in Biotechnology at the University of Mogi das Cruzes (UMC).
Funding
The work was support by grants from the São Paulo Research Foundation (FAPESP) (2016/16108–3) and the Foundation for Supporting Teaching and Research (FAEP). A.W.S. Hilsdorf is recipient of CNPq productivity scholarships (309570/2021–2).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Ethical approval
All applicable international, national, and/or institutional guidelines for animal testing, animal care and use of animals were followed by the authors.
Sampling and field studies
The fieldwork has been conducted in compliance with all applicable regulations.
Data availability
The microsatellite sequences are available on GenBank (MZ385602-MZ385610). All other data are available at request to the authors.
Author contribution
MSNG and AWSH conceived and organized the project; JRM, MSNG, and LRM performed the field and bench work, CAP carried out the in silico analysis of the data, AWSH, MSNG, and EMH wrote the manuscript, and all authors commented on the previous versions of the manuscript. All authors read and approved the final manuscript.
Additional information
Communicated by C. Chen
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Monteiro, J.R., Galvão, M.S.N., Perazza, C.A. et al. Assessment of genetic resources of the mangrove oyster Crassostrea gasar along the south and southeast coast of Brazil: baseline for its conservation and sustainable use. Mar. Biodivers. 53, 57 (2023). https://doi.org/10.1007/s12526-023-01367-8
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12526-023-01367-8