Abstract
This paper aims to provide a broad diet reconstruction for people buried in archaeologically defined contexts in Rome (first to third centuries CE), in order to combine archaeological and biological evidence focusing on dietary preferences in Imperial Rome. A sample of 214 human bones recovered from 6 funerary contexts was selected for carbon and nitrogen stable isotope analysis. The baseline for the terrestrial protein component of the diet was set using 17 coeval faunal remains recovered from excavations at Rome supplemented by previously published data for the same geographic and chronological frames. δ13C ranges from − 19.9 to − 14.8‰, whereas δ15N values are between 7.2 and 10.0‰. The values are consistent with an overall diet mainly based on terrestrial resources. All the human samples rely on a higher trophic level than the primary consumer faunal samples. Certainly, C3 plants played a pivotal role in the dietary habits. However, C4 plants also seem to have been consumed, albeit they were not as widespread and were not always used for human consumption. The environment played a critical role also for Romans of lower social classes. The topographical location determined the preferential consumption of food that people could obtain from their neighborhood.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Imperial Rome was one of the largest cities of Europe (Scheidel 2007; Lo Cascio 1994), and feeding its population was a severe concern for political authorities. Demographic surveys witness a peak in both urban and suburban Roman populations during the Imperial Age (first to third centuries CE, herein indicated by the capitalized word “Empire,” whereas the uncapitalized word “empire” refers to the geographical boundaries, as suggested by Boatwright et al. (2011)), revealing that about one million people lived in the city or within 50 km. Nearly 17% of the Italian population was concentrated in just 5% of Italian territory (Morley 1996; Scheidel 2009), which affected public health, administrative and social organization (Dyson 2010).
Roman authorities began to step in the food supply of the city in the mid-Republican period. The introduction of grain distribution by C. Sempronius Gracchus in 123 BCE is considered the first legal provision for supplying the citizens of Rome. According to this rule, each legal resident was entitled to receive a monthly allotment of essential foods at a discounted price or even for free. Because wheat supplied most of the calories citizens consumed, the government focused its interventions in the wheat market, especially for the poor, although meat and oil were also distributed in later years. Eligibility for the food allotment required an ever-watchful eye by the authorities. From the second half of the first century BCE, the names of those entitled to receive the frumentatio were recorded in dedicated registers. However, eligibility for the provision could also be acquired by donation or by the purchase of the frumentaria card, the tablet on which the eligible citizen’s name was engraved.
In the Principate, the Annona (the grain supply) was a critical element of the relationship between the Emperor and the citizens, and an influential political leader headed this office. Beyond the imperial estates’ production, the empire collected tax grain primarily in Sicily and Africa. Two ancient authors provide the best indication of the amount involved in this trade: Aurelius Victor, in his Liber de Caesaribus, reported that under Augustus, Rome annually received ca. 135,000 tons from Egypt, while Flavius Josephus in his De Bello Judaico, told us that under Agrippa II, the North African colonies granted Rome for up to 400,000 tons of grain. The obtained stock was distributed at the frumentationes, which fed a large part of the population but not its entirety. The primary conditions for accessing the public supply were Roman citizenship, residence in Rome, being male, and legal age, though there were many exceptions (Johnson 2013). The massive amount of grain imported and the strict regulation for its distribution clearly demonstrate that Rome depended on a grain supply, and if it were cut off, it would face hardship and even famine. Of course, the food requirements of Rome could not be fulfilled only by the central distribution of supplementary grain, and Roman social stratification in the city and suburbs created many related problems.
Archaeological evidence suggests the area outside the city walls, the Suburbium, was inhabited both by poor people, who could not afford the city lifestyle, and the upper strata of Roman society (Champlin 1982). Pliny the Younger in his Epistulae (Plin. Ep. 2.17) celebrated the countryside, where people wanted to spend their lives outside the unhealthy urbanized environment. However, this liminal area between the city and the open countryside also included marginal industries excluded from the city for religious or public safety reasons, such as landfills, quarry pits, brickmaking facilities, and funerary areas (Killgrove and Tykot 2013; Catalano 2015). Movements between the Urbs and the Suburbium were frequent, and the permanent Rome-ward migration from the countryside helped maintain the population size of Rome (Scheidel 2007). The migration to Rome was already testified by thousands of grave inscriptions and notorious contemporary authors such as Lucius Annaeus Seneca in his Ad Helviam matrem de consolatione (6.2-3) or Decimus Iunius Iuvenalis’s Satirae (3). However, the biomolecular analysis is currently supporting this topic, suggesting that the Rome population size was granted by people with different origins and cultural features (Killgrove and Tykot 2013; Antonio et al. 2019), including their dietary habits.
Roman diet was and continues to represent a fertile area of investigation, and the historical record provides a great deal of evidence of the variety of foodstuffs available to at least some of the Roman people. The broadest discussion of the diet of ancient Romans is provided by primary sources, such as novels and artworks (Purcell 2003; Wilkins and Hill 2006).
Several Latin authors handed information on dietary habits in ancient Rome, starting from the Republican time. One of the earliest treatises dealing with this topic was Cato’s De Agricultura. To provide the best moral behaviors to his son, he gave dietary prescriptions forming the basis for many of the recipes found in the following literary sources, such as the Marcus Terentius Varro’s De Re Rustica. However, the ancient Roman agronomist Columella left the most massive amount of information on agricultural techniques and food processing for the Imperial Age in his De Re Rustica, which represents the leading literary source for understanding the diet in that period. Pliny the Elder’s Naturalis Historia also provides us with a historical perspective of the Imperial age diet, as he described both faunal and horticultural landscapes.
Furthermore, Petronius and Apicius were the primary sources on Roman cuisine with their Satyricon and De Re Coquinaria, respectively, where hundreds of recipes were collected, and Martial’s Epigrams.
Food was a popular motif also in the decoration of Roman estates, where wealthy Romans enjoyed a fully catered lifestyle, especially in rooms associated with food consumption, such as kitchens and dining rooms (Yardley 1991). However, these luxury items were undoubtedly mainly produced by and for the upper social stratum, representing less than 2% of the population.
According to these primary sources, grain would have been the base of diet for Romans. This is not surprising since carbohydrates from grains would have accounted for about 70% of their daily energy intake (Delgado et al. 2017). Grain was used in various recipes, mainly as bread or puls, a grain pottage that could also be mixed with vegetables, meat, and cheese (Garnsey 1999). As previously stated, cereals were widely cultivated in the empire, and consistent importation came from Sicily and Egypt areas. The commercial value of grain was determined by the Edict of Diocletian, which set the maximum price of wheat, barley, and millet. Remarkably, the role of millet is still not completely understood, and it might have been mainly used for livestock fodder rather than for human sustenance (Spurr 1983) even though Dioscorides, a Roman physician, pharmacologist and botanist of Greek origin, mentioned the panicum in his 5-volume De Materia Medica. The pivotal role of cereals in the Empire is also attested by evidence concerning Roman skill in ensuring a continuous supply of those foodstuffs through diverse agricultural practices, artificial farming techniques, and food preservation methods (De Ligt 2006).
Along with the cereal backbone, a wide variety of vegetables, fruits, and legumes were eaten (or drunk, as in the case of wine) by Romans (Garnsey 1999; Prowse 2001).
Certainly, meat represented a critical element of an individual’s food consumption: livestock breeding and trade were rampant in the Roman world (Kron 2002; MacKinnon 2004) and the primary sources of meat were goats, sheep, lambs, and pigs (Brothwell and Brothwell 1998; MacKinnon 2004). Varro told us about a price list for the meat (Res Rust. 2.5.11), that clearly distinguished purchase for butchery from purchase for sacrifice, even though their consumption was not common amid the populace, as also referred by Pliny the Elder in his Historia Naturalis (“ex horto plebei macellum, quanto innocentiore”, that could be translated as “the populace market resides in the garden, carrying the simplest food”; HN 19.52). Furthermore, the role of fish in the Empire is unclear as this foodstuff was alternatively seen as an expensive or a common food (Purcell 2003) in various ecological contexts. In a simplistic view, preserved and fresh fish consumption was dependent on the social constraints (Marzano 2018). Generally, the consumption of certain types of fresh fish conferred status, as also reported in Juvenal’s Satura 4, whereas the exploitation of preserved and salty fish was more affordable across low social strata, as Cato reported in his De Agricultura (88).
According to Galen, marine fish were more highly valued than freshwater fish (De Alimentorum Facultatibus), and their consumption in ancient Rome increased with garum, the staple fish sauce.
Information about the Roman diet could also be provided by mounting archaeobotanical evidence found at roughly coeval sites, such as the floral remains from Pompeii and Herculaneum (Rowan 2017). Similarly, recovered faunal remains suggest the types of meat and fish available to Romans (King 1999; Cool 2006; Prowse et al. 2004, 2005).
The evaluation of human bone remains recovered in archaeological contexts could provide an even clearer glimpse into the lives of the people who lived and died in Rome. Indeed, human bones play a critical role in evaluating a community’s subsistence strategy through carbon and nitrogen stable isotope analysis of bone collagen (De Niro 1985; Ambrose and Norr 1993; O'Brien 2015).
Carbon and nitrogen isotopic analysis of collagen recruited through a bulk sampling of the bones reflects the latest years of life due to the turnover rate (Tsutaya and Yoneda 2015; Fahy et al. 2017). Carbon and nitrogen signatures derive primarily from consumed foodstuff and could, therefore, act as proxies to identify the diet. The carbon isotope ratio could be roughly used to differentiate between the consumption of plants with different photosynthetic pathways (C3 vs. C4) (Tykot 2014) or differentiate between terrestrial-based and marine-based resources in a C3 plant-based environment (Tykot 2014). Conversely, nitrogen isotope values provide information about the trophic level of an individual with an offset of 3–5‰ being detectable rising through the trophic levels. However, several confounding factors should be borne in mind in that evaluation. Metabolic and physiological processes could bias the straightforward relationship between stable isotopes and diet reconstruction (Bocherens et al. 1994; Cherel et al. 2005; Mekota et al. 2006; Waters-Rist and Katzenberg 2010; Pecquerie et al. 2010; Reitsema 2013; O’Connell 2017; Walter et al. 2020).
Despite the multiple primary information about diet in ancient Rome, the direct evidence for its analysis is not enough to clearly identify the food consumption of the common and poor people of Rome.
Several studies have provided isotopic data to reconstruct people’s diet in western communities of the Suburbium (Prowse et al. 2004, 2005, 2008, O'Connell et al. 2019) or in peri-urban Christian catacombs (Rutgers et al. 2009; Salesse et al. 2014; Salesse 2015). Similarly, evidence about the diet of commoners living close to the city walls has started to accumulate (Killgrove and Tykot 2013; Killgrove and Montgomery 2016; Killgrove and Tykot 2018), bridging the gap to a more comprehensive analysis of the diet in Imperial Rome.
The available isotopic evidence agrees that grain was a source of staple foods for Romans, to be mixed with vegetables, meat, and cheese (Prowse et al. 2004, 2005, 2008, Killgrove and Tykot 2013; Killgrove and Montgomery 2016; Killgrove and Tykot 2018). Only a few isotopic glimpses for C4 plant exploitation have been found (Killgrove and Tykot 2013), while domestic animals were the primary faunal resources, as venison consumption was only locally consumed (O'Connell et al. 2019) and fish exploitation could be pointed out for an increased preference among early Christians (Rutgers et al. 2009).
However, the already provided reconstructions miss some of the most significant cemeteries recently discovered in the Rome area, which can provide remarkable data for coping the complex bio-cultural variability of Rome. Indeed, the dietary habits in the whole city should have been heterogeneous, reflecting the multifaceted reality of the capital of one of the most influential Empires in the ancient World. Thus, we would contribute to the spread of investigations into ancient Rome diets by assessing a significant sample of commoners who were buried (and perhaps lived) in the nearby Suburbium is still far from being proficiently accounted for.
Dietary information represents a critical source of knowledge into complex societies such as ancient Rome as it has now been established that customs around food are a key tool for understanding the relationship between humans and their cultural and natural environment in the past (Smith 2006). Therefore, this paper aims to provide a broad diet reconstruction for people buried in Imperial Rome, to combine archaeological and biological evidence from recent excavation results focusing on commoners living in the Imperial Rome.
Materials and methods
Sample
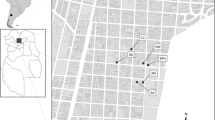
A sample of 214 human bones (Table 1) recovered from 6 funerary contexts (Fig. 1) were selected for carbon and nitrogen stable isotope analysis. The good preservation status of the skeletons according to the lack of soil infiltration in the cancellous bone tissue of the rib bones was the leading inclusion criterion for the recruitment. Information on sex and age at death for each individual were available from previous studies (Catalano 2015), in which the results of osteometric and paleopathological analyses were reported.
The necropolis of Castel Malnome was excavated in the southwestern suburbs (Catalano et al. 2010; Catalano et al. 2013). The sex ratio and juvenile index value, along with osteological suggestions related to musculoskeletal stress markers, push to consider that the funerary area was related to the salt flats unearthed close to the necropolis, where its living community might have worked (Caldarini et al. 2015).
The burial ground of Casal Bertone was set in the eastern suburbs close to the Aurelian walls, in proximity to a large productive area related to an ancient tannery (fullonica) (Musco et al. 2008). The funerary context was archaeologically subdivided into three sections: a mausoleum, a necropolis, and an area, named Area Q, contiguous to the production area. The demographic profile of the mausoleum and necropolis communities allows us to consider them as a unique population (De Angelis et al. 2015), and the analysis of skeletal stress markers suggests the population from both areas could have been engaged in work at the fullonica. Conversely, the demographic profile of Area Q is significantly dissimilar to the others and is characterized by a peculiar distribution of mortality, in which 48% were in the 0–6 years age range. This has been explained by the hazardous environmental conditions in Area Q, evidenced by the presence of pathological alterations likely caused by infectious diseases (De Angelis et al. 2015).
Quarto Cappello del Prete necropolis was established in the extreme eastern suburbs of Rome, along the Via Prenestina, near the ancient city of Gabii. Monumental structures, such as a circular basin and a nymphaeum, were found at the site, and the graves were located along the edges of a pool and in a hypogeum. More than 70% of the buried people were infants and juveniles; 50% of them were in the 0–6 years age range, and more than half of them seem to have suffered from dysmorphic alterations (De Angelis et al. 2015).
The funerary area of Via Padre Semeria is located on the southern side of Rome, along the Via Cristoforo Colombo (Catalano 2015) and close to the Aurelian walls. Land use was related to farming activities, as evidenced by the discovery of the ruins of a “villa rustica” and some hydraulic works (Ramieri 1992), as well as analysis of skeletal stress markers suggesting that females were also involved in agricultural activity (Caldarini et al. 2015).
The baseline for the terrestrial protein component of the diet was set using 17 coeval faunal remains recovered from excavations at Rome (6 from Castel Malnome, 2 from Via Padre Semeria and 9 coming from Colosseum Area), to be used as ecological reference data, supplemented by previously published data for the same geographic and chronological frames. These published data were downloaded from IsoArcH database in several queries performed on or before October 30, 2019 (Salesse et al. 2018; Prowse 2001; O'Connell et al. 2019).
Analytical methods
The extraction of collagen was individually performed at the Centre of Molecular Anthropology for Ancient DNA Studies, Department of Biology, University of Rome Tor Vergata, following Longin’s protocol modified by Brown et al. (1988), which was also simultaneously applied to a modern bovine sample as a reference. In order to obtain a satisfactory yield of collagen, the extraction was performed on about 500 mg of bone powder collected by drilling the bones. The ultrafiltration step was also performed for all the samples in order to magnify the collagen concentration through > 30 kDa Amicon® Ultra-4 Centrifugal Filter Units with Ultracel® membranes.
Each sample of collagen extract weighed 0.8–1.2 mg and was analyzed using an elemental analyzer isotope ratio mass spectrometer at the iCONa (isotope Carbon, Oxygen and Nitrogen analysis) Laboratory of the University of Campania. Carbon (δ13C) and nitrogen (δ15N) stable isotope ratios were measured in a single run on a Delta V Advantage isotope ratio mass spectrometer coupled to a Flash 1112 Elemental Analyser via a Conflow III interface (Thermo Scientific Milan, Italy). Results were expressed in δ notation (Coplen 1995) and reported in permille units. The measurements of δ13C were calibrated to the international standard VPDB with the standard reference materials IAEA-CH3, IAEA-CH6, and stable isotope ratio facility for environmental research at the University of Utah (SIRFER) yeast; δ15N measurements were calibrated to the international standard AIR with the standard reference materials USGS-34, IAEA-N-2, and SIRFER yeast.
To test reliability and exclude contamination from exogenous carbon and nitrogen sources, the samples were compared against established criteria to ascertain the percentages of carbon and nitrogen, atomic C/N ratios, and collagen yields (Ambrose 1990; Ambrose and Norr 1993; De Niro 1985; Van Klinken 1999). Analytical precision was ± 0.3‰ for δ15N, and ± 0.1‰ for δ13C.
Descriptive statistics and comparison tests were performed by R v.3.6.1 (R Core Team 2017).
The suggestions provided by Fraser et al. (2013) and recently further developed by Fontanals-Coll et al. (2016) were employed to detect the consumer’s role for humans compared to the available ecological resources. As described by the authors, this model uses the midpoint and the offsets between consecutive trophic levels to identify the effect of predators on their prey. Thus, the information based on faunal remains was organized according to typology (herbivores, omnivores, marine resources, freshwater organisms), and human data were plotted together in order to detect dietary preferences.
Results
The collagen extraction was performed for the whole sample, but the preservation status of the extracted collagen led us to exclude some individual data: carbon content greater than or equal to 30%, nitrogen content greater than or equal to 10% (Ambrose 1990), and an atomic C/N ratio between 2.9 and 3.6 (De Niro 1985) were the leading determinants for assessing suitable data. CM3 was depleted in elemental compositions, but its C/N ratio and the associated δ13C and δ15 results are consistent with conspecific samples.
The extraction yield was not used as a criterion (Ambrose 1990) because the ultrafiltration technique was used. Only samples with a yield of 0% were ruled out.
Faunal remains yielded enough collagen to be analyzed. Three bones of Canis sp. and two deer samples were recruited in Castel Malnome along with a cattle fragment and two herbivore fragments (sheep and cattle) from the Via Padre Semeria archaeological survey. The Colosseum Area domestics (one bird, one chicken, three pigs, two lambs, one hare, and one cattle) return valid values too (Table 2).
The obtained faunal δ13C values are consistent with a C3 European ecosystem (Schwarcz and Schoeninger 1991), and the δ15N signature suggests the proper trophic level for the identified species.
Out of 214 human samples, only 199 fit the quality criteria. Considering all 199 human individuals, δ13C ranges from – 19.9 to – 14.8‰, whereas δ15N values are between 7.2 and 10.0‰ (Table 3).
The overall data distribution and the density plots indicate a certain heterogeneity: δ13C and δ15N values range between 1.9 and 6.0‰ and between 3.2 and 6.6‰, respectively (Fig. 2).
Indeed, the wider range for δ13C than δ15N could be due to the presence of a few enriched outliers such as CM34 and CM52 (− 14.8‰ and − 17.0‰ for δ13C) as well as CBN1 (− 16.5‰ for δ13C) and CBQ13 (− 17.6‰ for δ13C). Likewise, some lower-δ15N outliers in Casal Bertone necropolis (CBN3, CBN4, and CBN18 with 9.3‰, 8.6‰, and 8.4‰ respectively) and the spanned values for QCP samples account for the wide range detected for δ15N.
The values are consistent with an overall diet mainly based on terrestrial resources. All the human samples rely on a higher trophic level than the primary consumer faunal samples, with no clear indication of exclusive marine food source consumption, although appreciable consumption of these cannot be ruled out, especially for some people at Castel Malnome and Casal Bertone, both at the necropolis and the mausoleum, due to the less negative δ13C data.
The sample stratification according to the necropolis could allow us to evaluate putative differences in food source exploitation. The descriptive statistics for δ13C and δ15N for the six funerary areas were calculated (Table 4).
The osteological evaluation of the human remains allowed us to determine the gender of all individuals, which led us to dissect the variability in food consumption between males and females, as summarized in Table 5.
Discussion
Data integration
Two previously analyzed samples from Casal Bertone necropolis and Casal Bertone mausoleum (Killgrove and Tykot 2013, Supplementary Table 1) were appended to the presented data in order to obtain a whole sample of 231 individual, whose basic descriptive statistics are listed in Table 6.
Furthermore, we are aware that the very restricted sample size for the faunal remains (17 animals) might be only minimally useful for representing the animal baseline, resulting in a bias for the dietary reconstruction of this large urban area. Thus, coeval data was collected by IsoArch Database and from the literature (O'Connell et al. 2019) in order to address this issue. The faunal remains of 48 animals from several species (Supplementary Table 2) made up the whole sample to support this data set as local ecological reference data for Imperial Rome.
A few diachronic samples (mid-fifth to early-sixth centuries CE) were included in the data set to provide additional data for marine species and Leporidae; their isotopic data were obtained by O'Connell et al. (2019) at the nearby Portus site. The data distribution for the faunal remains is consistent with the expected locations in the food net, and very few samples seem to be outliers. A bovine sample from Portus has a higher δ15N value than expected, and the pigs from Ostia (Portus and Isola Sacra) and Colosseum area seem to suggest different foraging strategies due to their different δ15N values. These could represent imported foodstuffs, consistent with the longstanding commercial connections between Rome and the nearby river and maritime ports of Portus and Ostia, and between Rome and other Mediterranean areas through the first centuries CE (O'Connell et al. 2019; Keay 2013). Furthermore, the local baselines for Castel Malnome, Via Padre Semeria, and Colosseum seem to roughly align with the ecological background determined for Portus and Isola Sacra for primary consumer herbivores. Accordingly, omnivores such as dogs from Rome lie one trophic level up and align with other Canidae from Isola Sacra and a bird from Portus. Unfortunately, no freshwater fish remains could be listed in the dataset, while the diachronic marine fish values are accordingly located at less negative δ13C values (Fig. 3).
The humans’ overall high trophic level (compared to the fauna) suggests that the livestock should be considered prey for humans (Fig. 4). This is also confirmed by the strong correlation between δ13C and δ15N (Pearson’s r (243) = 0.75, p < 0.01), excluding the fish data. An explanation for this correlation follows the interpretation of Murray and Schoeninger (1988), which observed a similar trend in their reconstruction of a terrestrial-based diet. Accordingly, our data seem to support the preference for a terrestrial diet based on C3 plant resources and their consumers, rather than a massive consumption of C4 plant and marine fish.
Diet reconstruction
The stable isotope analysis performed on the human remains recovered at the 6 funerary contexts in Imperial Rome (first to third CE) suggests people consumed a roughly heterogeneous diet based on C3 plant backbone resources. As previously reported, several classical authors wrote about agricultural and horticultural practices in the Roman world, confirming the leading role of such a productive activity. Although literary sources on horticulture focused on the cultivation of olives and grapes for their significance in elite production (Lomas 1993), these authors examined the production of cereal grains too, because they made up the bulk of most people’s diets as they were used to make bread and porridge (puls) (Brown 2011).
Our data do not support evidence for exclusive C4 plant exploitation, upholding the notion that animals mainly consumed them in Roman antiquity rather than humans, even though the livestock data reported here does not suggest a foundational role for these plants. Among these, millet represents a generic term for a large group of small-seeded grasses such as both Setaria italica and Panicum miliaceum. Millet is occasionally mentioned in ancient texts, and well-documented archaeological finds lack in archaeological surveys from Imperial Rome or its commercial hub (O'Connell et al. 2019). Despite millet being not Romans’ first choice, it also was not totally discarded by the Romans, though it seems to have been more appreciated far from Rome. The presence of iconographic sources at estates in Pompeii suggests that millet may have been consumed by the wealthy landowners even though they did not totally appreciate it (Jashemski 1992). Indeed, millet was found at several rural Campanian and southern Italy estates (Boscoreale, Herculaneum, and Matrice) (Spurr 1983; Murphy et al. 2013) and its role in cultual practices in northern Italy cannot be ruled out (Rottoli and Castiglioni 2011). Remarkably, millet was often noteworthy in relation to famines and food shortages (Spurr 1983; Garnsey 1999) due to its easy cultivation (Spurr 1983): Columella reports that millet sustained the population of a lot of Italian provinces and the commercial value of millet in the Empire was set in the Edict of Diocletian. Even though millet, which unlike wheat is a non-glutinous grain, can be used for making bread, this seed grass was preferentially used for animal fodder and birdseed rather than direct human consumption (Spurr 1983). Nevertheless, Panicum was recommended for several medical uses, and Pliny counseled that “roasted common millet checks looseness of the bowels and removes gripings” to indicate it particularly for regulating the digestive system (Murphy et al. 2013). Although Killgrove and Tykot (2013) found a consistent use of C4 plants in Castellaccio Europarco for a small sample of buried people, bioarchaeological data about this cereal grain is scarce. Recent archaeobotanical evidence (O'Connell et al. 2019) shows a consistent amount of cereal grains, mainly free-threshing wheats, emmer, einkorn, and barley, at the Roman harbor of Portus, where no C4 plants were recovered. This direct evidence, though biased for chance or for trade in the harbor, is consistent with the aforementioned Roman preference for C3 grains, along with pulses (lentils, peas, and broad beans were recovered) and fruits (a few grapes and elder berries were found in the flotation-sieved contexts at Portus).
The data distribution shows that there is no direct evidence of exclusive marine resource intake too. Although a few individuals, such as CM34, CM52, CBN1, and CBQ13, had less-negative values for δ13C, their moderate δ15N values do not clearly indicate massive marine fish consumption. Their isotopic signatures could be due to a diet consisting of a combination of marine resources and a mix of C3/C4 plant (or primary consumers who eat those plants) related to individual preferences and/or foodstuff availability. Nevertheless, their occasional consumption (along with freshwater resources) cannot be ruled out since up to 20% of the protein consumed could conceivably have come from marine ecosystems without any visible shift in collagen-derived values (Milner et al. 2004; Jim et al. 2006). The seashore vicinity of Castel Malnome hints at the role marine resources could play in the diet, and a local creek could have provided supplemental freshwater food sources. Additionally, people buried at Casal Bertone could have accessed these resources through markets due to their proximity to the city walls.
The lack of evidence for exclusive marine fish (or shellfish) consumption should not be confused with occasional consumption of fish through the Romans’ staple sauce, garum, which could be made with a variety of recipes. Much of the evidence about this ancient fish sauce comes from classical literary sources, which postulate that its popularity derived primarily from social forces influencing individual tastes. The peculiar smell and taste made garum a popular food among wealthy people, although the general populace probably also used it (Grainger 2018).
Recent archaeological findings in Portus turned up faunal remains representing a coeval dietary background (O'Connell et al. 2019). Sheep/pig-sized mammals made up the bulk of the findings, with the latter representing the most common species. We are aware that these findings cannot fully represent the local foodstuffs for the communities buried in the analyzed necropolises, but by the same token, the evidence provided by the harbor of Rome should not be undervalued.
We defined the average values in the dietary proxies for herbivores and omnivores from Rome and Portus/Ostia along with their variances, to draw the boxes where the prey could be set. These boxes are then shifted accounting for the predator-prey offsets, which have been estimated as + 1‰ for δ13C and + 4‰ for δ15N. These dietary markers increase with each trophic level and δ15N rises approximately + 3/+ 5‰, with deviations depending on species and dietary composition, which suggests using the median value (Robbins et al. 2005; Fraser et al. 2013; Fontanals-Coll et al. 2016).
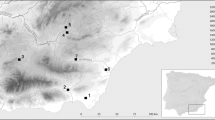
Unfortunately, no data on freshwater resources could be found in coeval and co-regional samples, so it could be tricky to model freshwater fish exploitation. Indeed, there is a paucity of archaeological evidence for the consumption of freshwater resources in the Empire because the archeozoological record rarely includes lacustrine or riverine faunal remains and, when it does include them, they are in minimal numbers and are difficult to obtain for analysis. Comparative data about this kind of prey have been collected for two diachronic samples from pre-Roman Britain (Jay 2008) and the late-Roman province of Pannonia (Hakenbeck et al. 2017) (Fig. 5). These datasets provide useful isotopic data concerning some freshwater resources, even though we are aware that the ecological background could result in biased values in freshwater fish isotopic signatures (Dufour et al. 1999). However, their isotopic signature cannot be used as specific end-members, but they could be leveraged for supporting the identification of putative freshwater resources consumption as representing the best approximation for such missing local data.
Linear model for the identification of prey-predator relationship. th, terrestrial herbivore; thc, box for terrestrial herbivore consumers; to, terrestrial omnivore; toc, box for terrestrial omnivore consumers; fwE, freshwater fish from England; fwEc, box for freshwater fish from England consumers; fwP, freshwater fish from Pannonia; fwPc, box for freshwater fish from Pannonia consumers; mf, marine fish; mfc, box for marine fish consumers. The dashed lines define consumers’ boxes. The cemeteries are identified as referred in Table 1
Despite the significant differences between the two samples (δ13C T-value 3.10, p < 0.01; δ15N T-value 4.27, p < 0.01), they consistently have low δ13C and high δ15N values, as those expected for these resources.
The estimation of the consumers’ boxes indicates that most individuals fall inside the boxes built for herbivore and omnivore consumers (Supplementary Fig. 1) even though 48 individuals fall beyond the threshold for a clear C3-derived omnivore consumer (Table 7). This evidence pushes us to reconsider the fraction of people whose diet was based on mixed C3/C4 plants and/or marine resources. The data stratification for those 48 individuals by site, sex, or age classes does not support any specific trend except for adult/child comparison for δ15N (δ13C: Kruskal-Wallis chi-squared = 8.44, df = 5, p value = 0.13 for site; Kruskal-Wallis chi-squared = 0.03, df = 2, p value = 0.98 for sex; Kruskal-Wallis chi-squared = 0.11, df = 1, p value = 0.73 for age class; δ15N: Kruskal-Wallis chi-squared = 9.62, df = 5, p value = 0.09 for site; Kruskal-Wallis chi-squared = 2.55, df = 2, p value = 0.28 for sex; Kruskal-Wallis chi-squared = 4.40, df = 1, p value = 0.04 for age class) suggesting a subtle stratification between age classes in that sub-sample. People from Casal Bertone (both necropolis and mausoleum) seem to be overrepresented (33 out 48 people). The moderate δ15N values offset between humans and marine fish (mean values 11.5‰ for adults and 11.0‰ for children vs 10.0‰ for marine resources herein considered) might deter to consider this shift due to marine resources exploitation exclusively. This could be supported by the notion that marine fish was considered expensive food in the Empire, suggesting that regular fish consumption may have been restricted to the upper strata of Roman society (Frayn 1993). However, the presence of several people buried in Casal Bertone (Musco et al. 2008) could advise to consider that they were a fairly wealthy group whose diet was varied and heterogeneous. This evidence is further supported by the topographical location of the cemetery, close to the city center. Hence, people buried in this area (and perhaps living and working at the same location, Catalano 2015) could easily access to market system featuring the city of Rome, where several horrea (large warehouses and other storage facilities in Ancient Rome) were located (Vera 2008; Burgers et al. 2015).
Via Padre Semeria necropolis was established in the hydrographic net of Almone river (Tallini et al. 2019), and its data distribution support a more than sporadic consumption of these foodstuffs (Supplementary Fig. 2), which could have represented a supplement food for this farming-based community (De Angelis et al. 2015).
The median value determined for QCP is apart from other cemeteries (Supplementary Fig. 2), suggesting a mostly farming-derived diet. This necropolis is related to a cultic site (Musco et al. 2001; Catalano 2015) where people sharing some biological characteristics related to osteo-dysmorphias seems to be collected, as suggested by osteological analysis (De Angelis et al. 2015). However, as 12 individuals from Quarto Cappello del Prete died before they were 3 years old, we assume that they were not wholly weaned, according to historical and bioarcheological data for ancient Rome (Dupras et al. 2001; Fulminante 2015). We are aware that their isotopic signature could be impacted by the breastfeeding effect (Fogel et al. 1989; Fuller et al. 2006; Beaumont et al. 2015). Accordingly, we removed their isotopic values for the further speculations about diet reconstruction as their δ15N signatures are different from those obtained from the adults in Quarto Cappello del Prete (T = 3.23; p < 0.01).
The median values calculated for Castel Malnome and Casal Bertone Area Q are quite similar (Table 4) and suggest a diet high in protein and mainly relied on C3 plants and C3-consumer species. This appears noteworthy considering that these necropolises were tied to manufacturing activities (saltworks in Castel Malnome and a tannery in Casal Bertone Area Q), where people should collectively be strong enough for their stressful tasks (De Angelis et al. 2015). The necropolis and the mausoleum of Casal Bertone appear to be shifted respect to Casal Bertone Area Q, suggesting a certain degree of heterogeneity in their diets. This evidence does not fit the archaeological data (Musco et al. 2008; Killgrove and Tykot 2013), which showed a clear difference in the social stratification between necropolis and mausoleum samples: the isotopic results flatten the social mismatch, at least for the dietary landscape.
Comparisons
To fully explore the dietary scenario, we have attempted to understand the roles of several factors that could be significant in the onset of the differences among the necropolises.
All the data are normally distributed (Supplementary Table 3) except for Castel Malnome (both δ13C and δ15N values) and Casal Bertone mausoleum δ15N values (Supplementary Figs. 3, 4, and 5).
The data distribution for Castel Malnome suggests the presence of diet-based groups, with some outliers. CM16 (male, 20–29 years old), CM20 (13–19 years, not available sex), CM21 (young female), CM23 (female, 30–39 years old), and CM33 (male, 40–49 years) feature significant low δ13C and δ15N values and those samples, along with CM66 (male, 30–39 years old), CM69 (male, 40–49 years old), and CM40 (male, 40–49 years old) seem to be clustered outside the normal distribution for δ13C, suggesting a diet mainly underpinned by plant-derived carbohydrate. Conversely, CM34 (male, 20–29 years old), CM52 (male, 50 or more years old), and CM55 (male, 30–39 years old) could have exploited marine fish and C4 plants, while CM29 (male, 20–29 years old), and CM47 (male, 20–29 years old) are outliers for δ15N, suggesting a protein-rich diet. The diet heterogeneity for Castel Malnome suggests that people buried in that site could have access to various dietary resources that would enable them to satisfy the nutritional requirements in carbohydrates and proteins for facing their everyday tasks related to the saltworks. The intra-community differences show that some people could have exploited more protein-rich stuff, paving the way to consider the hypothesis for the establishment of socially-heterogeneous strata in that working community, that cannot be detected by the osteological and archaeological records.
The δ15N values in Casal Bertone mausoleum highlight a sub-stratification too. CBM20 (child, 7–12 years old), CBM23 (child, 7–12 years old), CBM25 (teen, 13–19 years old), F10C (child, from Killgrove 2010), F04B, and F11A (two adult females, from Killgrove 2010) do not fit the normal distribution for low values, whereas CBM1 (male, 30–39 years old), CBM2 (female, 20–29 years old), and CBM24 (male, 40–49 years old) are at the upper limit of the data distribution, falling outside the normal distribution. These differences support previous archaeological analysis prompting the hypothesis that the mausoleum at Casal Bertone could host the tannery leadership and extended family, including slaves and freedmen (Musco et al. 2008; Catalano 2015), which could benefit from different dietary habits.
The sample stratification among all the necropolis was evaluated through the analysis of variance (ANOVA) to detect differences among the groups (Levene’s test for the homogeneity of the variances δ13C test statistic 1.45, p = 0.21; δ15N test statistic 1.48, p = 0.20) taking into account site, biological sex (male, female, or unknown due to skeletal immaturity), and age at death, with a dichotomic classification between adults and non-adults due to the variety of age classes reported, which was a result of different scoring methods used among the samples.
The site did not represent a significant determinant for the onset of δ13C differences among the samples (F: 1.18; p = 0.32), as well as sex and age featured similar δ13C values (sex F: 1.00, p = 0.32; age class F: 0.37, p = 0.54). However, the cemeteries are different for what concerns the δ15N (site F: 8.00, p < 0.01), while demographic differences are negligible (sex F: 01.75, p = 0.19; age F: 0.01, p = 0.91). Thus, we can conclude that the people buried in the analyzed necropolis were characterized by different diets regarding the introduction of proteins, but they were not sex-biased. Furthermore, the children more than 3 years old were fed like adults. This is in line with the previous findings for the greater Rome samples. Only Isola Sacra necropolis showed a sex-based differences in diet, even though people buried in that cemetery were considered a biased sample of Roman commoners, with better than average diets (Prowse et al. 2004, 2005). Despite the limited sample size, other samples from Imperial Rome did not point out differences in diet between males and females (Killgrove and Tykot 2013; Killgrove and Tykot 2018), and this is true also for the inhabitants of Velia, an Imperial port in southern Italy (Craig et al. 2009). The small amount of isotopic data pertaining the weaning practices in ancient Rome and the neighboring eastern suburbs are consistent with the identified weaning age-threshold (Rutgers et al. 2009; Killgrove and Tykot 2013; Killgrove and Tykot 2018), that is slightly beyond the suggestion provided for western communities (Prowse et al. 2004, 2005, 2008).
The Tukey HSD test (Maxwell and Delaney 2003; Dubitzky et al. 2013) was performed to determine which cemetery pairs underpin the differences in δ15N, accounting for multiple comparisons and maintaining experiment-wise alpha at 0.05 (Yuan and Maxwell 2005). The significant differences were found between Quarto Cappello del Prete and all the other cemeteries (Table 8). This result supports the peculiar status of the cultual site of Quarto Cappello del Prete respect to the other cemeteries in Imperial Rome that were instead related to low-social strata and working communities. Remarkably, Quarto Cappello del Prete isotopic values are not consistent with those obtained for the roughly coeval Gabines (Killgrove and Tykot 2018), the people inhabiting the ancient city of Gabii, a formerly independent city tackling a population contraction in Imperial age, just a few kilometers far from Quarto Cappello del Prete. The differences (δ13C F: 5.08, p = 0.03; δ15N F: 13.81, p < 0.01 that shift to δ13C F:4.05, p = 0.05; δ15N F: 14.63, p < 0.01 by excluding one outlier and three putatively breastfed children, according to Killgrove and Tykot 2018) seem to confirm that Quarto Cappello del Prete related to a site where shortlisted individuals rather than a living community were buried (De Angelis et al. 2015).
Comparisons with available data for Roman area
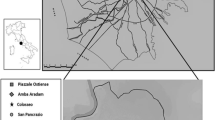
In an attempt to describe the food preferences of people buried in greater Rome area, we collected the data for other funerary contexts such as Castellaccio Europarco (Killgrove and Tykot 2013) and ANAS (Prowse 2004) via IsoArch, and the data for people buried in nearby Isola Sacra (Prowse 2001; 2004, Crowe et al. 2010) and Portus Tenuta del Duca, (O'Connell et al. 2019) are included for comparison.
The data evaluation suggested there were dissimilarities in both δ13C and δ15N distributions’ variances (δ 13C: F value = 8.45, p < 0.01; δ 15N: F value = 10.02, p < 0.01) (Supplementary Table 4).
The joint evaluation of the differences adjusted for multiple comparison allows us to determine common patterns among necropolises, which could be grouped both on δ 13C and δ 15N axes according to the significant differences (the mean values of these can be seen in Figs. 6 and 7, in the upper-right plots).
We can identify two segregated groups on the δ13C axis. One group (group A) comprises Via Padre Semeria, Casal Bertone Area Q, Castel Malnome, Quarto Cappello del Prete, and ANAS, which are different from a second cluster (group B) including Isola Sacra, Casal Bertone Necropolis, and Castellaccio Europarco. Tenuta del Duca (Portus) in the middle, sharing features from both group A and group B. This is only partially surprising if we consider the location of this latter site at the Rome harbor, where people from other areas and who could have eaten different diets might have been buried. The groups defined on the δ13C axis suggest that the individuals within them had slightly overall different dietary habits. Group A sites share a diet massively founded on C3 plants, that are consistent with the supposed diet reconstructions for Via Padre Semeria, Castel Malnome, and Casal Bertone Area Q and Quarto Cappello del Prete. Freshwater fauna could have represented a more than sporadic protein supplement for some of these communities that could have grasped these resources because of streaming creeks proximity (Castel Malnome and Via Padre Semeria were close to the Magliana and Galeria creeks, and in the Almone hydrogeological net, respectively). Casal Bertone Area Q was also set in a humid environment as it was established close to the ancient tannery, a factory which needed a massive amount of water, that was putatively provided by several streamlets involved in the Aniene aquifers, also by a subsidiary branch of the Aqua Virgo aqueduct (Musco et al. 2008). Indeed, the easy access to water was one of the leading aspects of considering the establishment of such a productive plant, so it is not surprising that people living in proximity could have exploited freshwater resources. Group B represents people with a more heterogeneous diet, where C4 plant and marine resources consumption cannot be ruled out, even though their exclusive exploitation should be denied, as previously determined for Isola Sacra and Castellaccio (Prowse et al. 2004, 2005; Killgrove and Tykot 2013). The mausoleum and the necropolis of Casal Bertone fall in this cluster, suggesting a more heterogeneous dietary supply than the other Roman areas.
Furthermore, δ15N values could be useful for determining two additional clusters. The first (group C) groups together ANAS, Castellaccio Europarco, and Quarto Cappello del Prete, while the second (group D) comprises Via Padre Semeria, Castel Malnome, Casal Bertone Area Q, and the samples from Portus and Isola Sacra.
The clusters on the δ15N axis should display meaningful differences in animal-derived protidic intake. Group C seems to feature lower animal protein intake than group D, where terrestrial fauna and lacustrine and riverine organisms likely provided needed dietary protein.
Again, Casal Bertone samples from the necropolis and mausoleum seem to feature intermediate characteristics. The archaeological differences between them (Musco et al. 2008) are not sustained by anthropological and isotopic evidence. Indeed, De Angelis et al. (2015) already recommended considering these contiguous areas pertaining to a single population related to the fullonica that should be decoupled from area Q, where the demographic profile (and dietary habits) suggests a separate community. Even though no certain archaeological data supports this hypothesis, it would be unusual for the funerary buildings of Area Q to be close to a productive area such as the tannery. In our opinion, we should envisage the partial diachronic establishment of the tannery and the funerary buildings comprising Area Q.
The topographical location in the Suburbium (eastern suburbs, eastern suburbs close to city walls, southwestern suburbs, southwestern area close to Aurelian walls, Portus and Ostia) seems to indicate some differences (δ13C Kruskal-Wallis chi square = 76.27, p < 0.01; δ15N Kruskal-Wallis chi square = 34.92, p < 0.01) even though it is hard to identify a cline as well as some other common patterns. Nevertheless, the heterogeneous landscape highlighted in the Ostia and Portus samples is significantly different from eastern (Quarto Cappello del Prete and, close to the city walls, the whole sample of Casal Bertone) and southwestern (Castel Malnome, Castellaccio Europarco, ANAS, and, close to the city walls, Via Padre Semeria) necropolises (Supplementary Tables 5 and 6).
Conclusions
The paper outlines the dietary landscape of Rome in the Imperial period. The evidence presented here provides a unique glimpse into the lives of the people who lived and died in Rome, whose bio-cultural profiles have been previously described through detailed osteological, anthropological, and archaeological evaluations.
The necropolises located outside the city walls are often related to individual low social classes, and people were tied to productive sites or rustic villae.
The dietary landscape we provide is heterogeneous and reflects the multifaceted reality of the capital of one of the most influential empires in Antiquity.
The complexity of Roman society remains hard to disentangle even from a dietary point of view, but some elements can be illuminated. One of these is the pivotal role of C3 plants, which is confirmed as the staple foodstuff of the lower class. However, C4 plants also seem to have been consumed, albeit they were not widespread. The environment played a critical role for Roman commoners. Even though administrative grain supplements partially sustained them, the topographical location of the settlements (and perhaps of the necropolises where people were buried) determined the preferential consumption of food that people could easily obtain from their neighborhood. People could gain the protidic intake needed to sustain active lifestyles by farming and/or livestock breeding as well as by gathering (fishing and/or hunting). Nevertheless, the complexity of Roman society and trade that passed through Rome during the Imperial period accounted for the broader range of foodstuffs that people could access, making a portrait of the nutritional habits of Romans challenging. However, the meticulous selection of burial grounds in this paper could lead to a less biased reconstruction. Indeed, exotic foods were only partially accessible to commoners, who mainly relied on local food resources, even though markets were accessible, especially to people living close to the city center. The proposed approach represents a powerful tool able to shed light on a crucial aspect of the biological characteristics of this ancient human population but pushing beyond the biological feature. Dietary patterns should be understood as one of the most long-lasting markers of the cultural identity of a population, and the information provided herein represents a step forward in the understanding of the social organization of this ancient society, to be complemented by genomic and isotopic data related to migration, both in synchronic and diachronic perspectives (Killgrove and Montgomery 2016; Antonio et al. 2019; De Angelis et al. in prep). Accordingly, the steady deepening of a combined archaeological and anthropological evaluation will allow us to stratify the Roman sample concerning the bio-cultural factors that impacted the lives of a significant sample of the Roman population.
References
Ambrose SH (1990) Preparation and characterization of bone and tooth collagen for isotopic analysis. J Archaeol Sci 17:431–451
Ambrose SH, Norr L (1993) Experimental evidence for the relationship of the carbon isotope ratios of whole diet and dietary protein to those of bone collagen and carbonate. In Lambert JB & Grupe G (Eds.) Prehistoric human bone: archaeology at the molecular level. Springer-Verlag, New York, pp1-37
Antonio ML, Gao Z, Moots HM, Lucci M, Candilio F, Sawyer S, Oberreiter V, Calderon D, Devitofranceschi K, Aikens RC, Aneli S, Bartoli F, Bedini A, Cheronet O, Cotter DJ, Fernandes DM, Gasperetti G, Grifoni R, Guidi A, la Pastina F, Loreti E, Manacorda D, Matullo G, Morretta S, Nava A, Fiocchi Nicolai V, Nomi F, Pavolini C, Pentiricci M, Pergola P, Piranomonte M, Schmidt R, Spinola G, Sperduti A, Rubini M, Bondioli L, Coppa A, Pinhasi R, Pritchard JK (2019) Ancient Rome: a genetic crossroads of Europe and the Mediterranean. Science 366:708–714
Beaumont J, Montgomery J, Buckberry J, Jay M (2015) Infant mortality and isotopic complexity: New approaches to stress, maternal health, and weaning. American Journal of Physical Anthropology 157:441–457
Boatwright MT, Gargola DJ, Lenski N, Talbert RJA (2011) From village to empire: a history of Rome from earliest times to the end of the Western Empire. Oxford University Press, Oxford
Bocherens H, Fizet M, Mariotti A (1994) Diet, physiology and ecology of fossil mammals as inferred from stable carbon and nitrogen isotope biogeochemistry: implications for Pleistocene bears. Palaeogeogr Palaeoclimatol Palaeoecol 107:213–225. https://doi.org/10.1016/0031-0182(94)90095-7
Brothwell D, Brothwell P (1998) Food in antiquity: a survey of the diet of early peoples. Johns Hopkins University Press, Baltimore
Brown M (2011) Grain, pulses and olives: an attempt toward a quantitative approach to diet in Ancient Rome. J Wash Acad Sci 97:1–24
Brown TA, Nelson DE, Vogel JS, Southon JR (1988) Improved collagen extraction by modified Longin method. Radiocarbon 30:71–77
Burgers GLM, Kok RA, Sebastiani R (2015) The Imperial Horrea of the Porticus Aemilia. Babesch Bull Antieke Beschaving 90:199–212
Caldarini C, Zavaroni F, Benassi V (2015) Indicatori scheletrici di lavoro: marcatori muscoloscheletrici, artropatie e traumi. Med Secoli 27:893–956
Catalano P (2015) Gli scheletri degli antichi romani raccontano. Indagini antropologiche su 11 sepolcreti di età imperiale del suburbio romano. Med Secoli 27:773–786
Catalano P, Benassi V, Caldarini C, Cianfriglia L, Mosticone R, Nava A, Pantano W, Porreca F (2010) Health status and lifestyle in Castel Malnome (Rome, I-II cent. A. D.). Med Secoli 22:111–128
Catalano P, Caldarini C, Mosticone R et al (2013) Il contributo dell’analisi traumatologica nella ricostruzione dello stile di vita della comunità di Castel Malnome (Roma, I- II sec. dC). Med Secoli 25:101–118
Champlin E (1982) The suburbium of Rome. Am J Ancient Hist 7:97–117
Cherel Y, Hobson KA, Bailleul F, Groscolas R (2005) Nutrition, physiology, and stable isotopes: new information from fasting and molting penguins. Ecology 86:2881–2888
Cool HEM (2006) Eating and drinking in Roman Britain. Cambridge University Press, Cambridge
Coplen TB (1995) Reporting of stable hydrogen, carbon, and oxygen isotopic abundances. Geothermics 24:707–712
Craig OE, Biazzo M, O’Connell TC et al (2009) Stable isotopic evidence for diet at the Imperial Roman coastal site of Velia (1st and 2nd Centuries AD) in Southern Italy. Am J Phys Anthropol 139:572–583
Crowe F, Sperduti A, O'Connell TC, Craig OE, Kirsanow K, Germoni P, Macchiarelli R, Garnsey P, Bondioli L (2010) Water-related occupations and diet in two Roman coastal communities (Italy, first to third century AD): correlation between stable carbon and nitrogen isotope values and auricular exostosis prevalence. Am J Phys Anthropol 142:355–366
De Angelis F, Pantano W, Battistini A (2015) L’analisi paleodemografica. Med Secoli 27:805–872
De Ligt L (2006) The economy: agrarian change during the second century. In Rosenstein N, Morstein-Marx R (Eds.) A companion to the Roman Republic. Oxford, pp 590–605.
De Niro MJ (1985) Postmortem preservation and alteration of in vivo bone collagen isotope ratios in relation to palaeodietary reconstruction. Nature 317:806–809
Delgado AM, Vaz Almeida MD, Parisi S (2017) Chemistry of the Mediterranean diet. Springer International Publishing, Switzerland
Dubitzky W, Wolkenhauer O, Cho KH, Yokota H (2013) Tukey’s HSD Test. In: Encyclopedia of Systems Biology. Springer, New York
Dufour E, Bocherens H, Mariotti A (1999) Palaeodietary implications of isotopic variability in Eurasian Lacustrine Fish. J Archaeol Sci 26:617–627
Dupras TL, Schwarcz HP, Fairgrieve SI (2001) Infant feeding and weaning practices in Roman Egypt. Am J Phys Anthropol 115:204–212
Dyson SL (2010) Rome—a living portrait of an ancient city a living portrait of an ancient city. Johns Hopkins University Press, Baltimore
Fahy GE, Deter C, Pitfield R, Miszkiewicz JJ, Mahoney P (2017) Bone deep: variation in stable isotope ratios and histomorphometric measurements of bone remodelling within adult humans. J Archaeol Sci 87:10–16
Fogel ML, Tuross N, Owsley DW (1989) Nitrogen isotope tracers of human lactation in modern and archaeological populations. Carnegie Institution of Washington Yearbook 88:111–117
Fontanals-Coll M, Eulàlia Subirà M, Díaz-Zorita Bonilla M, Gibaja JF (2016) First insight into the Neolithic subsistence economy in the north-east Iberian Peninsula: paleodietary reconstruction through stable isotopes. Am J Phys Anthropol 62:36–50
Fraser RA, Bogaard A, Schäfer M, Arbogast R, Heaton THE (2013) Integrating botanical faunal and human stable carbon and nitrogen isotope values to reconstruct land use and palaeodiet at LBK Vaihingen an der Enz Baden-Wurttemberg. World Archaeol 45:492–517
Frayn JM (1993) Markets and fairs in Roman Italy: their social and economic importance from the Second Century BC to the Third Century AD. Clarendon Press, Oxford
Fulminante F (2015) Infant feeding practices in Europe and the Mediterranean from Prehistory to the Middle Ages: a comparison between the historical sources and bioarchaeology. Child Past 8:24–47
Fuller BT, Fuller JL, Harris DA, Hedges REM (2006) Detection of breastfeeding and weaning in modern human infants with carbon and nitrogen stable isotope ratios. American Journal of Physical Anthropology 129:279–293
Garnsey P (1999) Food and society in classical antiquity. University Press, Cambridge
Grainger S (2018) Garum and Liquamen, What’s in a name? J Marit Archaeol 13:247–261
Hakenbeck SE, Evans J, Chapman H, Fóthi E (2017) Practising pastoralism in an agricultural environment: an isotopic analysis of the impact of the Hunnic incursions on Pannonian populations. PLoS One 12:e0173079
Jashemski WF (1992) The gardens of Pompeii, Herculaneum and the villas destroyed by Vesuvius. J Garden Hist 12:102–125
Jay M (2008) Iron Age diet at Glastonbury lake village: the isotopic evidence for negligible aquatic resource consumption. Oxf J Archaeol 27:201–216
Jim S, Jones V, Ambrose SH, Evershed RP (2006) Quantifying dietary macronutrient sources of carbon for bone collagen biosynthesis using natural abundance stable carbon isotope analysis. Br J Nutr 95:1055–1062
Johnson PS (2013) Economic evidence and the changing nature of urban space in late antique Rome. University Press, Barcelona
Keay S (2013) Rome, Portus and the Mediterranean. The British School at Rome, London
Killgrove K (2010) Identifying immigrants to Imperial Rome using strontium isotope analysis. J Roman Archaeol Suppl 78:157–174
Killgrove K, Montgomery J (2016) All roads lead to Rome: exploring human migration to the eternal city through biochemistry of skeletons from two Imperial-Era cemeteries (1st-3rd c AD). PLoS One 11:e0147585
Killgrove K, Tykot R (2013) Food for Rome: a stable isotope investigation of diet in the Imperial period (1st-3rd centuries AD). J Anthropol Archaeol 32:28–38
Killgrove K, Tykot R (2018) Diet and collapse: a stable isotope study of Imperial-era Gabii (1st–3rd centuries AD). J Archeol Sci Rep 19:1041–1049
King A (1999) Diet in the Roman world: a regional inter-site comparison of the mammal bones. J Roman Archaeol 12:168–202
Kron G (2002) Comparative perspectives on nutrition and social inequality in the Roman World. In: Erdkamp P, Holleran C (eds) Diet and nutrition in the Roman World. Routledge, London, pp 156–174
Lo Cascio E (1994) The size of the Roman population: Beloch and the meaning of the Augustan Census Figures1. J Roman Stud 84:23–40. https://doi.org/10.2307/300868
Lomas K (1993) Rome and the Western Greeks, 350 BC - AD 200: conquest and acculturation in Southern Italy. Routledge, London
MacKinnon M (2004) Production and consumption of animals in Roman Italy: integrating the zooarchaeological and textual evidence. Journal of Roman Archaeology, Rhode Island
Marzano A (2018) Fish and fishing in the Roman World. J Marit Archaeol 13:437–447. https://doi.org/10.1007/s11457-018-9195-1
Maxwell SE, Delaney HD (2003) Designing experiments and analyzing data: a model comparison perspective, 2nd edn. Routledge, London
Mekota A-M, Grupe G, Ufer S, Cuntz U (2006) Serial analysis of stable nitrogen and carbon isotopes in hair: monitoring starvation and recovery phases of patients suffering from anorexia nervosa. Rapid Commun Mass Spectrom 20:1604–1610
Milner N, Craig OE, Bailey GN, Pedersen K, Andersen SH (2004) Something fishy in the Neolithic? A re-evaluation of stable isotope analysis of Mesolithic and Neolithic coastal populations. Antiquity 78:9–22
Morley N (1996) Metropolis and Hinterland. The city of Rome and the Italian economy 200 B.C.–A.D. 200. University Press, Cambridge.
Murphy C, Thompson G, Fuller DQ (2013) Roman food refuse: urban archaeobotany in Pompeii, Regio VI, Insula 1. Veget Hist Archaeobot 22:409–419
Murray ML, Schoeninger MJ (1988) Diet, status, and complex social structure in Iron Age Central Europe: some contributions of the bone chemistry. In: Gibson DB, Geselowitz MN (eds) Tribe and Polity in Late Prehistoric Europe. Plenum Press, New York, pp 155–176
Musco S, Petrassi L, Pracchia S (2001) Luoghi e paesaggi archeologici del suburbio orientale di Roma. Pegaso: Rome
Musco S, Catalano P, Caspio A et al (2008) Le complèxe archéologique de Casal Bertone. Les Dorriers d’Archèologie 330:32–39
O’Connell TC (2017) ‘Trophic’ and ‘source’ amino acids in trophic estimation: a likely metabolic explanation. Oecologia 184:317–326
O'Brien DM (2015) Stable isotope ratios as biomarkers of diet for health research. Annu Rev Nutr 35:565–594
O'Connell TC, Ballantyne RM, Sheila Hamilton-Dyer S et al (2019) Living and dying at the Portus Romae. Antiquity 93:719–734
Pecquerie L, Nisbet RM, Fablet R, Lorrain A, Kooijman SALM (2010) The impact of metabolism on stable isotope dynamics: a theoretical framework. Philos Trans R Soc Lond Ser B Biol Sci 365:3455–3468
Prowse TL (2001) Isotopic and dental evidence for diet from the necropolis of Isola Sacra (1st-3rd centuries AD), Italy (PhD dissertation). McMaster University, Hamilton
Prowse T, Schwarcz HP, Saunders S et al (2004) Isotopic paleodiet studies of skeletons from the Imperial Roman-age cemetery of Isola Sacra, Rome, Italy. J Archaeol Sci 31:259–272
Prowse T, Schwarcz HP, Saunders S et al (2005) Isotopic evidence for age-related variation in diet from Isola Sacra, Italy. Am J Phys Anthropol 128:2–13
Prowse TL, Saunders SR, Schwarcz HP, Garnsey P, Macchiarelli R, Bondioli L (2008) Isotopic and dental evidence for infant and young child feeding practices in an imperial Roman skeletal sample. Am J Phys Anthropol 137:294–308
Purcell N (2003) The way we used to eat: diet, community, and history at Rome. Am J Philol 124:329–358
R Core Team (2017) R: a language and environment for statistical computing https://www.R-project.org
Ramieri AM (1992) La cisterna Romana in via Cristoforo Colombo. SIP, Rome
Reitsema LJ (2013) Beyond diet reconstruction: stable isotope applications to human physiology, health, and nutrition. Am J Hum Biol 25:445–456
Robbins CT, Felicetti LA, Sponheimer M (2005) The effect of dietary protein quality on nitrogen isotope discrimination in mammals and birds. Oecologia 144:534–540
Rottoli M, Castiglioni E (2011) Plant offerings from Roman cremations in northern Italy: a review. Veg Hist Archaeobotany 20:495–506
Rowan E (2017) Bioarchaeological preservation and non-elite diet in the Bay of Naples: an analysis of the food remains from the Cardo V sewer at the Roman site of Herculaneum. Environ Archaeol 22:318–336
Rutgers LV, van Strydonck M, Boudin M (2009) Stable isotope data from the early Christian catacombs of ancient Rome: new insights into the dietary habits of Rome’s early Christians. J Archaeol Sci 36:1127–1134
Salesse K (2015) Archéo-biogéochimie isotopique, reconstitutions des régimes alimentaires et des schémas de mobilité, et interactions bioculturelles. Les sépultures plurielles de la catacombe des Saints Pierre-et-Marcellin (Rome, Ier-IIIe s. ap. J.-C.): Les sépultures plurielles de la région X de la catacombe des Saints Pierre-et-Marcellin (Rome, Ier-IIIe s. ap. J.-C.). 353
Salesse K, Dufour E, Lebon M et al (2014) Variability of bone preservation in a confined environment: the case of the catacomb of Sts Peter and Marcellinus (Rome, Italy). Palaeogeogr Palaeoclimatol Palaeoecol 416:43–54. https://doi.org/10.1016/j.palaeo.2014.07.021
Salesse K, Fernandes R, de Rochefort X et al (2018) IsoArcH.eu: an open-access and collaborative isotope database for bioarcheological samples from Graeco-Roman World and its margins. J Archaeol Sci Rep 19:1050–1055
Scheidel W (2007) Roman population size: the logic of the debate. Princeton/Stanford Working Papers in Classics Paper No. 070706. Princeton/Stanford University: https://ssrn.com/abstract=1096415 or https://doi.org/10.2139/ssrn.1096415
Scheidel W (2009) Disease and death in the ancient city of Rome. Princeton/Stanford Working Papers in Classics Paper No. 070706. Princeton/Stanford University: https://ssrn.com/abstract=1347510 or https://doi.org/10.2139/ssrn.1347510
Schwarcz HP, Schoeninger MJ (1991) Stable isotope analyses in human nutritional ecology. Yearb Phys Anthropol 34:283–321
Smith ML (2006) The archaeology of food preference. Am Anthropol 108:480–493
Spurr MS (1983) The cultivation of millet in Roman Italy. Papers of the British School at Rome 51: 1–15.
Tallini M, Di Leo A, Rossetti C, Berardi F (2019) The Sacred Almone River of the Appian Way Regional Park in Rome: ancient myths, a ritual link between Rome and Asia Minor and water uses in the modern age. Water Supply 13:727–734
Tsutaya T, Yoneda M (2015) Reconstruction of breastfeeding and weaning practices using stable isotope and trace element analyses: a review. Am J Phys Anthropol 156:2–21
Tykot RH (2014) Bone chemistry and ancient diet. In: Smith C (ed) Encyclopedia of Global Archaeology. Springer, New York
Van Klinken GJ (1999) Bone collagen quality indicators for palaeodietary and radiocarbon measurements. J Archaeol Sci 26:687–695
Vera D (2008) Gli horrea frumentari dell’Italia tardoantica: tipi, funzioni, personale. Mélanges de l'école française de Rome 120:323–336
Walter BS, DeWitte SN, Dupras T, Beaumont J (2020) Assessment of nutritional stress in famine burials using stable isotope analysis. Am J Phys Anthropol 172:214–226
Waters-Rist AL, Katzenberg MA (2010) The effect of growth on stable nitrogen isotope ratios in subadult bone collagen. Int J Osteoarchaeol 20:172–191
Wilkins JM, Hill S (2006) Food in the ancient world. Blackwell Publishing Ltd, Oxford
Yardley JC (1991) The symposium in Roman elegy. In: Slater WJ (ed) Dining in a classical context. University of Michigan, Ann Arbor, pp 149–155
Yuan KH, Maxwell S (2005) On the post hoc power in testing mean differences. J Educ Behav Stat 30:141–167
Acknowledgments
The authors would like to acknowledge Andy Bolduc and Martin Bennet for providing language help.
Funding
Open access funding provided by Università degli Studi di Roma Tor Vergata within the CRUI-CARE Agreement. This work was supported by the Italian Ministry of Education, Universities and Research (MIUR) through PRIN 2015 (Diseases, health and lifestyles in Rome: from the Empire to the Early Middle Age, Grant ID: 2015PJ7H3K) allotted to CML, RSV, and VG.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 485 kb)
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
De Angelis, F., Varano, S., Battistini, A. et al. Food at the heart of the Empire: dietary reconstruction for Imperial Rome inhabitants. Archaeol Anthropol Sci 12, 244 (2020). https://doi.org/10.1007/s12520-020-01194-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12520-020-01194-z