Abstract
Background
The prevalence of congenital diaphragmatic hernia (CDH) varies across countries, with limited information available on its epidemiology in China. Our study aimed to investigate the prevalence, time trends, and perinatal outcomes of CDH in China, as well as its associated malformations and potential associations with maternal and infant characteristics.
Methods
This study included all birth and CDH cases from the Chinese Birth Defects Monitoring Network between 2007 and 2019, with CDH cases classified as either isolated or associated. We employed the joinpoint regression model to calculate the trends of prevalence and the annual percent change, with Poisson regression used for adjusted prevalence rate ratios. A P value ≤ 0.05 was considered statistically significant.
Results
A total of 4397 CDH cases were identified among 24,158,029 births in the study period, yielding prevalence rates of 1.82, 1.13 and 0.69 per 10,000 for overall, isolated, and associated CDH, respectively. The prevalence of each type of CDH increased over time. The prevalence of overall CDH varied significantly by infant sex (male vs. female, 1.91/10,000 vs. 1.63/10,000), maternal residence (urban vs. rural, 2.13/10,000 vs. 1.45/10,000), maternal age (< 20 years, 1.31/10,000; 20–24 years, 1.63/10,000; 25–29 years, 1.80/10,000; 30–34 years, 1.87/10,000; ≥ 35 years, 2.22/10,000), and geographic region (central, 1.64/10,000; east, 2.45/10,000; west, 1.37/10,000). Cardiovascular anomalies were the most common malformations associated with CDH. Infants with associated CDH had a higher risk of premature birth and perinatal death than those with isolated CDH.
Conclusion
The increasing prevalence and high perinatal mortality rate of CDH highlight the need for further etiological, epidemiological, and clinical studies among the Chinese population.
Video Abstract
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Congenital diaphragmatic hernia (CDH) is a structural birth defect characterized by incomplete formation of the diaphragm, which allows abdominal organs to herniate into the thoracic cavity and impair pulmonary and vascular development. The diaphragm typically develops between the 4th and 8th weeks of gestation [1]. Most cases of CDH are left-sided but right-sided, and in rare cases, bilateral hernias can also occur [2]. The vast majority of CDH cases are isolated, with only approximately one-third being associated with other anomalies [3].
Previous studies have reported that the prevalence rate of CDH in the Chinese population was 0.7 per 10,000 births during the period from 1986 to 1993 [4]. In contrast, the prevalence of CDH in other countries has been reported to range from 1.93 to 3.80 per 10,000 births, depending on the study population and time period [1, 5,6,7,8,9]. Regardless of the specific type of CDH, all cases are associated with a poor prognosis. The overall perinatal mortality rate for CDH has been reported to be as high as 84.1% in China [4], 65.5% in Australia [9] and 32.7% for stillbirths and early neonatal death in the United States [7]. Although the pathogenesis of CDH remains largely unknown, several potential teratogenic causes have been proposed, including quinine, phenmetrazine, nitrofen, and vitamin A deficiency [10,11,12]. In addition, maternal age, maternal residence, and male birth have also been suggested as factors associated with the prevalence of CDH, but the correlations are inconsistent [1, 6, 7, 13, 14]. In recent decades, China has undergone significant changes in its economy, environment, and maternal and child health [15]. To gain new insights into the epidemiology of CDH, we conducted a study using data from the Chinese Birth Defects Monitoring Network (CBDMN) from 2007 to 2019. Our study aimed to investigate the prevalence and time trends of CDH, with a particular focus on perinatal outcomes, associated malformations, and potential associations with maternal and infant characteristics.
Methods
Data source and quality
The CBDMN is a well-established, nationwide hospital-based birth defect surveillance system with the largest sample size and geographical coverage in China, managed by the National Health Commission. It covers 763 member hospitals in 31 provinces, municipalities, or autonomous regions and currently monitors approximately 1.6 million births annually, accounting for more than 10% of annual births in China [16]. For this study, data from 1 January 2007 to 31 December 2019 were derived from the CBDMN. All births (live birth, stillbirths, and terminations of pregnancy) with a gestational age ≥ 28 weeks, obtained from delivery or medical records in member hospitals, were examined for congenital malformations by trained obstetrics, pediatrics, and ultrasound experts. The maximum diagnosis time for a congenital malformation case was the seventh day after birth. At each member hospital, trained professionals collected data on births and birth defect cases using standardized forms. All anomalies in the CBDMN database were coded according to the International Classification of Disease 10th version (ICD-10) by a national panel. A detailed description of the three-level (county, provincial, and national) data collection and quality management network can be found elsewhere [17, 18]. Quality control of the data was performed routinely through an independent retrospective survey. A panel of senior health professionals at the three levels evaluated the completeness, accuracy, and timeliness of the data. At the county level, all data reported by member hospitals were investigated quarterly. At the provincial and national levels, data reported by approximately one-third of the member hospitals were investigated semiannually and annually, respectively. More details about the CBDMN have been published elsewhere [16, 18,19,20]. Ethical approval was not necessary since the study was based on anonymized routine surveillance data with no identifiable information on mothers. Permission was obtained from the National Health Commission of China to use the data for this analysis.
Case identification and classification
The CBDMN defined CDH as a congenital malformation characterized by herniation of abdominal contents into the thorax through a diaphragmatic defect, including total absence of the diaphragm but excluding diaphragmatic paralysis, diaphragmatic eventration, or hiatal hernia [13, 21]. CDH corresponds to ICD-10 code “Q79.0”. Diagnostic approaches commonly used to identify CDH cases include ultrasound sonography, magnetic resonance imaging, and X rays. In our study, CDH cases were classified into two mutually exclusive groups: isolated, defined as the absence of any other Q or ICD-10 codes other than Q79.0 in the CBDMN register, and associated or non-isolated, when other additional codes existed.
Statistical standards and methods
The prevalence rate of CDH was calculated as the number of cases of CDH per 10,000 births (live births, stillbirths and terminations of greater than or equal to 28 weeks of gestational age). The rates were analyzed according to the following demographic characteristics: year of birth, geographic location, maternal residence, maternal age, and infant sex. Maternal residence was defined as the mothers’ residence during pregnancy and was classified as urban (cities and urbanized areas/towns) or rural (villages or countryside) based on the mother’s last residence for at least one year [17]. In our study, we classified China’s 31 provinces into three regions based on their geographical location and level of economic development [18]. The central region included 10 provinces: Hebei, Shanxi, Jilin, Heilongjiang, Anhui, Jiangxi, Henan, Hubei, Hunan, and Hainan. The eastern region included nine provinces: Beijing, Tianjin, Liaoning, Shanghai, Jiangsu, Zhejiang, Fujian, Shandong, and Guangzhou. The western region included 12 provinces: Inner Mongolia, Guangxi, Sichuan, Chongqing, Guizhou, Yunnan, Xizang, Shaanxi, Gansu, Qinghai, Ningxia and Xinjiang. Maternal age was categorized into five age groups: < 20 years, 20–24 years, 25–29 years, 30–34 years, and ≥ 35 years [20]. We also compared the timing of diagnosis (postpartum and antenatal), distribution of gestational age (< 37, 37–42, and ≥ 42 weeks of gestation), and perinatal outcome (stillbirths/terminations, early neonate death and alive within 7 days) between isolated and associated cases. Additionally, we analyzed the differences in demographic and associated deformity characteristics between CDH cases that survived and those that did not.
Prevalence rates and their 95% confidence intervals (95% CI) were estimated using the Poisson distribution. Poisson regression was used to calculate the adjusted prevalence rate ratios (aPRRs) and their 95% CIs. When calculating the aPRR for each factor (birth year, maternal residence, maternal age, geographic region, and infant sex), we controlled for the effects of others. Time trends in prevalence over the study period were analyzed using joinpoint regression. Changes in the prevalence of overall, isolated and associated CDH are presented as the average annual percentage change. Chi-square tests were used to examine differences in the timing of diagnosis and perinatal outcomes between isolated and associated cases, as well as differences in demographic characteristics and associated deformities between surviving and deceased cases. Data analysis was performed using R version 4.0.2 (the Comprehensive R Archive Network: http://cran.r-project.org). Additionally, Joinpoint regression was conducted using the Joinpoint Regression Program (version 4.9.0.1; Statistical Research and Applications Branch, National Cancer Institute, Bethesda, MD, USA). The statistical significance level for α was set at 0.05.
Results
Prevalence rates of CDH
Between 2007 and 2019, we identified a total of 4397 cases of CDH among 24,158,029 births. Of these cases, 2737 (62.2%) were isolated, and 1660 (37.8%) were associated with other conditions. The prevalence rate of overall CDH was 1.82 per 10,000 births (95% CI = 1.77–1.87), with a prevalence of 1.13 per 10,000 births (95% CI = 1.09–1.18) for isolated CDH and 0.69 per 10,000 births (95% CI = 0.65–0.72) for associated CDH.
Table 1 displays the prevalence of CDH by selected demographic characteristics. The prevalence of both overall and isolated CDH showed a considerable male predominance. Mothers residing in urban areas had a significantly higher probability of having infants with CDH than those residing in rural areas (Table 1, Fig. 1). An upward trend was observed for maternal age-specific prevalence rates of overall, isolated, and associated CDH (Table 1). However, after accounting for heterogeneity between maternal residence, geographic region, infant sex and year of delivery, no statistically significant associations were found with maternal age (Fig. 1). Compared to newborns whose mothers resided in western regions, newborns whose mothers resided in central regions had a 1.10–1.37-fold higher prevalence rate of overall, isolated and associated CDH, while newborns whose mothers resided in eastern regions had a 1.72–1.89-fold higher prevalence rate of these conditions (Table 1, Fig. 1).
The trend of congenital diaphragmatic hernia
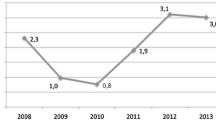
From 2007 to 2019, the annual prevalence rates of overall, isolated, and associated CDH increased from 1.06/10,000 to 2.51/10,000, from 0.75/10,000 to 1.53/10,000, and from 0.31/10,000 to 0.99/10,000, respectively (Table 1 and Fig. 2). The prevalence rate of associated CDH showed the highest upward trend, with an annual percent change of 9.4%, followed by a significant increase in the overall prevalence rate of 7.3%. In comparison, the prevalence of isolated cases rose moderately by 6.2% per year. Compared to 2007, the prevalence of isolated CDH in 2019 increased by one time, while the prevalence of associated CDH increased by three times.
Prenatal detection
As shown in Supplementary Table 1, antenatal diagnosis was available for 87.4% of the overall cases. Among the 3843 patients, 2669 (69.5%) underwent termination of pregnancy. The median gestational age at prenatal diagnosis for overall, isolated and associated cases was 25, 26 and 25 weeks, respectively.
Associated anomalies
Of the 1660 CDH cases with additional anomalies, 49.5% of associated cases had only one additional malformation, while 50.5% had two or more extra anomalies. Cardiovascular anomalies were the most commonly associated with CDH, occurring in 999 (60.2%) cases, followed by musculoskeletal anomalies (18.7%). Chromosomal abnormalities were present in 51 (3.1%) cases (Table 2).
Perinatal outcomes
Table 3 shows the perinatal outcomes of CDH cases. Preterm births accounted for 66.2% of overall cases, with more associated CDHs born prematurely than isolated cases. Only 29.8% of CDH cases survived the perinatal period, with a lower survival rate among associated cases (19.9%). The perinatal mortality rates were 70.3% for overall cases, 80.1% for associated CDH, and 64.2% for isolated cases.
Among surviving cases, a higher proportion were male infants or from the eastern geographic regions. Among deceased cases, almost 95.0% were diagnosed prenatally, and 86.6% were of low gestational age (Supplementary Table 2). The percentage of concomitant cardiovascular system abnormalities was higher among surviving cases, while nervous system and musculoskeletal system abnormalities were more common among deceased cases (Supplementary Table 3).
Discussion
Our study found that the prevalence of CDH was 1.82 per 10,000 births and showed an increasing trend over time. Higher prevalence rates were observed among male fetuses, older mothers, and mothers residing in urban areas, particularly in the eastern and central regions. Additionally, infants with associated CDH had a higher risk of premature birth and perinatal death compared to those with isolated CDH.
The prevalence rate of overall CDH is lower than rates reported in studies from the United States [7], France [22], Australia [9], Finland [23], Croatia [24], Sweden [25], and other European countries [1]. One possible explanation for the difference between our study and others is that our surveillance is hospital-based, while others are population-based. However, when compared to other hospital-based surveillance programs in Argentina, Cuba, Japan, and Saudi Arabia, our prevalence is still lower [21, 26]. Our study also revealed a lower prevalence in both isolated (1.13/10,000 births) and associated cases (0.69/10,000 births) [1, 6]. Factors such as birth definition, racial characteristics, and time range may contribute to these differences in prevalence rates.
An increasing trend in the prevalence of CDH was observed during the study period. However, trends in CDH prevalence varied across different countries and regions. For example, no change was seen in California from 1989 to 1997 [6] or in the United States from 1995 to 2002 [7]. In contrast, our analysis showed an upward trend in CDH prevalence over time, consistent with the studies conducted in the Middle East, Europe, North America, Central America, and South America [1, 21]. Our upward trend was even more significant, with an average annual percent change of 7.32%, compared to 0.47% in the aforementioned regions [21]. The rising trend of CDH prevalence in China may be attributed to improvements in prenatal diagnosis, including better and more accessible ultrasonography. Changes in maternal environmental exposures and other factors associated with CDH may also contribute to the increasing trend.
Our study showed a higher risk of CDH among older mothers, consistent with previous studies [6, 7, 27, 28], which was also comparable to studies that found no association or observed a slight, non-significant increase in prevalence among older maternal age groups [1, 6, 29]. Furthermore, our finding that males are at an approximately 20% higher risk than females for developing CDH is in general agreement with previous studies [6, 7, 13, 30]. In our study, higher prevalence rates were found in the eastern and central regions and in urban areas, suggesting a possible role for environmental factors in the pathogenesis of CDH.
CDH can be diagnosed prenatally or postnatally. The percentage of patients with CDHs diagnosed prenatally by ultrasound has significantly increased over the last 20 years, from 15% to 50%–75% [31]. In our study, 87.4% of the overall CDH cases were diagnosed prenatally, close to the 84.1% reported in other findings [32]. This is likely due to advances in ultrasonic diagnosis technology. Consistent with previous studies [1, 22, 33, 34], we found that the prenatal detection rate was higher for the non-isolated CDH cases than for the isolated cases. This may be because non-isolated CDH can be detected earlier by prenatal ultrasound [22, 33]. Our study and others confirm that there is large national variation in the prenatal detection rate of CDH due to differences in policies regarding antenatal routine ultrasound screening [1]. Associated anomalies may also contribute to the prenatal detection of CDH fetuses.
CDH is known to be associated with other structural anomalies and chromosome abnormalities, with the proportion of associated anomalies ranging from 28.2% to 85.3% [1, 6, 9, 13]. The large range of changes may be due to the differences in the CDH case confirmation and data sources. In our analysis, approximately one-third of CDH patients had associated anomalies, consistent with other studies [3, 13, 21, 31, 35]. Specifically, we observed the highest frequency of co-occurring cardiovascular anomalies, followed by musculoskeletal anomalies and nervous system malformations, which are the same as those of previous investigations [6]. Accompanying the high incidence of congenital heart disease in CDH is increasing evidence of fetal ventricular hypoplasia, characterized by a narrowing and elongation of the left ventricle [36,37,38,39]. The development of fetal left ventricle hypoplasia is likely multifactorial, secondary to direct mechanical compression of the left ventricle by the herniated abdominal viscera and flow-related mechanisms [40]. Left ventricle hypoplasia could be a risk factor for early postnatal ventricular dysfunction, increasingly recognized as a contributor to CDH pathophysiology and outcome [41].
Mortality rates for CDH patients vary considerably in the literature. Our study found an overall perinatal mortality rate of 70.3%, higher than the mortality of 42%–68% reported in other studies [9, 42, 43]. Differences in measuring mortality among CDH patients make it difficult to accurately evaluate variations between studies, which may be due to the presence of “hidden mortality” [42, 44]. “Hidden mortality” refers to the exclusion of intrauterine deaths and induced terminations from institution-based studies. Therefore, recently reported increases in survival rates should be interpreted with caution. Our study found that the mortality rate of stillbirths and terminations was 62.2%, which partially accounted for the “hidden mortality”. Despite accounting for “hidden deaths” in our mortality calculations, our study still reported a higher perinatal mortality rate compared to the literature [22, 42, 45]. This variation may be attributed to differences in the type of registry (national hospital-based vs. regional population-based) [22], the time period considered (perinatal vs. neonatal) [42], and the populations studied (all births vs. postmortems) [45]. Because 29.8% of CDH cases survived the perinatal period, we were unable to obtain information on subsequent treatment. However, other studies have confirmed that live births with CDH almost always receive treatment [24].
The rate of preterm delivery in our study appeared to be higher than that reported by another registry [7, 46]. CDH cases with a gestational age of less than 37 weeks were classified as premature, regardless of whether they resulted in live births, stillbirths, or terminations in our study. After excluding cases of pregnancy termination, the rate in our study was slightly lower than that reported in other studies [7, 46], although the difference was not statistically significant. As such, we hypothesized that the high rate of preterm birth observed in our study may be partly attributable to the high rate of pregnancy termination. In agreement with the findings of Shanmugam et al. [46], our study found that CDH cases with associated anomalies were more likely to result in premature delivery than isolated CDH cases. Similarly, when terminations were excluded from our analysis, a higher proportion of CDH cases with associated anomalies were born prematurely compared to isolated cases.
Infants with associated CDH were at a higher risk of perinatal death (80.1%) compared with those affected by isolated CDH (63.2%), consistent with previous reports [1, 22, 43]. In addition to cardiovascular system defects, there were more musculoskeletal diseases and neurological diseases among deceased patients, indicating that multiple malformations, especially circulatory, musculoskeletal, and central nervous system anomalies, are important factors in perinatal mortality.
Using 13 years of surveillance data and covering 24 million births, this study represents the most extensive investigation to date on the birth prevalence and time trend of CDH in the Chinese population. The wide geographical coverage, consistent case ascertainment methods, and adjustment for several characteristics ensure reliable estimates of the prevalence of CDH and the potential relationship between the maternal and infant characteristics and risk for CDH.
This study has several limitations. Firstly, the observed CDH prevalence may be underestimated due to hospital-based samples with a short monitoring period and incomplete population coverage. Secondly, information on chromosomal anomalies and syndromes for some CDH was unavailable due to limited chromosomal testing and syndromic case ascertainment in some member hospitals. Finally, CDH cases could not be classified based on the anatomical position of the defect, as the hernia types were not included in the routine data collection.
In conclusion, our study found that the Chinese population has a relatively low risk for CDH, but challenges remain. Cardiovascular anomalies are most commonly associated with CDH. The increasing trend in prevalence, demographic risk factors, and high perinatal mortality rate highlight the need for further research on the etiology, epidemiology, and clinical management of CDH in the contemporary Chinese population.
Data availability
The Chinese Birth Defects Monitoring Network database is not open access publicly available. The corresponding author obtained permission to use the data for this analysis from the National Health Commission of China. The datasets used and analyzed during the study are available from the corresponding author on reasonable request.
References
McGivern MR, Best KE, Rankin J, Wellesley D, Greenlees R, Addor MC, et al. Epidemiology of congenital diaphragmatic hernia in Europe: a register-based study. Arch Dis Child Fetal Neonatal Ed. 2015;100:F137–44.
Larsen UL, Jepsen S, Strøm T, Qvist N, Toft P. Congenital diaphragmatic hernia presenting with symptoms within the first day of life; outcomes from a non-ECMO centre in Denmark. BMC Pediatr. 2020;20:196.
Hidaka N, Ishii K, Mabuchi A, Yamashita A, Ota S, Sasahara J, et al. Associated anomalies in congenital diaphragmatic hernia: perinatal characteristics and impact on postnatal survival. J Perinat Med. 2015;43:245–52.
Zhu J, Wang Y, Miao L. Epidemiological studies on 321 children with congenital diaphragmatic hernia in China. Zhonghua Yu Fang Yi Xue Za Zhi. 1997;31:266–8 (in Chinese).
Torfs CP, Curry CJ, Bateson TF, Honoré LH. A population-based study of congenital diaphragmatic hernia. Teratology. 1992;46:555–65.
Yang W, Carmichael SL, Harris JA, Shaw GM. Epidemiologic characteristics of congenital diaphragmatic hernia among 2.5 million California births, 1989–1997. Birth Defects Res A Clin Mol Teratol. 2006;76:170–4.
Balayla J, Abenhaim HA. Incidence, predictors and outcomes of congenital diaphragmatic hernia: a population-based study of 32 million births in the United States. J Matern Fetal Neonatal Med. 2014;27:1438–44.
Wright JC, Budd JL, Field DJ, Draper ES. Epidemiology and outcome of congenital diaphragmatic hernia: a 9-year experience. Paediatr Perinat Epidemiol. 2011;25:144–9.
Colvin J, Bower C, Dickinson JE, Sokol J. Outcomes of congenital diaphragmatic hernia: a population-based study in Western Australia. Pediatrics. 2005;116:e356–63.
Tibboel D, Gaag AV. Etiologic and genetic factors in congenital diaphragmatic hernia. Clin Perinatol. 1996;23:689–99.
Tenbrinck R, Tibboel D, Gaillard JL, Kluth D, Bos AP, Lachmann B, et al. Experimentally induced congenital diaphragmatic hernia in rats. J Pediatr Surg. 1990;25:426–9.
Greer JJ, Babiuk RP, Thebaud B. Etiology of congenital diaphragmatic hernia: the retinoid hypothesis. Pediatr Res. 2003;53:726–30.
Dott MM, Wong LYC, Rasmussen SA. Population-based study of congenital diaphragmatic hernia: risk factors and survival in Metropolitan Atlanta, 1968–1999. Birth Defects Res A Clin Mol Teratol. 2003;67:261–7.
García AM, Machicado S, Gracia G, Zarante IM. Risk factors for congenital diaphragmatic hernia in the Bogota birth defects surveillance and follow-up program. Colombia Pediatr Surg Int. 2016;32:227–34.
Qiao J, Wang Y, Li X, Jiang F, Zhang Y, Ma J, et al. A Lancet Commission on 70 years of women’s reproductive, maternal, newborn, child, and adolescent health in China. Lancet. 2021;397:2497–536.
Gao Y, Xu W, Li W, Chen Z, Li Q, Liu Z, et al. Epidemiology and prevalence of pulmonary sequestration in Chinese population, 2010–2019. BMC Pulm Med. 2023;23:8.
Deng K, Dai L, Yi L, Deng C, Li X, Zhu J. Epidemiologic characteristics and time trend in the prevalence of anotia and microtia in China. Birth Defects Res A Clin Mol Teratol. 2016;106:88–94.
Dai L, Zhu J, Mao M, Li Y, Deng Y, Wang Y, et al. Time trends in oral clefts in Chinese newborns: data from the Chinese National Birth Defects Monitoring Network. Birth Defects Res A Clin Mol Teratol. 2010;88:41–7.
Dai L, Zhu J, Liang J, Wang YP, Wang H, Mao M. Birth defects surveillance in China. World J Pediatr. 2011;7:302–10.
Xu W, Gao Y, Li W, Chen Z, Li Q, Li R, et al. A national descriptive epidemiologic analysis on congenital pulmonary airway malformation in China, 2010–2019. Pediatr Pulmonol. 2022;57:674–81.
Politis MD, Bermejo-Sánchez E, Canfield MA, Contiero P, Cragan JD, Dastgiri S, et al. Prevalence and mortality in children with congenital diaphragmatic hernia: a multicountry study. Ann Epidemiol. 2021;56:61–9.e3.
Gallot D, Boda C, Ughetto S, Perthus I, Robert-Gnansia E, Francannet C, et al. Prenatal detection and outcome of congenital diaphragmatic hernia: a French registry-based study. Ultrasound Obstet Gynecol. 2007;29:276–83.
Hautala J, Karstunen E, Ritvanen A, Rintala R, Mattila IP, Räsänen J, et al. Congenital diaphragmatic hernia with heart defect has a high risk for hypoplastic left heart syndrome and major extra-cardiac malformations: 10-year national cohort from Finland. Acta Obstet Gynecol Scand. 2018;97:204–11.
Grizelj R, Bojanić K, Vuković J, Novak M, Rodin U, Ćorić T, et al. Epidemiology and outcomes of congenital diaphragmatic hernia in Croatia: a population-based study. Paediatr Perinat Epidemiol. 2016;30:336–45.
Mesas Burgos C, Ehrén H, Conner P, Frenckner B. Maternal risk factors and perinatal characteristics in congenital diaphragmatic hernia: a nationwide population-based study. Fetal Diagn Ther. 2019;46:385–91.
International Clearinghouse for Birth Defects Surveillance and Research. Annual report. 2014. http://www.icbdsr.org/resources/annual-report/. Accessed 6 Sep 2023.
Yang P, Khoury MJ, Stewart WF, Beaty TH, Chee E, Beatty JC, et al. Comparative epidemiology of selected midline congenital abnormalities. Genet Epidemiol. 1994;11:141–54.
Hollier LM, Leveno KJ, Kelly MA, MCIntire DD, Cunningham FG. Maternal age and malformations in singleton births. Obstet Gynecol. 2000;96:701–6.
Robert E, Källén B, Harris J. The epidemiology of diaphragmatic hernia. Eur J Epidemiol. 1997;13:665–73.
Mohamed MA, Aly H. Birth region, race and sex may affect the prevalence of congenital diaphragmatic hernia, abdominal wall and neural tube defects among US newborns. J Perinatol. 2012;32:861–8.
Kalanj J, Salevic P, Rsovac S, Medjo B, Antunovic SS, Simic D. Congenital diaphragmatic hernia—a Belgrade single center experience. J Perinat Med. 2016;44:913–8.
Snoek KG, Greenough A, van Rosmalen J, Capolupo I, Schaible T, Ali K, et al. Congenital diaphragmatic hernia: 10-year evaluation of survival, extracorporeal membrane oxygenation, and foetoscopic endotracheal occlusion in four high-volume centres. Neonatology. 2018;113:63–8.
Gallot D, Coste K, Francannet C, Laurichesse H, Boda C, Ughetto S, et al. Antenatal detection and impact on outcome of congenital diaphragmatic hernia: a 12-year experience in Auvergne, France. Eur J Obstet Gynecol Reprod Biol. 2006;125:202–5.
Garne E, Haeusler M, Barisic I, Gjergja R, Stoll C, Clementi M. Congenital diaphragmatic hernia: evaluation of prenatal diagnosis in 20 European regions. Ultrasound Obstet Gynecol. 2002;19:329–33.
Skari H, Bjornland K, Haugen G, Egeland T, Emblem R. Congenital diaphragmatic hernia: a meta-analysis of mortality factors. J Pediatr Surg. 2000;35:1187–97.
Byrne FA, Keller RL, Meadows J, Miniati D, Brook MM, Silverman NH, et al. Severe left diaphragmatic hernia limits size of fetal left heart more than does right diaphragmatic hernia. Ultrasound Obstet Gynecol. 2015;46:688–94.
Ferguson DM, Gupta VS, Lally PA, Luco M, Tsao K, Lally KP, et al. Early, postnatal pulmonary hypertension severity predicts inpatient outcomes in congenital diaphragmatic hernia. Neonatology. 2021;118:147–54.
Massolo AC, Romiti A, Viggiano M, Vassallo C, Ledingham MA, Lanzone A, et al. Fetal cardiac dimensions in congenital diaphragmatic hernia: relationship with gestational age and postnatal outcomes. J Perinatol. 2021;41:1651–9.
Vogel M, McElhinney DB, Marcus E, Morash D, Jennings RW, Tworetzky W. Significance and outcome of left heart hypoplasia in fetal congenital diaphragmatic hernia. Ultrasound Obstet Gynecol. 2010;35:310–7.
Zani A, Chung WK, Deprest J, Harting MT, Jancelewicz T, Kunisaki SM, et al. Congenital diaphragmatic hernia. Nat Rev Dis Primers. 2022;8:37.
Patel N, Massolo AC, Kipfmueller F. Congenital diaphragmatic hernia-associated cardiac dysfunction. Semin Perinatol. 2020;44:151168.
Mah VK, Zamakhshary M, Mah DY, Cameron B, Bass J, Bohn D, et al. Absolute vs. relative improvements in congenital diaphragmatic hernia survival: what happened to “hidden mortality.” J Pediatr Surg. 2009;44:877–82.
Tennant PWG, Pearce MS, Bythell M, Rankin J. 20-year survival of children born with congenital anomalies: a population-based study. Lancet. 2010;375:649–56.
Harrison MR, Bjordal RI, Langmark F, Knutrud O. Congenital diaphragmatic hernia: the hidden mortality. J Pediatr Surg. 1978;13:227–30.
Brownlee EM, Howatson AG, Davis CF, Sabharwal AJ. The hidden mortality of congenital diaphragmatic hernia: a 20-year review. J Pediatr Surg. 2009;44:317–20.
Shanmugam H, Brunelli L, Botto LD, Krikov S, Feldkamp ML. Epidemiology and prognosis of congenital diaphragmatic hernia: a population-based cohort study in Utah. Birth Defects Res. 2017;109:1451–9.
Acknowledgements
The authors thank the obstetricians, pediatricians, pathologists, and other participants involved in the Chinese Birth Defects Monitoring Network, as well as Zhen-Yu Guo, Fan-Long Bu, and Zhi-Hua Wan for providing linguistic assistance. The content of this article is solely the responsibility of the authors and do not represent the official view of the National Center for Birth Defects Monitoring.
Funding
This project was supported by National Health Commission of China.
Author information
Authors and Affiliations
Contributions
CZY contributed to concept and design, statistical analysis and drafting of the manuscript, and revision of the manuscript. TJ contributed to statistical analysis and drafting of the manuscript. XWL, GYY, LWY and LZ contributed to data collection and preparation, and revision of the manuscript. ZJY contributed to data collection and preparation. DL contributed to concept and design, and revision of the manuscript. All authors had read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethical approval
The current study analyzed de-identified secondary data from the Chinese Birth Defects Monitoring Network. Ethical approval was not necessary since the study was based on anonymized routine project monitoring data with no identifiable information on mothers. Permission was obtained from the National Health Commission of China to use the data for this analysis. All procedures were performed in accordance with relevant guidelines.
Conflict of interest
No financial or non-financial benefits have been received or will be received from any party related directly or indirectly to the subject of this article. The authors have no conflict of interest to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Chen, ZY., Tao, J., Xu, WL. et al. Epidemiology of congenital diaphragmatic hernia among 24 million Chinese births: a hospital-based surveillance study. World J Pediatr 20, 712–722 (2024). https://doi.org/10.1007/s12519-023-00774-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12519-023-00774-y