Abstract
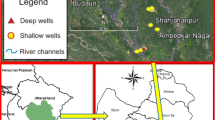
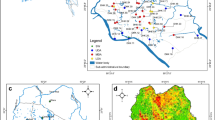
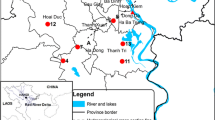
This paper conceptualises the factors responsible for water quality change in the aquifer on the basis of a study of flow dynamics, hydro-chemical, and bacteriological processes in a gypsum, lignite, oil, and gas-rich Barmer sedimentary basin in India. The change in colouration, odour, and presence of gases in flowing wells has been analysed. The study revealed that such a change in the quality of the water is mainly occurring in those wells that are located in the transition zone between unconfined and confined parts of the same aquifer. The field and lab analyses have further revealed the presence of pyrites, other iron-rich minerals, hydrogen sulphide (H2S), carbon dioxide (CO2), and methane (CH4) gases in the aquifer. The study suggests that changes in flow dynamics, hydro-chemical equilibrium, and microbiological processes like sulphate reduction, methanogenesis, and reverse methanogenesis are responsible for such changes in groundwater quality. The change in water quality in the unconfined parts of the aquifer is mainly due to in situ pyrite dissolution, bacteria-mediated sulphate reduction, or reverse methanogenesis taking place in the nearby confined parts of the aquifer. The presence of CH4 in non-flowing wells or while drilling new wells appears to be biogenic in nature and has migrated from deeper, confined parts of the same aquifer. The overexploitation of the aquifer has changed the flow dynamics and hydro-chemical equilibrium within the aquifer, and it has prompted the migration of gases from deeper, confined parts of the same aquifer. The water quality change is more prominent in the transitional zone between unconfined and confined parts of the aquifer.

















Similar content being viewed by others
References
Andersen, MS, Nyvang V, Postma D (2001) Geochemical processes at a freshwater / seawater interface in a shallow sandy aquifer, Skansehage Denmark. First International Conference on Saltwater Intrusion and Coastal Aquifers—Monitoring, Modeling, and Management. Essaouira, Morocco, April 23–25, 2001
Beeman RE, Suflita JM (1990) Environmental factors influencing methanogenesis in a shallow anoxic aquifer: a field and laboratory study. J Industr Microbiol 5:45–57. https://doi.org/10.1007/BF01569605
Celia ZR, Mathew KL, Peter BM, Janice MG, Justin TK, Michael JS, Andrew GH, David HS, Tracy AD (2021) Groundwater quality of aquifers overlying the oxnard oil field, Ventura County California. Sci Total Environ 771:144822. https://doi.org/10.1016/j.scitotenv.2020.144822
Central Ground Water Board (2019) Report on National compilation on dynamic ground water resources of India - 2017, India. https://cgwb.gov.in/GW-Assessment/GWRA-2017-National-Compilation.pdf (accessed 1 July 2003)
Chander S, Briceno A (1987) A kinetics of pyrite oxidation. J Mining Metal Explor 4:171–176. https://doi.org/10.1007/BF03402687
Compton PM (2009) The geology of the Barmer basin, Rajasthan, India, and the origins of its major oil reservoir, the Fatehgarh formation. J Petrol Geosci 15(2):117–130. https://doi.org/10.1144/1354-079309-827
Conrad O, Bechtel B, Bock M, Dietrich H, Fischer E, Gerlitz L, Wehberg J, Wichmann V, Boehner J (2015) System for Automated Geoscientific Analyses (SAGA) v. 2.1.4. Geosci Model Dev 8:1991–2007. https://doi.org/10.5194/gmd-8-1991-2015
David L. Sturrock, Ashok Kumar, Anurag Pattnaik, Nigel Banks, and Michael Price, (2008). Geology and geocellular modelling of a saline aquifer: from integration of hydrogeology and petroleum geology in a study of the Thumbli Formation, Rajasthan, North-West India. GEO India Expo XXI, Noida, New Delhi, India 2008©AAPG Search and Discovery. DOI: https://doi.org/10.1007/BF01809852
Ghaderi SM, Keith DW, Lavoie R, Leonenko Y (2011) Evolution of hydrogen sulfide in sour saline aquifers during carbon dioxide sequestration. Int J Greenhouse Gas Control 5(2):347–355. https://doi.org/10.1016/j.ijggc.2010.09.008
Harbaugh AW, Langevin CD, Hughes JD, Niswonger RN, Konikow LF (2017) MODFLOW-2005 version 1.12.00, the U.S. Geological Survey modular groundwater model: U.S. Geological Survey Software Release, 03 February 2017, https://doi.org/10.5066/F7RF5S7G
Heinen W and Lauwers AM (1996) Organic sulfur compounds resulting from the interaction of iron sulfide, hydrogen sulfide and carbon dioxide in an anaerobic aqueous environment. Origins of life and evolution of the biosphere volume 26, pages131–150 (1996).
http://www.searchanddiscovery.com/abstracts/html/2008/geo_india/abstracts/sturrock.htm
https://olemiss.edu/sciencenet/saltnet/swica1/Andersen-Nyvang-Postma-exabs.pdf
Humez P, Mayer B, Nightingale M, Becker V, Kingston A, Taylor S, Bayegnak G, Millot R, Kloppmann W (2016) Redox controls on methane formation, migration and fate in shallow aquifers. Hydrol Earth Syst Sci 20:2759–2777. https://doi.org/10.5194/hess-20-2759-2016
Jakobsen R, Postma D (1999) Redox zoning, rates of sulfate reduction and interactions with Fe-reduction and methanogenesis in a shallow sandy aquifer, Rømø. Denmark Geochimica Et Cosmochimica Acta 63(1):137–151. https://doi.org/10.1016/S0016-7037(98)00272-5
Junfeng Z, Thomas MP, Charles JT, Steven E, Webb BD, Richard S, Stephen DR, Lisa JM, Jenna SK, Ann PS (2018) Assessing methane in shallow groundwater in unconventional oil and gas play areas Eastern Kentucky. Groundwater J 56(3):413–424. https://doi.org/10.1111/gwat.12583
Kimura H, Nashimoto H, Shimizu M, Hattori S, Yamada K, Koba K, Yoshida N, Kato K (2010) Microbial methane production in deep aquifer associated with the accretionary prism in Southwest Japan. ISME J 4:531–541. https://doi.org/10.1038/ismej.2009.132
Knöllera K, Schubertb M (2010) Interaction of dissolved and sedimentary sulfur compounds in contaminated aquifers. Chem Geol 276(3–4):284–293. https://doi.org/10.1016/j.chemgeo.2010.06.013
Mayrhofer C, Niessner R, Baumann T (2014) Hydrochemistry and hydrogen sulfide generating processes in the Malm aquifer, Bavarian Molasse Basin Germany. Hydrogeol J 22:151–162. https://doi.org/10.1007/s10040-013-1064-2
McFarland ML, Provin TL (1998) Hydrogen sulfide in drinking water - causes and treatment alternatives. Report, Agrilife Extension, Texas A & University, Oregon. http://soiltesting.tamu.edu/publications/L-5312.pdf
Michael TW, Peter BM, Matthew KL, Justin TK (2019) Groundwater quality of a public supply aquifer in proximity to oil development, Fruitvale oil field Bakersfield California. Appl Geochemistry 106(2019):82–95. https://doi.org/10.1016/j.apgeochem.2019.05.003
Mukhopadhyay A, Al-Haddad A, Al-Otaibi M, Al-Senafy, (2007) Occurrence of hydrogen sulfide in the ground water of Kuwait. Environ Geol 52:1151–1161. https://doi.org/10.1007/s00254-006-0552-2
Nelson D (2002) Natural variations in the composition of groundwater, drinking water program, Groundwater Foundation annual meeting, Report, Department of Human Services, Oregon, https://pdfs.semanticscholar.org/1006/667a49c78e3944ba4f55ac6637391c349d04.pdf
Parkhurst DL, Appelo C AJ (2020) Description of input and examples for PHREEQC Version 3 - a computer program for speciation, batch-reaction, one-dimensional transport, and inverse geochemical calculations. https://water.usgs.gov/water-resources/software/PHREEQC/documentation/phreeqc3-html/phreeqc3.htm
Rathore SS (2001) The corrosive well water of Bhadkha village, Block – Sheo, District – Barmer, Unpublished report, Public Health Engineering Dept, Govt. of Rajasthan, Barmer
Santo E CD, Silva J CM, Duarte HA (2016) Pyrite oxidation mechanism by oxygen in aqueous medium, The Journal of Physical Chemistry, Universidade Federal de Minas Gerais, Brazil, 31.270–901:2760–2768, https://pubs.acs.org/doi/pdf/https://doi.org/10.1021/acs.jpcc.5b10949
Talebmorad H, Ostad-Ali-Askari K (2022) Hydro geo-sphere integrated hydrologic model in modeling of wide basins. Sustain. Water Resour Manag.8, 118 (2022). https://doi.org/10.1007/s40899-022-00689-y
Timmers P HA, Welte CU, Koehorst JJ, Plugge CM, Jetten MSM, Stams AJM (2017) Reverse methanogenesis and respiration in methanotrophic archaea, Archaea, 2017, Article ID 1654237, 22 pages, https://doi.org/10.1155/2017/1654237
Winston RB (2019) ModelMuse version 4—a graphical user interface for MODFLOW 6: U.S. Geological Survey Scientific Investigations Report 2019–5036, 10 p. https://doi.org/10.3133/sir20195036
Yoshioka H, Takeuchi M, Sakata S, Takahashi Hiroshi A, Takahashi M, Tanabe S, Hayashi T, Inamura A, Yasuhara M (2020) Microbial methane production and oxidation in the Holocene mud beneath the Kanto Plain of central Japan. Geochemical J 54(4):243–254. https://doi.org/10.2343/geochemj.2.0597
Zhi-P W, Zhang L, Wang B, Long-Y H, Chun-W X, Xi-M Z, Xing-G H (2018) Dissolved methane in groundwater of domestic wells and its potential emissions in arid and semi-arid regions of Inner Mongolia China. Sci Total Environ 626:1193–1199. https://doi.org/10.1016/j.scitotenv.2018.01.205
Acknowledgements
The author acknowledges Cairn Oil and Gas for providing the opportunity for this preliminary study while working with the company. The author is also thankful to Mr. Gopal Singh Bhati, Chief Engineer (Retd.), Public Health Engineering Department, Government of Rajasthan, for sharing the historical field data and his field observations while executing water well drilling projects in the area. The author also acknowledges the support of Ranjan Sinha, Hanuman Panwar, and Imran Khan of Cairn Oil and Gas for their assistance in the collection of field data. The opinion expressed in this paper is in no way to be treated as the opinion of Cairn Oil and Gas.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The author declares no competing interests.
Additional information
Responsible Editor: Broder J. Merkel
Highlights
• Over exploitation of groundwater changed flow dynamics and hydro-geo-chemical conditions in the aquifer.
• In a shallow aquifer, hydrogen sulphide, carbon dioxide, and methane gases are present.
• Both hydrogeochemical and microbiological processes are responsible water quality change in aquifer.
• Hydro-chemical change is dominant in the transitional zone of unconfined and confined parts of the aquifer.
Appendix 1
Appendix 1
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kumar, A. Conceptualization of the recent change in groundwater quality of the shallow aquifer of the hydrocarbon-rich Barmer sedimentary basin, Rajasthan, India. Arab J Geosci 16, 306 (2023). https://doi.org/10.1007/s12517-023-11396-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12517-023-11396-z