Abstract
Background
82Rb PET and [15O]H2O PET are both validated tracers for myocardical perfusion imaging but have not previously been compared clinically. During our site’s transition from 82Rb to [15O]H2O PET, we performed a head-to-head comparison in a mixed population with suspected ischemic heart disease.
Methods
A total of 37 patients referred for perfusion imaging due to suspicion of coronary stenosis were examined with both 82Rb and [15O]H2O PET on the same day in rest and during adenosine-induced stress. The exams were rated by two blinded readers as normal, regional ischemia, globally reduced myocardial perfusion, or myocardial scarring. For [15O]H2O PET, regional ischemia was defined as two neighboring segments with average stress perfusion ≤ 2.3 mL/(min·g). Further, we evaluated a total perfusion deficit (TPD) of ≥ 10% as a more conservative marker of ischemia.
Results
[15O]H2O PET identified more patients with regional ischemia: 17(46%) vs 9(24%), agreement: 59% corresponding to a Cohen’s kappa of .31 [95%CI .08-.53], (P < .001). Using the more conservative TPD ≥ 10%, the agreement increased to 86% corresponding to a kappa of .62 [95%CI .33-.92], (P = .001). For the subgroup of patients with no known heart disease (n = 18), the agreement was 94%. Interrater agreement was 95% corresponding to a kappa of .89 [95%CI .74-1.00] (P < .001).
Conclusions
In clinical transition from 82Rb to [15O]H2O PET, it is important to take into account the higher frequency of patients with regional ischemia detected by [15O]H2O PET.
Graphical Abstract

Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Identification of patients with coronary artery disease (CAD) that will most likely benefit from revascularization is guided by non-invasive work-up e.g. myocardial perfusion imaging.1 For decades, myocardial perfusion imaging was used with single photon emission computed tomography (SPECT) with e.g. [99mTc]Sestamibi or other irreversible tracers which are extracted into the myocardium dependent on the level of perfusion and not significantly redistributed back into the vascular system. For these irreversible tracers, regionally reduced perfusion during stress but not during rest is referred to as stress-induced ischemia. Reduced perfusion during both stress and rest, matched defects, are considered to be scarring.
A number of sites have switched to PET using 82Rb eluted from an 82Sr generator. The PET technology results in high-resolution images, and due to the short half-life of 82Rb (76 s), rest and stress tests can be performed shortly after each other reducing the entire examination from a two-day protocol to no more than 30 minutes. 82Rb is actively transported into the myocytes through the Na+/K+ pump where the tracer is trapped allowing for static imaging. Hence, the resulting perfusion images are comparable to SPECT images but with better resolution. Further, the software packages for 82Rb PET support the quantification of myocardial blood perfusion (MBF). The increase in MBF referred to as the coronary flow reserve (CFR) allows detection of globally reduced perfusion e.g. in triple-vessel disease (balanced ischemia) or microvascular disease.2 The quantification method differs between software packages,3 but are generally based on a one-tissue compartment model:
CT is the myocardium time-activity-curve and CA is the arterial input function both obtained from the dynamic PET series and ⊗ denotes the convolution operation. K1 and k2 are the influx and efflux rate constants. For the irreversible 82Rb, k2 is close to zero. The measured influx K1 represents MBF and is subsequently corrected for partial volume, motion and arterial blood volume by software package specific corrections.3
Another PET perfusion tracer, [15O]H2O is a cyclotron product with a short physical half-life (122 s) requiring an on-site cyclotron. With the rapidly developing cyclotron technology, our site has established a mini-cyclotron GENtrace (GE healthcare, Uppsala, Sweden) dedicated for production of 15O and subsequent synthesis to [15O]H2O. In contrast to the irreversible PET tracer 82Rb, [15O]H2O is a freely diffusible tracer. The software package calculation for [15O]H2O is also based on the one-tissue compartment model (Eq. 1) but contrary to 82Rb, the efflux rate constant k2 is used to achieve a more robust estimation for MBF.4,5 k2 represents the clearance of tracer from the tissue and is—at least in theory—more independent of partial volume, motion,4,6 and even attenuation correction5 which affect only the influx as k2 is measured relative to the influx. Thus, [15O]H2O PET allows for parametric images with robust MBF estimates at the expense of static robust high-resolution images obtained using 82Rb PET.
While water is freely diffusible with an extraction fraction of 1 in myocardial tissue allowing for direct measurements of perfusion, 82Rb has a limited extraction which is reduced at higher perfusion rates and correction for the lower extraction is necessary. The extraction corrections are based on the Renkin-Crone model7,8:
with PS being the permeability-surface product and E the extraction. The 82Rb quantification with correction for decreased extraction has been validated against [15O]H2O PET.9 Corrections are typically not applied to the static images used for both visual interpretation and comparison to a normal database but only to the additional quantitative MBF and CFR.
[15O]H2O has been used mainly in research since the mid 1980’s to quantify MBF. [15O]H2O PET is quantitatively validated against microspheres in pigs, which is considered the reference standard10 and to invasive pressure-measurements in coronary vessels (Fractional Flow Reserve, FFR), thermodilution and has shown prognostic value.11,12,13 In fact, early studies on FFR used [15O]H2O PET as the reference standard.14 For [15O]H2O PET, the most accurate metric for predicting an FFR-positive coronary stenosis in patients without previous cardiac disease is MBF during stress (MBFstress) in two neighboring segments ≤ 2.3 mL/(min·g) in a 17 segment model.15
The clinical transition from [99mTc]Sestamibi SPECT to 82Rb PET is straightforward with better image quality and patient experience, while the clinical transition to [15O]H2O PET may be more complicated due to the aforementioned differences in uptake mechanisms. 82Rb PET and [15O]H2O PET are both validated for heart perfusion but have not previously been compared for clinical performance in a prospective cohort of patients suspected for myocardial ischemia. During our clinical transition from 82Rb PET to [15O]H2O PET, we performed a head-to-head comparison in a mixed population with suspected myocardial ischemia and a high fraction of patients with previous heart disease. We hypothesized that there is no significant difference in the overall occurrence of detected clinically relevant myocardial ischemia as determined with 82Rb PET and [15O]H2O PET and that in the vast majority of cases, the patients would be assessed similarly.
Methods
Study population
We prospectively and consecutively included patients referred from the Department of Cardiology, Copenhagen University Hospital Bispebjerg from January to April 2022. The study was initiated when [15O]H2O production was approved at our site and during a four month overlap with continued clinical use of 82Rb. Thus all [15O]H2O PET scans were performed during the startup period. The center has a high-throughput with around 1,500 heart PET examinations per year16 and during the startup period, a number of exams were co-reviewed by experts from Turku PET Centre to ensure quality. To increase the likelihood of ischemia in the cohort, we included patients with typical anginal chest pain and with at least one of the following risk factors: a family history of cardiac disease (< 55 years for male and < 65 years for female family members), smoking, diabetes, hypertension, BMI > 30 or hyperlipidemia. Exclusion criteria were unstable angina, significant chronic obstructive lung disease or asthma, claustrophobia, acute severe illness or a significant language barrier. The study was approved by the Research Ethics Committee of the Capital Region of Denmark (ID: H-21016899) and written consent to participate were obtained from all individuals after receiving oral and written information according to the Helsinki declaration. All data were handled according to regulations by The Danish Data Protection Agency.
[15O]H2O production
[15O]H2O was produced in two steps. First, a target gas mixture (97.5% 15N2, 2.5% O2) was continuously bombarded for a few minutes with a 7.8 MeV proton beam in a GenTrace cyclotron (GE, Uppsala, Sweden) dedicated to the production of 15O. Second, for administration of [15O]H2O, the target was mixed with the transport gas (N2 with 4% H2) and pushed into an oven with a 400 °C hot palladium wire. The resulting radioactive water vapor was fed into a bedside automated production system (Hidex RWG, Hidex Oy, Turku, Finland) consisting of a dual membrane system to mix physiological saline with [15O]H2O. The resulting radioactive saline solution was injected into the patient without further user interaction.
PET scans
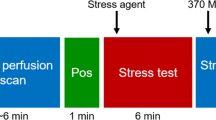
All subjects refrained from using caffeine-containing beverages and food or theophylline-containing medication for 24 hours before examination. Furthermore, phosphodiesterase type 5 inhibitors were withheld five days before examination, antithrombotic medicine containing Dipyridamole or Nicorandil two days before, extended-release nitrates 12 hours before and short-acting nitroglycerin two hours before examination. All patients were scanned using a Discovery 710 PET/CT scanner (GE Healthcare, Milwaukee, WI, USA). After a CT scan for attenuation correction and for anatomical localization, a 5 minutes dynamic emission scan in list mode was performed during resting condition after intravenous injection of 1100 MBq of 82Rb eluted from an 82Sr/82Rb generator (CardioGen-82; Bracco, Princeton, NJ). [15O]H2O PET in resting condition was mean performed earliest 10 minutes after 82Rb PET, and a dose of 394 MBq (range: 345-563 MBq) [15O]H2O was injected intravenously using a synthesis and injection system, Hidex RadioWaterGenerator (Hidex, Turku, Finland) and a 5-minutes scan was initiated simultaneously with the bolus arrival. After a 10 minutes interval to allow for decay of radioactivity, an identical PET sequence was performed during stress conditions induced by intravenous adenosine infusion (140 μg/kg/min) for 6 minutes. Adenosine was started 2 minutes prior to the stress PET scans to achieve maximum hyperaemia. We did not randomize the order of 82Rb and [15O]H2O PET as several patients could not cooperate to two adenosine infusions and we aimed to ensure a clinically useful 82Rb PET examination. Reconstruction of dynamic PET images was performed using ordered subset expectation maximization (OSEM) with Time of Flight (ToF) (2 iterations, 24 subsets and 6.4 mm in-plane filtering). For 82Rb a static reconstruction for the last 150 s was used. The Corridor 4DM software version 2018 (Invia Medical Imaging Solutions, Ann Arbor, MI, USA) was used for the analysis of 82Rb PET data, while CarimasCE software version 1.3.1. (Turku, Finland) was used for [15O]H2O PET data. Corridor 4DM estimates quantitative MBF from 82Rb K1 measurements using a modified version of Equation 2 according to Lortie17:
PET interpretation
82Rb PET
82Rb PET was evaluated as part of daily clinical routine by a nuclear medicine specialist with > 10 years of experience in myocardial imaging. Previous studies have shown good inter-observer agreement.18,19 82Rb PET were assessed visually using the ‘splash’ images using polar plots with relative differences compared to a normal database. The degree of relative defects within the myocardium was rated using 17 segments and 4 degrees of reduction, i.e. maximum total score 68. A stress defect score of 7 or more (i.e. ≈10%) of the myocardial wall was considered significant. Based on the clinical readings, the 82Rb PET was classified into four groups (normal, regional ischemia, globally reduced myocardial perfusion and myocardial scarring, see Table 1 for definitions). To further simplify data, the classifications were reduced to two groups of normal and regional ischemia, the latter included global reduction with suspicion of triple-vessel disease. The vascular territory involved was noted as left anterior descending artery (LAD), left circumflex artery (LCX) or right coronary artery (RCA).
[15O]H2O PET
[15O]H2O PET was assessed according to Danad15 with a cut-off of two neighboring segments with MBFstress≤ 2.3 mL/(min·g). Additionally, in line with 82Rb, total perfusion deficit (TPD) was calculated as a score of segmental reduction in percentage according to the following limits: normal (MBFstress > 2.3; score 0), mildly reduced (2.0< MBFstress ≤ 2.3; score 1), moderately reduced (1.7< MBFstress ≤ 2.0; score 2), severely reduced (1.4< MBFstress ≤ 1.7; score 3), and very severely reduced (MBFstress ≤ 1.4; score 4). TPD ≥ 10% was tentatively considered significant for ischemia. TPD is not a validated measure in [15O]H2O PET but was introduced in our clinic during the training period as we quickly realized that the threshold of two segments ≤ 2.3 mL/(min·g) identified too many patients with ischemia in the current patient population, which was also demonstrated in a recent study in patients with previous myocardial infarction or PCI.20 All [15O]H2O PET images were evaluated after the startup period had ended and [15O]H2O PET had been clinical routine for 2 months. Readers had > 10 years of experience in nuclear cardiology (UT and MK). [15O]H2O PET images were randomized and readers were blinded to the result of the 82Rb PET but with full access to all clinical data to simulate daily clinical routine. As for 82Rb, each [15O]H2O PET examination was classified into four groups based on the two segment threshold with MBFstress≤ 2.3 mL/(min·g) and two groups based on the aforementioned TPD of 10% (see Table 1 for definitions). The involved vascular territories (LAD, LCX and/or RCA) were noted. In case of discrepancy between the readers, consensus reading was performed. If a scan was classified with both a regional ischemia and global perfusion reduction or scarring, the regional defect overruled the other findings. To assess how previous heart disease affected the agreement, we performed a subgroup analysis of patients without known heart disease.
Statistics
Data are reported with mean and standard deviation or median and interquartile range. Differences in heart rate response during adenosine infusion were compared using a paired Student’s t-test. Using SPSS (IBM SPSS Statistic version 25). Agreement was defined as the number of identical classification divided by the total number of patients. Cohen’s kappa was used to compare agreement between methods and between readers for the two and four group comparisons when sample sizes were sufficient. Kappa-values were evaluated according to Altman: κ ≤ .2: poor, .2 < κ < .4: fair, .4 < κ < .6: moderate, .6 < κ < .8: good, κ > .8: very good.21 McNemar’s test were used to test for agreement between tracers with the 2 × 2 contingency tables. Pearson’s correlation coefficient was used to compare the measured perfusion with the two methods. The K1 values were calculated from 82Rb MBF estimates using Equation 3 to compare the apparent 82Rb uptake in static images to the parametric [15O]H2O PET MBF images.
Results
A total of 57 patients were included. A number of subjects were subsequently excluded due to cancellation (n = 1), [15O]H2O production failure (n = 3), intravenous access failure (n = 1), not able to receive adenosine twice (n = 4), or technical failure (n = 5) leaving 43 patients for the analysis. During reading, 6 additional examinations were excluded due to excessive movement artefacts (n = 4) or insufficient vasodilatation (n = 2) leaving 37 patients with diagnostic scans with both tracers (Suppl. Figure 1). Table 2 lists demographic and clinical data of the included patients. Please note the high number of patients with significant previous heart disease. Infusion of adenosine elicited a heart rate response from on average 67.0 (± SD 9.1) to 89.6 (± 12.1) during 82Rb scan and a slightly diminished (p < .005) response from 69.6 (± 11.7) to 87.8 (± 13.1) during the [15O]H2O scan, which was probably due to habituation.
Please refer to Table 3 for comparison of the 82Rb PET and consensus [15O]H2O PET ratings for the entire group and Table 4 for patients without previous heart disease. We found more positive findings with [15O]H2O than 82Rb PET as 82Rb PET identified 9 (24%) patients with regional ischemia vs 17 (46%) patients by [15O]H2O PET. Of the 9 positive findings with 82Rb PET only one was negative with [15O]H2O PET. Agreement between the tracers when rating into four groups was 59% corresponding to a fair kappa of .31 [95%CI .08-.53], (P < .001) for the entire population (Table 3A), and 72% for patients with no previous heart disease (Table 4A).
Rating into two groups with and without a TPD > 10%, the number of positive findings was reduced from 17 to 10 for [15O]H2O PET and from 9 to 8 for 82Rb PET, indicating the importance of selecting a proper cut-off. The agreement increased to 86% for the entire population corresponding to a good kappa of .62 [95%CI .33-.92], (P = .001) (Table 3B) and 94% for the patients with no previous heart disease (Table 4B). No significant differences between tracers was found for the entire population or the patients with no previous heart disease (McNemar, P = .16 and P = .50, respectively). A total of six patients had a TPD > 10% for both tracers and five of these had identical scoring of vascular territory.
Table 5 shows the cases with discrepancies, and Figure 1 shows examples of agreement and disagreement. The inter-reader agreement of [15O]H2O PET was 84% for four group classification corresponding to a good kappa of .73 [95%CI .54-.91] (P < .001), and for classification in two groups, the agreement was 95% corresponding to a very good kappa of .89 [95%CI .74-1.00] (P < .001).
Patient A: Example of agreement. 61 year old man with angina but without prior known CAD. Both tracers showed large perfusion defects (82Rb: 25% and [15O]H2O: 54%) during stress in LAD and RCA. Patient B. Example of disagreement. 74 year old man with angina, a number of risk factors for CAD and dilated left atrium. 82Rb showed homogenous tracer uptake while [15O]H2O showed a defect of 15% in LCX and/or RCA
MBF obtained with the two methods correlated significantly but lower values were measured at high perfusion for 82Rb compared to [15O]H2O—even after correction for the lower extraction of 82Rb (see Figure 2): Figure 2a: Rest: r = .75 [95%CI .56-.86], P < .000001; stress: r = .62 [95%CI .37-.79], P < .0001; and Figure 2b for uncorrected 82Rb K1 and [15O]H2O MBF values: Rest: r = .77 [95%CI .59-.87], slope = .33, P < .0000001 and stress: r = .62 [95%CI .38-.79], slope = .10, P < .0001.
A Correlation between myocardial blood flow (MBF) measured with 82Rb as a function of MBF measured with [15O]H2O. (Rest: r = .75 [95%CI .56-.86], P < .000001; stress: r = .62 [95%CI .37-.79], P < .0001). The 82Rb measures are corrected for a flow-dependent lower extraction according to Lortie 17. B: Correlation between uncorrected 82Rb K1 and [15O]H2O MBF values (rest: r = .77 [95%CI .59-.87], slope = .33, P < .0000001; stress: r = .62 [95%CI .38-.79], slope = .10, P < .0001). Note the clear bias with lower perfusion values using 82Rb, even when correcting for reduced extraction in A. Blue represent resting conditions and red represent stress conditions
The CFR obtained with the two tracers also correlated significantly (Figure 3) (r = .66 [95%CI .42-.81], P < .0001) and also with systematically lower values for 82Rb compared to [15O]H2O. Note that the cut-off from the literature as depicted by the solid lines is also lower for 82Rb. A total of 6 patients (marked red) would be classified differently in terms of CFR by the two tracers and one of these had different final ratings.
CFR compared for the two tracers (r = .66 [95%CI .42-.81], P < .0001). Note that the cut-offs for reduced CFR are different15. It is noteworthy that four subjects (marked red) were regarded normal according to the 82Rb scan and had too low CFR according to the [15O]H2O scan, and additional two subjects (also marked red) were opposite below normal for 82Rb but not for [15O]H2O
Discussion
This is the first direct comparison of myocardial perfusion PET with 82Rb and [15O]H2O for diagnosing obstructive CAD in a mixed population of patients with more than 50% having previous heart disease. Only fair agreement (P < .001) was found with [15O]H2O PET reporting ischemia more frequently than 82Rb PET. Applying a TPD cut-off of 10% improved agreement with 82Rb but still identified three more subjects with ischemia.
Part of the disagreement between 82Rb and [15O]H2O can be explained by the application of the cut-off of 2.3 mL/(min·g) which was determined in patients without prior CAD15 that were slightly younger (61 vs 66 years), weighed less (80 vs 86kg) and included more women (42% vs 20%). Thus, the cut-off is likely to identify too many patients with ischemia in a mixed population. This finding is in accordance with the PACIFIC 2 study,20 showing reduced specificity of [15O]H2O PET in patients with previous ischemic heart disease. However, in the subpopulation of the patients with no previous heart disease, [15O]H2O PET also seem to find regional ischemia more frequently than 82Rb PET (Table 4A). The solely use of relative perfusion defects for 82Rb may be less sensitive. We do not know to what degree [15O]H2O PET identifies patients with signs of hemodynamically significant coronary artery stenosis as determined by invasive coronary angiography (ICA) since only patients with stress-induced defects on 82Rb PET were considered for ICA. None of the patients identified with perfusion defects by [15O]H2O PET who had a normal 82Rb PET scan had subsequent angiographic imaging or were subsequently hospitalized suspected for myocardial infarction but the limited number of patients and short time of follow-up of 12 months does not rule out that the patients did indeed have significant coronary artery stenosis (see Table 5 for details of the patients with discrepancy). We anticipate that a less dichotomous interpretation of [15O]H2O PET than simply applying an MBF cut-off value of 2.3 mL/(min·g) will be needed in a mixed population, e.g. taking into account other factors potentially contributing to globally decreased stress myocardial perfusion such as previous revascularization, atrial fibrillation, decreased left ventricular ejection fraction, diabetes, and other conditions involving microvascular dysfunction. Segmental relative CFR calculation could be a possible tool to evaluate perfusion in these patients.22
82Rb has a flow-dependent lower extraction of tracer compared to the freely diffusible [15O]H2O. This is partly corrected in the quantitative measures, achieving a high correlation between MBF and CFR measured with the two methods (Figures 2, 3), although a flow-dependent bias is still clearly visible in Figure 2a. The bias in our study is more pronounced than previous studies have reported23 and must be kept in mind when comparing MBF measured with the two methods. But more importantly, the 82Rb images which are visually assessed using ‘splash’ images or polar plots are indeed images of tracer uptake, i.e. uncorrected K1 values which show a much more pronounced bias. Figure 2b shows that differences in 82Rb K1 correspond to much larger differences in the k2-derived MBF using [15O]H2O, especially for stress images, which have a slope of only .10, i.e. visual differences are about 10 times greater for [15O]H2O compared to 82Rb. This is not only true between subjects as in Figure 2b but also within subjects. Thus, the higher extraction likely explains the higher frequency of patients with regional ischemia detected by [15O]H2O.
It was shown by Danad et al15 that rest images of [15O]H2O PET were not prognostic in a setting of patients with no prior heart disease. Indeed, in some sites only stress images are obtained resulting in a highly efficient workflow. At our own institution, half of our patients had previous heart disease (Table 2) and traditionally, rest 82Rb images were used to differentiate ischemia from previous infarcts/fibrosis/scarring. Two factors reduce the clinical usefulness of rest [15O]H2O images for differentiating between scarring and ischemia. Firstly, as explained above, [15O]H2O PET shows higher variation in MBF and differentiating between true infarcts and minor relative changes may be difficult. Secondly, and more importantly, the efflux constant k2 is used instead of the influx constant K1 to assess MBF (see Equation 1). While the influx K1 is highly sensitive not only to movement and attenuation artefacts but also to tissue defects and old infarcts, k2 is a rate constant for tracer leaving myocytes, i.e. independent of infarcts and non-perfused areas. [15O]H2O PET robustly measures the perfusion of the remaining viable tissue at the expense of obscuring areas with partial fibrosis/subendocardial infarcts. Indeed, in our material three subjects were classified with scarring using 82Rb, while appearing normal on resting [15O]H2O PET (Table 3a). It is, however, not known to which degree true infarctions were present. A tissue fraction measure (perfusable tissue fraction, PTF) may be able to identify areas of non-perfused tissue6,24,25 but this has not been thoroughly validated in a population with different risk and probability for CAD and did not seem robust in our sample. Thus, PTF was not included in the present study. We found high to very high agreement between readers of [15O]H2O PET, suggesting a robust assessment tool, although the agreement must be interpreted with the notion that a number of subjects were excluded due to noisy data or motion artefacts. This may be attributed to the patients receiving two rather unpleasant infusions of adenosine combined with less routine by the technical staff in the startup period as we currently rarely experience such artefacts. Limitations include the lack of a reference standard. A reference standard as ICA with FFR measurements would result in a more selected population while a reference standard involving clinical follow-up would require a much larger sample than was possible in the present setting. Sample size is limited as a large number of patients were excluded due to production failure or noisy [15O]H2O data which was not possible to interpret and we cannot rule out that agreement would been different if the excluded cases were part of the analysis. Based on the experience, we have now applied and received permission to increase the standard injected [15O]H2O dose to 600 MBq. The population was mixed with a high fraction of patients having had previous CAD with PCI or coronary by-pass surgery, which diminished agreement between the two methods. Indeed, patients were only included if they had relatively high pre-test probability of ischemia to ensure a data set with a considerable number of patients with ischemia. Lack of randomization of the order of 82Rb and [15O]H2O PET is another limitation as the stress period of the first adenosine infusion may interfere with physiological reactions to the second infusion, which may limit the quantitative comparisons of perfusion between the methods.
New knowledge gained
In a clinical setting, we found only moderate agreement between [15O]H2O and 82Rb perfusion PET as [15O]H2O PET identifies a higher frequency of patients with regional ischemia.
Conclusion
[15O]H2O perfusion PET is a sensitive imaging modality for myocardial ischemia with a high interrater agreement. However, a number of differences exist between 82Rb and [15O]H2O perfusion PET. Primarily, we found only a moderate agreement as [15O]H2O PET identifies a higher frequency of patients with regional ischemia especially if using the literature cut-off of 2.3 mL/(min·g) determined in individuals without prior CAD. An improved agreement was found using the more conservative TPD of ≥ 10% but future studies are warranted to establish [15O]H2O PET interpretation criteria in a mixed population. Secondly, matched rest and stress perfusion defects using 82Rb interpreted as scarring are not always detected by [15O]H2O PET which likely detects only transmural scars.
Data availability
Anonymised datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- CAD:
-
Coronary artery disease
- PCI:
-
Percutaneous coronary intervention
- CABG:
-
Coronary artery bypass grafting
- ICA:
-
Invasive coronary angiography
- SPECT:
-
Single photon emission computed tomography
- PET:
-
Positron emission tomography
- CFR:
-
Coronary flow reserve
- MBF:
-
Myocardial perfusion
- MBFstress :
-
Myocardial perfusion during adenosine vasodilation
- TPD:
-
Total perfusion deficit
References
Knuuti J, Wijns W, Saraste A, Capodanno D, Barbato E, Funck-Brentano C. 2019 ESC Guidelines for the diagnosis and management of chronic coronary syndromes. Eur Heart J 2020;41:407‐77.
Højstrup S, Hansen KW, Talleruphuus U, Marner L, Galatius S, Rauf M, et al. Coronary microvascular disease assessed by 82Rb PET myocardial perfusion imaging is associated with small vessel disease of the kidney and brain. J Am Heart Assoc e028767;2023.
Nesterov SV, Deshayes E, Sciagra R, Settimo L, Declerck JM, Pan XB, et al. Quantification of myocardial blood flow in absolute terms using 82Rb PET imaging: the RUBY-10 study. JACC Cardiovasc Imaging 2014;7:1119‐27.
Koshino K, Watabe H, Enmi J, Hirano Y, Zeniya T, Hasegawa S, et al. Effects of patient movement on measurements of myocardial blood flow and viability in resting 15O-water PET studies. J Nucl Cardiol 2012;19:524‐33.
Lubberink M, Harms HJ, Halbmeijer R, de Haan S, Knaapen P, Lammertsma AA. Low-dose quantitative myocardial blood flow imaging using 15O-water and PET without attenuation correction. J Nucl Med 2010;51:575‐80.
Iida H, Kanno I, Takahashi A, Miura S, Murakami M, Takahashi K, et al. Measurement of absolute myocardial blood flow with H215O and dynamic positron-emission tomography. Strategy for quantification in relation to the partial-volume effect. Circulation 1988;78:104‐15.
Crone C. The permeability of capillaries in various organs as determined by use of the “indicator diffusion” method. Acta Physiol Scand 1963;58:292‐305.
Renkin EM. Transport of potassium-42 from blood to tissue in isolated mammalian skeletal muscles. Am J Physiol 1959;197:1205‐10.
Prior JO, Allenbach G, Valenta I, Kosinski M, Burger C, Verdun FR, et al. Quantification of myocardial blood flow with 82Rb positron emission tomography: clinical validation with 15O-water. Eur J Nucl Med Mol Imaging 2012;39:1037‐47.
Rimoldi O, Schafers KP, Boellaard R, Turkheimer F, Stegger L, Law MP, et al. Quantification of subendocardial and subepicardial blood flow using 15O-labeled water and PET: experimental validation. J Nucl Med 2006;47:163‐72.
Bom MJ, van Diemen PA, Driessen RS, Everaars H, Schumacher SP, Wijmenga JT, et al. Prognostic value of [15O]H2O positron emission tomography-derived global and regional myocardial perfusion. Eur Heart J Cardiovasc Imaging 2020;21:777‐86.
Driessen RS, Danad I, Stuijfzand WJ, Schumacher SP, Knuuti J, Maki M, et al. Impact of revascularization on absolute myocardial blood flow as assessed by serial [15O]H2O positron emission tomography imaging: a comparison with fractional flow reserve. Circ Cardiovasc Imaging 2018;11:e007417.
Everaars H, de Waard GA, Schumacher SP, Zimmermann FM, Bom MJ, van de Ven PM, et al. Continuous thermodilution to assess absolute flow and microvascular resistance: validation in humans using [15O]H2O positron emission tomography. Eur Heart J 2019;40:2350‐9.
De Bruyne B, Baudhuin T, Melin JA, Pijls NH, Sys SU, Bol A, et al. Coronary flow reserve calculated from pressure measurements in humans. Validation with positron emission tomography. Circulation 1994;89:1013‐22.
Danad I, Uusitalo V, Kero T, Saraste A, Raijmakers PG, Lammertsma AA, et al. Quantitative assessment of myocardial perfusion in the detection of significant coronary artery disease: cutoff values and diagnostic accuracy of quantitative [15O]H2O PET imaging. J Am Coll Cardiol 2014;64:1464‐75.
Rauf M, Hansen KW, Galatius S, Wiinberg N, Brinth LS, Hojstrup S, et al. Prognostic implications of myocardial perfusion imaging by 82-rubidium positron emission tomography in male and female patients with angina and no perfusion defects. Eur Heart J Cardiovasc Imaging 2023;24:212‐22.
Lortie M, Beanlands RS, Yoshinaga K, Klein R, Dasilva JN, DeKemp RA. Quantification of myocardial blood flow with 82Rb dynamic PET imaging. Eur J Nucl Med Mol Imaging 2007;34:1765‐74.
Berti V, Sciagra R, Neglia D, Pietila M, Scholte AJ, Nekolla S, et al. Segmental quantitative myocardial perfusion with PET for the detection of significant coronary artery disease in patients with stable angina. Eur J Nucl Med Mol Imaging 2016;43:1522‐9.
Nesterov SV, Deshayes E, Juarez-Orozco LE, de Kemp RE, Sciagra R, Malaspina S, et al. Myocardial perfusion quantification with Rb-82 PET: good interobserver agreement of Carimas software on global, regional, and segmental levels. Ann Nucl Med 2022;36:507‐14.
Driessen RS, van Diemen PA, Raijmakers PG, Knuuti J, Maaniitty T, Underwood SR, et al. Functional stress imaging to predict abnormal coronary fractional flow reserve: the PACIFIC 2 study. Eur Heart J 2022;43:3118‐28.
Altman DG. Some common problems in medical research. In: Practical Statistics for Medical Research. London: Chapman & Hall; 1991. p. 396‐439.
Stuijfzand WJ, Uusitalo V, Kero T, Danad I, Rijnierse MT, Saraste A, et al. Relative flow reserve derived from quantitative perfusion imaging may not outperform stress myocardial blood flow for identification of hemodynamically significant coronary artery disease. Circ Cardiovasc Imaging 2015;8:e002400.
Yoshinaga K, Manabe O, Katoh C, Chen L, Klein R, Naya M, et al. Quantitative analysis of coronary endothelial function with generator-produced 82Rb PET: comparison with 15O-labelled water PET. Eur J Nucl Med Mol Imaging 2010;37:2233‐41.
Iida H, Rhodes CG, de Silva R, Yamamoto Y, Araujo LI, Maseri A, et al. Myocardial tissue fraction–correction for partial volume effects and measure of tissue viability. J Nucl Med 1991;32:2169‐75.
Gronman M, Tarkia M, Stark C, Vahasilta T, Kiviniemi T, Lubberink M, et al. Assessment of myocardial viability with [15O]water PET: a validation study in experimental myocardial infarction. J Nucl Cardiol 2021;28:1271‐80.
Acknowledgements
Radiographer Daniel Ask Riisberg and Kathrine Marie Erenskjold are thanked for operating the GENTrace mini-cyclotron and the scanner. We are grateful for the installation of the GENTrace cyclotron by GE Healthcare and the assistance during the Covid-19 challenged time. We are very thankful to Professor Juhani Knuuti for training and reviewing our heart scan assessments the first months. Last but not least, we highly appreciate the visionary idea of a mini cyclotron at Bispebjerg Hospital, which was nurtured and negotiated by Lars Friberg.
Funding
Open access funding provided by Royal Library, Copenhagen University Library. The study did not receive external funding.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosures
Martin Krakauer, Afefah Ismail, Ulrik Talleruphuus, Alexander Cuculiza Henriksen, Markus N. Lonsdale, Inge Lise Rasmussen, Stefan Fuglsang, Eva Prescott, Peter Hovind, and Lisbeth Marner report no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The authors of this article have provided a PowerPoint file, available for download at SpringerLink, which summarises the contents of the paper and is free for re-use at meetings and presentations. Search for the article DOI on SpringerLink.com.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Krakauer, M., Ismail, A., Talleruphuus, U. et al. 82Rb and [15O]H2O myocardial perfusion PET imaging: a prospective head to head comparison. J. Nucl. Cardiol. 30, 2790–2802 (2023). https://doi.org/10.1007/s12350-023-03372-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12350-023-03372-7