Abstract
Background
An open-label, single-center, randomized controlled prospective trial was performed to assess the efficiency and safety of an insulin loading procedure to obtain high-quality cardiac 18F-FDG PET/CT images for patients with coronary artery disease (CAD).
Methods
Between November 22, 2018 and August 15, 2019, 60 patients with CAD scheduled for cardiac 18F-FDG PET/CT imaging in our department were randomly allocated in a 1:1 ratio to receive an insulin or standardized glucose loading procedure for cardiac 18F-FDG imaging. The primary outcome was the ratio of interpretable images (high-quality images defined as myocardium-to-liver ratios ≥ 1). The secondary outcome was the patient preparation time (time interval between administration of insulin/glucose and 18F-FDG injection). Hypoglycemia events were recorded.
Results
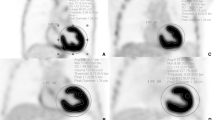
The ratio of interpretable cardiac PET images in the insulin loading group surpassed the glucose loading group (30/30 vs. 25/30, P = 0.026). Preparation time was 71±2 min shorter for the insulin loading group than for the glucose loading group (P < 0.01). Two and six hypoglycemia cases occurred in the insulin and glucose loading groups, respectively.
Conclusion
The insulin loading protocol was a quicker, more efficient, and safer preparation for gaining high-quality cardiac 18F-FDG images.



Similar content being viewed by others
Abbreviations
- CAD:
-
Coronary artery disease
- 18F-FDG:
-
F18-Fluorodeoxyglucose
- PET:
-
Positron emission tomography
- SPECT:
-
Ssingle photon emission computerized tomography
- BG:
-
Blood glucose
- GLUT4:
-
Glucose transporter 4
- VOI:
-
Volume of interest
- SUV:
-
Standardized uptake value
References
Dilsizian V, Bacharach SL, Beanlands RS, Bergmann SR, Delbeke D, Dorbala S, et al. ASNC imaging guidelines/SNMMI procedure standard for positron emission tomography (PET) nuclear cardiology procedures. J Nucl Cardiol. 2016;23:1187–226.
Panza JA, Ellis AM, Al-Khalidi HR, Holly TA, Berman DS, Oh JK, et al. Myocardial viability and long-term outcomes in ischemic cardiomyopathy. N Engl J Med. 2019;381:739–48.
Ling LF, Marwick TH, Flores DR, Jaber WA, Brunken RC, Cerqueira MD, et al. Identification of therapeutic benefit from revascularization in patients with left ventricular systolic dysfunction: inducible ischemia versus hibernating myocardium. Circ Cardiovasc Imaging. 2013;6:363–72.
Anavekar NS, Chareonthaitawee P, Narula J, Gersh BJ. Revascularization in patients with severe left ventricular dysfunction: is the assessment of viability still viable? J Am Coll Cardiol. 2016;67:2874–87.
Valentin RC, Farag A, Hage FG, Bhambhvani P. Non-diagnostic 18F-FDG PET myocardial viability studies in type-2 diabetic patients. J Nucl Cardiol. 2019;26(5):1775–6.
Gibb AA, Hill BG. Metabolic coordination of physiological and pathological cardiac remodeling. Circ Res. 2018;123:107–28.
Fazakerley DJ, Lawrence SP, Lizunov VA, Cushman SW, Holman GD. A common trafficking route for GLUT4 in cardiomyocytes in response to insulin, contraction and energy-status signalling. J Cell Sci. 2009;122:727–34.
Leto D, Saltiel AR. Regulation of glucose transport by insulin: traffic control of GLUT4. Nat Rev Mol Cell Biol. 2012;13:383–96.
Jaldin-Fincati JR, Pavarotti M, Frendo-Cumbo S, Bilan PJ, Klip A. Update on GLUT4 vesicle traffic: a cornerstone of insulin action. Trends Endocrinol Metab. 2017;28:597–611.
Zhu Y, Pereira RO, O’Neill BT, Riehle C, Ilkun O, Wende AR, et al. Cardiac PI3K-Akt impairs insulin-stimulated glucose uptake independent of mTORC1 and GLUT4 translocation. Mol Endocrinol. 2013;27:172–84.
Chareonthaitawee P, Gersh BJ, Araoz PA, Gibbons RJ. Revascularization in severe left ventricular dysfunction: the role of viability testing. J Am Coll Cardiol. 2005;46:567–74.
American Diabetes Association. 6. glycemic targets: standards of medical care in diabetes-2018. Diabetes Care. 2018;41:S55–64.
Wahl RL, Jacene H, Kasamon Y, Lodge MA. From RECIST to PERCIST: evolving considerations for PET response criteria in solid tumors. J Nucl Med. 2009;50:122S–50S.
Markendorf S, Benz DC, Messerli M, Grossmann M, Giannopoulos AA, Patriki D, et al. Value of 12-lead electrocardiogram to predict myocardial scar on FDG PET in heart failure patients. J Nucl Cardiol. 2019. https://doi.org/10.1007/s12350-019-01841-6.
Slart RH, Bax JJ, van Veldhuisen DJ, van der Wall EE, Dierckx RA, de Boer J, et al. Prediction of functional recovery after revascularization in patients with coronary artery disease and left ventricular dysfunction by gated FDG-PET. J Nucl Cardiol. 2006;13:210–9.
Fuchs TA, Ghadri JR, Stehli J, Gebhard C, Kazakauskaite E, Klaeser B, et al. Hypodense regions in unenhanced CT identify nonviable myocardium: validation versus 18F-FDG PET. Eur J Nucl Med Mol Imaging. 2012;39:1920–6.
Bax JJ, Visser FC, Poldermans D, van Lingen A, Elhendy A, Boersma E, et al. Feasibility, safety and image quality of cardiac FDG studies during hyperinsulinaemic-euglycaemic clamping. Eur J Nucl Med Mol Imaging. 2002;29:452–7.
Lizunov VA, Stenkula KG, Lisinski I, Gavrilova O, Yver DR, Chadt A, et al. Insulin stimulates fusion, but not tethering, of GLUT4 vesicles in skeletal muscle of HA-GLUT4-GFP transgenic mice. Am J Physiol Endocrinol Metab. 2012;302:E950–60.
Jezewski AJ, Larson JJ, Wysocki B, Davis PH, Wysocki T. A novel method for simulating insulin mediated GLUT4 translocation. Biotechnol Bioeng. 2014;111:2454–65.
Szablewski L. Glucose transporters in healthy heart and in cardiac disease. Int J Cardiol. 2017;230:70–5.
Vitale GD, deKemp RA, Ruddy TD, Williams K, Beanlands RS. Myocardial glucose utilization and optimization of (18)F-FDG PET imaging in patients with non-insulin-dependent diabetes mellitus, coronary artery disease, and left ventricular dysfunction. J Nucl Med. 2001;42:1730–6.
Kobylecka M, Mazurek T, Fronczewska-Wieniawska K, Fojt A, Słowikowska A, Mączewska J, et al. Assessment of the myocardial FDG-PET image quality with the use of maximal Standardized Uptake Value myocardial to background index. Application of the results in regard to semiquantitative assessment of myocardial viability with cardiac dedicated software. Nucl Med Rev Cent East Eur. 2017;20:69-75.
Schinkel AF, Bax JJ, Valkema R, Elhendy A, van Domburg RT, Vourvouri EC, et al. Effect of diabetes mellitus on myocardial 18F-FDG SPECT using acipimox for the assessment of myocardial viability. J Nucl Med. 2003;44:877–83.
Cai X, Han X, Zhou X, Zhou L, Zhang S, Ji L. Associated factors with biochemical hypoglycemia during an oral glucose tolerance test in a Chinese population. J Diabetes Res. 2017;3212814. https://doi.org/10.1155/2017/3212814
Morio R, Hyogo H, Hatooka M, Morio K, Kan H, Kobayashi T, et al. The risk of transient postprandial oxyhypoglycemia in nonalcoholic fatty liver disease. J Gastroenterol. 2017;52:253–62.
Sherwin RS, Kramer KJ, Tobin JD, Insel PA, Liljenquist JE, Berman M, et al. A model of the kinetics of insulin in man. J Clin Invest. 1974;53:1481–92.
Acknowledgements
The trial was supported by Natural Science Foundation of Fujian Province (2015J01516, 2018J01202), Quanzhou Science and Technology Commission (2019C023R), and National Natural Science Foundation of China (81601525).
Conflict of Interest
No disclosures.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Chen, Y.C., Wang, Q.Q., Wang, Y.H. et al. Intravenous regular insulin is an efficient and safe procedure for obtaining high-quality cardiac 18F-FDG PET images: an open-label, single-center, randomized controlled prospective trial. J. Nucl. Cardiol. 29, 239–247 (2022). https://doi.org/10.1007/s12350-020-02219-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12350-020-02219-9