Abstract
Introduction
Eosinophilic esophagitis (EoE) is a chronic, immune-mediated esophageal disease for which there is currently no approved treatment in the USA and only one approved therapy in Europe. EoE can significantly affect the lives of patients and caregivers; however, little has been published about patients’ experiences from symptom onset to diagnosis and treatment.
Methods
This was a cross-sectional, qualitative research study. During one-on-one semi-structured interviews, patients with EoE in the USA and their caregivers provided information about their experiences of EoE before and during diagnosis, and their current experiences. Qualitative data were analyzed using a content analysis approach.
Results
The study included children aged 6–11 years with EoE (n = 4) and their caregivers (n = 4); adolescents aged 12–17 years with EoE (n = 7) and their caregivers (n = 7); and adults aged ≥ 18 years with EoE (n = 20). The diagnosis of EoE was often arduous and took many years. Patients and caregivers were frequently frustrated with the complexities of diagnosing EoE, which often involved multiple healthcare providers and procedures. Patients reported physical and psychosocial burdens associated with EoE before diagnosis, including interference with social activities, school, and work. Patients also reported feeling frustrated or anxious. These burdens frequently remained after diagnosis. Caregivers also reported anxiety and, in some cases, interference with their ability to work.
Conclusion
This study of EoE describes the difficult journey faced by patients and their caregivers from symptom onset to diagnosis and beyond.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Why carry out this study? |
Eosinophilic esophagitis (EoE) is a chronic, immune-mediated disease, which can significantly affect the lives of patients and caregivers; however, little has been published about patients’ experiences from symptom onset to diagnosis and treatment. |
The objective of this study was therefore to improve our understanding of the clinical and humanistic burden on patients with EoE and their caregivers, and to gain insights into the patient journey. |
What was learned from the study? |
Patients with EoE and their families reported feeling frustrated by the long and frequently complicated journey to diagnosis. |
EoE affected patients and their families physically, emotionally and socially, often interfering with school and work. |
The burdens associated with EoE typically remained even after diagnosis. |
Introduction
Eosinophilic esophagitis (EoE) is an immune-mediated disease characterized by eosinophilic infiltration of the esophagus [1], which can cause chronic inflammation. If left untreated, fibrosis, the formation of strictures, and esophageal narrowing can occur, resulting in esophageal dysfunction [1]. In younger children and infants, the primary signs and symptoms of EoE include abdominal pain, failure to thrive, feeding difficulties, reflux, and vomiting [1]. In older children and adults with EoE, solid-food dysphagia, food impaction, and chest pain are more commonly observed [1]. In the USA, the prevalence of EoE has been reported to range from 7.3 (1995–2004) to 71.1 (2011–2014) per 100,000 among children [2, 3], and from 9.5 (2008–2009) to 58.9 (2009–2011) per 100,000 among adults [4, 5]. EoE is estimated to affect 13–49 individuals per 100,000 globally [1], with the prevalence having increased over recent years [6]. Despite this, there is currently no pharmacological therapy approved by the US Food and Drug Administration (FDA) for the treatment of patients with EoE, and only a single product is approved by the European Medicines Agency (budesonide oral dispersible tablet; market authorization received 8 January 2018) [7]. Patients in the USA thus use dietary modifications, as well as existing medications off-label for EoE, including topical corticosteroids and proton pump inhibitors [8, 9].
Although EoE does not appear to limit the life expectancy of patients, it can have a significant financial impact on healthcare services and patients and impair patients’ health-related quality of life (HRQoL) [10,11,12,13,14]. A US database study estimated that medical resource utilization costs for patients with EoE were significantly higher than for matched controls (median total annual cost, US$3304 vs. $1001, respectively; p < 0.001) [10]. As a result, EoE has been estimated to cost the US healthcare service $350–$947 million per year [10]. Furthermore, existing off-label pharmacologic therapies and dietary therapies are not typically covered by health insurance policies, thus increasing out-of-pocket expenses for patients with EoE in the USA [11, 12].
Only a limited number of qualitative studies have been conducted over the last decade to identify disease-specific concerns and assess the impact of EoE on patients’ HRQoL [15,16,17]. These studies identified consistent themes raised by patients and their caregivers, and included: concerns and anxiety about the disease and associated symptoms; feelings of isolation and frustration; and the negative impact the disease had on school, work, and social activities [15,16,17]. More recently, a systematic review of the literature identified only 13 studies describing the impact of EoE on HRQoL in children and adults [14]. The systematic review reported that EoE disrupted and restricted patients’ daily lives and, in some cases, the lives of their families [14]. Currently, relatively little has been published about how EoE affects patients and their families in terms of the process leading to a formal diagnosis, managing symptoms and associated triggers, and living with the disease after diagnosis.
The objectives of this cross-sectional, qualitative research study were to improve our understanding of the clinical and humanistic burden on patients with EoE and their caregivers, and to gain insights into the patient journey from symptom onset to diagnosis and beyond.
Methods
Study Design
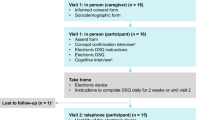
This was a cross-sectional, qualitative research study involving one-on-one, semi-structured interviews of patients with EoE and their caregivers in the USA. Caregivers were interviewed about their children with EoE (children aged 6–11 years and adolescents aged 12–17 years). In addition, caregivers were interviewed to determine the impact of an EoE diagnosis on their own HRQoL. Adolescents (aged 12–17 years) and adults (aged ≥ 18 years) with EoE were also interviewed separately. All interviews were completed by telephone. Caregivers and their adolescent children were interviewed independently of each other to ensure that both their perspectives were represented; however, a caregiver could attend the child’s interview session if preferred. Only one caregiver per patient participated in the study. This qualitative study was designed to obtain patient and caregiver perspectives on the humanistic burden of EoE, and, as such, was not powered to allow direct comparisons between groups. Furthermore, EoE is a rare condition [1], and thus responses were elicited directly from identified patients.
All procedures performed in this study were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. The study protocol and research materials were approved by an institutional review board (IRB; Ethical & Independent Review Services, Independence, MO, USA); study number 16102-01 (approved September 2, 2016). Once the central IRB review/approval was received (Ethical & Independent Review Services, Independence, MO, USA), Evidera designed, managed, collected, analyzed, and reported the study.
Study Participants
Participants were recruited by US-based patient advocacy organizations (the American Partnership for Eosinophilic Disorders (APFED) and the Campaign Urging Research for Eosinophilic Disease (CURED)) using their patient registries. Participants were recruited in the following planned cohorts: children with EoE (aged 6–11 years) and their caregivers; adolescents with EoE (aged 12–17 years) and their caregivers; and adults with EoE (aged ≥ 18 years). Patients and their caregivers were contacted by email, through promotional materials, and through social media advertisements. A purposive sampling strategy was used, with efforts made to recruit participants with diverse demographic characteristics (e.g., age and race/ethnicity), including at least 25% female participants and at least 40% male participants in each patient cohort; and to include participants from diverse backgrounds in terms of age, race, and ethnicity.
To be eligible, participants had to: have a caregiver report or self-report of a clinician-confirmed diagnosis of EoE; be able to read and speak English; be willing to participate in an audio-recorded telephone interview; and provide written informed consent (adults and caregivers) or assent (children and adolescents). Caregivers of children and adolescents with EoE also had to be aged 18 years or older, be the primary guardian, and have resided with the child for at least 6 months prior to participating in this study. Participants were excluded if they had any cognitive, visual, or hearing impairment that would interfere with participation in the study. Furthermore, only one caregiver of each child with EoE could participate in the study. Children with any clinically relevant and/or serious chronic medical conditions, which, in the opinion of the investigators, may have confounded the reporting of signs and symptoms of EoE, were excluded from this study. This included: malignancy; history of organ transplantation; congenital abnormalities; Ehler–Danlos Syndrome; vasculitis; Lupus; hypereosinophilic syndrome; other eosinophilic disorders (e.g., eosinophilic gastroenteritis); and other gastrointestinal diseases (e.g., Crohn’s disease). Concomitant gastroesophageal reflux disease (GERD), asthma, allergic rhinitis, or food allergies were not exclusionary.
Potential participants were screened by a trained qualitative researcher during a telephone call, and eligibility criteria collected using a recruitment and screening script. Eligibility data were then reviewed and confirmed by the investigation team. Participant contact information was sent via a secure, password-protected electronic document with permission from each individual and/or caregiver. Each participant received a US$100 gift certificate for their participation in the study. If a caregiver participated alone (e.g., for the cohort aged 6–11 years), only the caregiver received $100. In instances where caregivers and adolescents participated, both received $100.
Study Conduct and Data Collection
Participants were asked to complete a questionnaire before the telephone interview to help facilitate discussions. A trained staff member then conducted qualitative telephone interviews according to the study protocol using (1) the completed pre-interview questionnaire, and (2) a semi-structured qualitative interview guide. Following the interview, participants were asked to complete a sociodemographic survey. Repeat interviews were not conducted. The pre-interview questionnaire and qualitative interview guide were developed based on a targeted literature review with input from two physicians with clinical and research expertise in EoE. The questionnaire and interview guide were not previously validated or pilot-tested, but were designed specifically for this study. The research team was led by two qualified outcomes research scientists [R.M.P. (PhD, MHA; female) and G.H. (MA; female), Evidera] and supported by research associates [including A.B. (BS; female), Evidera] and other qualified qualitative researchers from Evidera. An outcomes research scientist [L.T. (MBA; female)] and a clinician [J.W. (MD; male)] from the sponsor (Shire, a member of the Takeda group of companies) helped develop and refine the study methods. Participants were aware of the interviewer’s credentials and the purpose of the study, but did not speak with the interviewer until taking part in the study. A total of five staff members performed the interviews. This study utilized standardized recruitment and screening scripts, standardized qualitative interview guides, and standardized surveys/questionnaires for each cohort in the study in order to reduce any potential interviewer bias. All interviewers were trained on how to use the interview guide and followed the same guide.
Pre-Interview Questionnaire
The pre-interview questionnaire and other appropriate documents (e.g., the consent and assent forms, where applicable) were sent to participants via post for completion ahead of the interviews, and comprised questions about the patient’s journey leading to a diagnosis of EoE, their management of the disease, and their current experiences with EoE. It included questions about the patient’s medical history, signs and symptoms [pre-diagnosis and post-diagnosis (3 months before interview)], use of pharmacological treatments and dietary modifications, and the physical and psychosocial impact of the disease (pre- and post-diagnosis). Participants were also asked about episodic experiences, steps taken to modify their lifestyle to avoid symptoms, and whether specific triggers had been identified (e.g., allergy or food intolerance). Different versions of the pre-interview questionnaire were used for caregivers, adults, and adolescents.
Participants were provided with a brief introduction to the interview process, informed that the session would be audio-recorded, and given the opportunity to ask questions before the interview.
Qualitative Interview Guide
The interview sessions captured data on symptom experience, procedure history, diagnosis, clinical milestones, and other personal impact. Each interview lasted approximately 90 min, during which interviewers collected information as notes on the data collection form. A semi-structured qualitative interview guide was used in conjunction with results from the pre-interview questionnaire. It included open-ended questions about the participant’s experiences with EoE symptoms, the pathway to diagnosis of EoE (including medical milestones), the impact of EoE on HRQoL for both the patient and caregiver (including social activity, daily activity, physical well-being, emotional well-being, and relationships), and experience with treatments. Different versions of the qualitative interview guide were used for caregivers, adults, and adolescents.
Post-Interview Sociodemographic Survey
Two different versions of the sociodemographic survey were used: one for caregivers and one for adults with EoE. Sociodemographic data for children and adolescents were collected using a caregiver version of the sociodemographic survey. The caregiver survey collected information about the caregiver and their child or adolescent with EoE, including age, sex, relationship to patient, and level of schooling. The adult survey included questions about age, sex, race, ethnicity, living situation, employment status, and educational background.
Qualitative and Quantitative Analysis
Qualitative data collected from interviewer notes and audio files were analyzed using a content analysis approach with thematic analysis in which qualitative data are organized by topic [18, 19]. Emerging concepts were tracked by interviewers in a concept tracking table. Quantitative data were summarized by cohort using descriptive statistics, including frequencies or means and standard deviations, in Microsoft Excel.
Results
Study Participants
Overall, 65 individuals responded to the advertisement; of these, 25 were not screened due to difficulties with contacting them or the study recruitment quotas being reached. Of the 40 participants who were screened, two were not US residents and were thus not eligible for inclusion in this study (Supplementary Fig. S1 in the ESM). Thirty-eight participants were enrolled from 22 September to 28 November 2016 and completed the qualitative interviews (flow diagram presented in Supplementary Fig. S1 in the ESM). This included caregivers of children with EoE aged 6–11 years (n = 4; 75.0% of the children were male), adolescents with EoE aged 12–17 years (n = 7; 42.9% male) and their caregivers (n = 7), and adults with EoE aged ≥ 18 years (n = 20; 40.0% male). All 11 caregivers (for children, n = 4; and for adolescents, n = 7) were female and married (Table 1), and all were White (not Hispanic or Latino). The mean [standard deviation (SD)] age at first signs and symptoms for children, adolescents, and adults with EoE was 0.6 (1.0), 3.8 (4.0), and 17.2 (12.7) years, respectively. The mean (SD) age at diagnosis for these groups was 4.9 (3.9), 8.1 (4.1), and 27.3 (11.3) years, respectively (Table 1).
Pre-Diagnosis Signs and Symptoms and Burden of EoE
Signs and symptoms before diagnosis varied by cohort (Fig. 1). Pre-diagnosis signs and symptoms for children and adolescents were based on caregiver report. For children with EoE (n = 4; caregiver report), the most commonly reported signs and symptoms pre-diagnosis were: abdominal pain (100.0%); avoiding or refusing food; chest pain; regurgitation/reflux/GERD; and vomiting (each reported by 75.0% of caregivers). These findings were similar for adolescents with EoE (n = 7; caregiver report), with the most common signs and symptoms before diagnosis being: abdominal pain (100.0%); avoiding or refusing food; regurgitation/reflux/GERD; and vomiting (each reported by 57.1% of caregivers). In contrast, adults (n = 20; self-report) with EoE experienced a broader range of signs and symptoms before diagnosis, but the most common were: difficulty or discomfort swallowing solid foods and purees (90.0%); chest pain; feeling an obstruction in the throat (globus sensation); and odynophagia (each reported by 75.0% of adults). Furthermore, only adults with EoE reported globus sensation (75.0%); and food impaction/obstruction (60.0%).

Child and adolescent information reported is based on caregiver report. More than one sign or symptom could have been selected for each patient. aThree children had other symptoms, which were: cessation of linear growth (n = 1); coughing and sneezing (n = 1); eating slowly (n = 1); headaches and stomachaches (n = 1); and pain in legs (n = 1). bFour adolescents had other symptoms, which were: constipation (n = 1); dizziness (n = 1); difficulty sleeping (n = 2); eczema (n = 2); rashes/hives (n = 1); and pain in legs (n = 1). cFive adults reported other symptoms, which were: weight loss (n = 2); slow eating (n = 1); joint pain (n = 1); ulcers (n = 1); and “food-poisoning symptoms” (n = 1). EoE eosinophilic esophagitis, GERD gastroesophageal reflux disease
Signs and symptoms of EoE prior to diagnosisa–c.
Caregivers and adults with EoE were also asked about the physical and psychosocial impact of EoE before diagnosis (Table 2). Caregivers (n = 11) most commonly reported feeling fatigued/tired (72.7%); difficulties with completing daily activities at home, work, or school (63.6%); difficulties with participating in hobbies or leisure activities (72.7%); and spending more time to care for their child with EoE versus their other children (90.9%). Caregivers also commonly reported feeling frustrated/irritated/moody (81.8%); concerned/worried/anxious (100.0%); and out of control/helpless (81.8%). Caregivers reported that their children (n = 4) most commonly reported fatigue/tiredness (50.0%); difficulties participating in social activities with others (75.0%); feeling frustrated/irritated/moody (75.0%); and feeling like their life was centered or revolved around EoE (75.0%). Caregivers reported that their adolescent children (n = 7) most commonly experienced a reduced appetite/ability to eat (85.7%); fatigue/tiredness (71.4%); and weakness/reduced energy levels (71.4%). Caregivers also indicated that their adolescent children most commonly reported difficulties participating in activities for fun and enjoyment (85.7%); difficulties with participating in social activities with others (57.1%); and an impact on their relationships with family, friends, or other children (71.4%). Furthermore, all adolescents reported feeling frustrated/anxious/moody (100.0%); feeling concerned/worried/anxious as a result of having EoE was also common (71.4%). Similarly, adults with EoE (n = 20) most commonly reported fatigue/tiredness (70.0%); reduced appetite/reduced ability to eat (75.0%); and weakness/reduced energy level (70.0%). Adults also most commonly reported difficulties participating in social activities with others (75.0%); and impact on relationships with family, friends, or other children (65.0%). Adults, like caregivers and adolescents with EoE, reported feeling concerned/worried/anxious (90.0%), but also reported feeling embarrassed by their condition (75.0%).
EoE was reported to have an impact on adult patients’ and caregivers’ work productivity. Caregivers of children and adolescents with EoE and adults with EoE reported that they had to stop working [caregivers, 45.5% (5/11); adults, 20.0% (4/20)], or had to reduce the number of hours they worked owing to EoE [caregivers, 27.3% (3/11); adults, 15.0% (3/20)]. Two adults with EoE also had to work extra hours or more than one job to cover the costs of living with EoE (Supplementary Table S1 in the ESM).
Reaching a Formal Diagnosis of EoE
For most participants, a formal diagnosis of EoE was obtained after consultation with a range of healthcare providers. This usually included a gastroenterologist or pediatric gastroenterologist, an allergist/immunologist, and a primary healthcare provider (Fig. 2a). Participants reported that they had undergone a range of procedures prior to receiving a diagnosis of EoE; all participants (children, adolescents, and adults) had undergone an upper endoscopy with biopsy (Fig. 2b). For children, adolescents, and adults with EoE, there was a mean (range) delay of 4.3 (0.9–9.7) years, 4.3 (1.0–6.9) years, and 11.1 (0.0–34.0) years, respectively, in time from onset of first signs and symptoms of EoE to diagnosis. The burden of reaching a formal diagnosis of EoE was described as “a great deal” or “a very great deal” for 75.0% of children (3/4), 71.4% of adolescents (5/7), and 70.0% of adults (14/20) with EoE (Fig. 2c). Qualitative feedback was similar across cohorts, including reports of being incorrectly diagnosed, feeling frustrated with healthcare professionals because they could not provide a diagnosis, stress related to obtaining a diagnosis, difficulty in pinpointing the reason for signs and symptoms, and having to attend multiple doctor visits to obtain a diagnosis (data not shown).

Child and adolescent information reported is based on caregiver report. More than one category could have been selected for each patient. aOther healthcare providers for two adolescents with EoE were: “Naturopath” doctor (n = 1; 2 visits); and integrative medical doctor (n = 1; 10 visits). Other healthcare providers for adults with EoE were: chiropractor/ “energy” doctor (n = 1); cardiologist (n = 1); ear, nose, and throat specialist (n = 1); and neurologist (n = 2). bOther procedures for two adolescents with EoE were: colonoscopy (n = 2); and lactose intolerance test (n = 1). Other procedures for adults with EoE were: esophageal manometry test (n = 1); and pH probe study (n = 1). EoE eosinophilic esophagitis, ER emergency room
Burden of obtaining a formal diagnosis of EoE, as measured by a the range of healthcare providers visited before reaching a diagnosis;a b the range of procedures that occurred before reaching a diagnosis;b and c the burden related to obtaining a formal diagnosis of EoE as rated by the caregiver or patient.
Post-Diagnosis Signs and Symptoms and Burden of EoE Reported During the 3 Months Before Interview
Signs and symptoms of EoE persisted for all cohorts even after diagnosis (Fig. 3a, b), the caregiver and self-report for adolescents were comparable (Fig. 3b), and EoE continued to have a physical and psychosocial impact on all patient cohorts after diagnosis (Table 3). Patients and caregivers also reported worries about the cost of EoE, in terms of expensive dietary supplements or dietary modifications and travel expenses that were not covered by their health insurance policies. Patients and caregivers highlighted the cost of their monthly or yearly deductibles on their insurance policies; some participants also reported worries about their lack of insurance coverage (data not shown).

More than one response option could have been selected for each patient. aTwo children (caregiver report) had other symptoms, which were: slow linear growth (n = 1); pain in throat (n = 1); headaches (n = 1); and leg pain (n = 1). bOne adolescent (caregiver report) had excessive throat clearing (n = 1). cFour adults reported (self-report): anxiety (n = 1); esophageal spasms (n = 1); pain in throat (n = 1); joint pain (n = 1); small intestine dysmotility (n = 1); and digestion problems (n = 1). dOne adolescent reported (self-report) dizziness (n = 1). EoE eosinophilic esophagitis, GERD gastroesophageal reflux disease
Post-diagnosis signs and symptoms reported in the 3 months before interview for a children (caregiver report), adolescents (caregiver report), and adults (self-report) with EoE, and b comparison between the caregiver report and self-report for adolescents with EoEa–d.
Triggers of EoE and Trigger Avoidance
In the 3 months before interview, the most common triggers for children with EoE (n = 4) were food triggers (100.0%) and environmental allergens (50.0%). Similarly, common triggers for adolescents (caregiver report) with EoE (n = 7), were: food triggers (100.0%) and environmental allergens (42.9%); stress was also reported (42.9%). Adolescents (self-report) additionally identified infection as a common trigger (57.1%). For adults with EoE (n = 20), food triggers (80.0%) and stress (50.0%) were reported as their most common triggers (Supplementary Table S2 in the ESM). Triggers could be avoided by the majority of patients [children, 100.0% (4/4); adolescents, 71.4% (5/7) according to caregivers and adolescents themselves; adults, 60.0% (12/20)], although most patients also had times when EoE triggers could not be avoided [children, 75.0% (3/4); adolescents, 71.4% (5/7) according to caregivers and adolescents themselves; adults, 75.0% (15/20)] (Supplementary Table S2 in the ESM).
Medications and Dietary Therapy/Modification for EoE
The majority of participants (all cohorts) were receiving medication for the treatment of EoE [children, 100.0% (4/4); adolescents, 100.0% (7/7); adults, 70.0% (14/20)]. For all cohorts, the most common medications used were proton pump inhibitors and orally administered topical corticosteroids (e.g., administered as a slurry) (Supplementary Table S3 in the ESM). Rates of dietary elimination therapy were similarly high [children, 50.0% (2/4); adolescents, 71.4% (5/7); adults, 80.0% (16/20)]. The most common was an empiric 6-food elimination diet (soy, egg, dairy products, wheat, nuts, and seafood) (Supplementary Table S4 in the ESM). Some participants were on a combination of pharmacologic therapy and dietary modification [children, 50.0% (2/4); adolescents, 71.4% (5/7)]; and adults, 55.0% (11/20)].
Discussion
Since clinical guidelines on EoE were first established in 2007 [20], the treatment and understanding of this disease have advanced rapidly [1, 8, 21]. However, there is limited information about the journey patients and caregivers face during the progression from symptom onset to diagnosis and treatment. In this cross-sectional, qualitative research study of caregivers, children, adolescents, and adults with EoE, we report that EoE not only had a physical impact but often also a significant psychosocial impact on patients and caregivers. Obtaining a formal diagnosis of EoE from onset of first signs or symptoms was often arduous, with a mean time to diagnosis ranging from 4.3 to 11.1 years. For most patients with EoE, a formal diagnosis was obtained after consultation with a range of healthcare providers and after several different procedures, including invasive techniques such as upper endoscopy with biopsy. The majority of participants subsequently reported the process of obtaining a formal diagnosis of EoE as burdensome, highlighting the need to streamline the diagnostic process for this disease. These delays likely contributed in part to the feelings of frustration experienced by patients and caregivers.
Patients found it difficult to participate in social activities or hobbies, and the disease could also interfere with school or work, and time spent with family and friends. Patients also reported often feeling frustrated or anxious, and reported fatigue/tiredness and reduced energy levels. Physical and psychosocial impacts of EoE, especially disruption of school and work and psychological problems, remained even after diagnosis and treatment, highlighting the unmet need of patients with EoE.
Signs and symptoms of EoE reported in this study were generally consistent across age cohorts, but adults reported more severe signs and symptoms, including food impaction/obstruction. These differences between the symptom profiles of younger children and infants compared with adolescents and adults have been previously reported [1] and could be indicative of disease progression. This finding was also mirrored in the “Severity of EoE” and “Overall health” ratings reported by caregivers of children and adolescents with EoE and adult patients with EoE. Many signs and symptoms persisted after diagnosis, despite the majority of patients receiving either medication or dietary therapy, likely owing to the persistent nature of EoE [22], and potentially a lack of effective medications for treating the disease. As a result, patients required multiple medications, including corticosteroids and proton-pump inhibitors, as well as immunotherapies (all currently used off-label [11]), and complex, restrictive dietary modifications to manage the disease. The cost of dietary supplements or modifications was also discussed as a concern by patients and caregivers, particularly as these may not be covered by their health insurance policies. Furthermore, patients may experience difficulties in obtaining insurance coverage for medications used off-label [11]. To help improve this situation, in 2017, the American Gastroenterological Association Center for Diagnostics and Therapeutics released a white paper on advancing drug development for EoE, which included recommendations for industry on appropriate diagnostic criteria, and endpoints for different patient populations in clinical trials [11].
Although EoE is regarded as not impacting life expectancy, it has been reported to significantly affect patients’ HRQoL and that of their caregivers and families [14,15,16,17]. However, only a limited number of HRQoL studies have been conducted in EoE, exemplified by the fact that only 13 studies were included in a recent systematic review of the literature [14]. Despite this, a number of qualitative studies highlighted that EoE has an impact on the psychosocial wellbeing and daily lives of patients [15, 17]. Consistent themes reported here and in the literature include feelings of anger and frustration, irritability, and moodiness among patients with EoE, and that the disease makes it difficult to participate in social activities [15, 17]. An important additional finding of this study was that EoE also strongly affected the psychosocial function of caregivers. Before a diagnosis of EoE, caregivers reported feeling anxious, irritated, frustrated, and helpless about their child’s condition, and that their lives centered around it. Indeed, in some cases, caregivers had stopped working or were working less because of their child’s condition. For caregivers, reaching a diagnosis helped; however, they continued to be anxious about their child’s well-being and found the process of reaching a diagnosis frustrating. The diagnostic burden associated with EoE has been recently highlighted in a case report describing a 34-year-old woman who experienced progressively worsening intermittent dysphagia (in the absence of other signs and symptoms) for 2 years before being referred for an esophagogastroduodenoscopy (EGD). The EGD identified furrows and rings in the esophagus, which prompted the collection of four biopsies from the proximal and distal esophagus. A diagnosis of EoE was confirmed based on histological examination [23].
A strength of this study was that it employed a questionnaire developed with clinician and patient-reported outcomes researcher input (based on a systematic review of the literature) that allowed all patient and caregiver experiences related to signs, symptoms, and impact of EoE to be fully captured. As noted in the recent US FDA draft guidance for EoE drug development, there are no widely available patient-reported or observer-reported outcome instruments specific to EoE [24]. Future research studies could therefore explore the fit of disease-specific tools [25,26,27] or more generic HRQoL measures [28, 29] for use in EoE, so that the sample could be compared with other studies and the burden of EoE better defined compared with more common conditions.
Limitations of this study were the relatively small sample size, which could have affected the precision of the estimates; the studied population may not be representative of the overall population of patients with EoE in the USA; the time lag between events and interviews may have impacted patient and caregiver recall; and that EoE diagnosis was based on patient or caregiver report. In addition, the adult and adolescent subgroups of this study had a greater percentage of females than would be expected for this male predominant disease [6]; furthermore, the study did not include single parent or male caregivers, and the proportion of patients currently on dietary therapy in this study may not be representative of the general EoE population. These differences between the study population and the overall EoE population in the USA are likely because patients in this study were recruited via patient advocacy groups, and likely represent a more actively engaged and supported population. The limitation of caregiver report or self-report of an EoE diagnosis was mitigated by recruitment through patient advocacy groups.
Conclusion
In conclusion, this qualitative study describes the difficult journey faced by children, adolescents, and adults with EoE and the impact on their caregivers, from the onset of signs and symptoms to diagnosis and beyond. This includes difficulties dealing with not only physical signs and symptoms, the burdens of the diagnostic process, and ongoing care of EoE, but also managing personal, social, and family difficulties that are associated with the disease. Knowledge of the patient and caregiver experience is important for improving the diagnostic and treatment journey of patients and caregivers who are impacted by EoE.
References
Lucendo AJ, Molina-Infante J, Arias A, von Arnim U, Bredenoord AJ, Bussmann C, et al. Guidelines on eosinophilic esophagitis: evidence-based statements and recommendations for diagnosis and management in children and adults. United European Gastroenterol J. 2017;5:335–58.
Gill R, Durst P, Rewalt M, Elitsur Y. Eosinophilic esophagitis disease in children from West Virginia: a review of the last decade (1995–2004). Am J Gastroenterol. 2007;102:2281–5.
Maradey-Romero C, Prakash R, Lewis S, Perzynski A, Fass R. The 2011–2014 prevalence of eosinophilic oesophagitis in the elderly amongst 10 million patients in the United States. Aliment Pharmacol Ther. 2015;41:1016–22.
Dellon ES, Jensen ET, Martin CF, Shaheen NJ, Kappelman MD. Prevalence of eosinophilic esophagitis in the United States. Clin Gastroenterol Hepatol. 2014;12:589–96.
Ally MR, Maydonovitch CL, Betteridge JD, Veerappan GR, Moawad FJ. Prevalence of eosinophilic esophagitis in a United States military health-care population. Dis Esophagus. 2015;28:505–11.
Shaheen NJ, Mukkada V, Eichinger CS, Schofield H, Todorova L, Falk GW. Natural history of eosinophilic esophagitis: a systematic review of epidemiology and disease course. Dis Esophagus. 2018. https://doi.org/10.1093/dote/doy015.
European Medicines Agency. EPAR summary for the public. Jorveza (budesonide). https://www.ema.europa.eu/documents/overview/jorveza-epar-summary-public_en.pdf. Accessed 7 Apr 2020.
Dellon ES, Liacouras CA, Molina-Infante J, Furuta GT, Spergel JM, Zevit N, et al. Updated international consensus diagnostic criteria for eosinophilic esophagitis: proceedings of the AGREE conference. Gastroenterology. 2018;155:1022–33.
Hirano I, Chan ES, Rank MA, Sharaf RN, Stollman NH, Stukus DR, et al. AGA institute and the joint task force on allergy-immunology practice parameters clinical guidelines for the management of eosinophilic esophagitis. Gastroenterology. 2020;158:1776–86.
Jensen ET, Kappelman MD, Martin CF, Dellon ES. Health-care utilization, costs, and the burden of disease related to eosinophilic esophagitis in the United States. Am J Gastroenterol. 2015;110:626–32.
Hirano I, Spechler S, Furuta G, Dellon ES. White paper AGA: drug development for eosinophilic esophagitis. Clin Gastroenterol Hepatol. 2017;15:1173–83.
Gonsalves N. Dietary therapy for eosinophilic esophagitis. Gastroenterol Hepatol. 2015;11:267–76.
Leiman DA, Kochar B, Posner S, Fan C, Patel A, Shaheen O, et al. A diagnosis of eosinophilic esophagitis is associated with increased life insurance premiums. Dis Esophagus. 2018;31:1–5.
Mukkada V, Falk GW, Eichinger CS, King D, Todorova L, Shaheen NJ. Health-related quality of life and costs associated with eosinophilic esophagitis: a systematic review. Clin Gastroenterol Hepatol. 2018;16:495–503.
Taft TH, Kern E, Keefer L, Burstein D, Hirano I. Qualitative assessment of patient-reported outcomes in adults with eosinophilic esophagitis. J Clin Gastroenterol. 2011;45:769–74.
Franciosi JP, Hommel KA, DeBrosse CW, Greenberg AB, Greenler AJ, Abonia JP, et al. Quality of life in paediatric eosinophilic oesophagitis: what is important to patients? Child Care Health Dev. 2012;38:477–83.
Flood EM, Beusterien KM, Amonkar MM, Jurgensen CH, Dewit OE, Kahl LP, et al. Patient and caregiver perspective on pediatric eosinophilic esophagitis and newly developed symptom questionnaires*. Curr Med Res Opin. 2008;24:3369–81.
Braun V, Clarke V. Using thematic analysis in psychology. Qual Res Psychol. 2006;3:77–101.
Patrick DL, Burke LB, Gwaltney CJ, Leidy NK, Martin ML, Molsen E, et al. Content validity—establishing and reporting the evidence in newly developed patient-reported outcomes (PRO) instruments for medical product evaluation: ISPOR PRO good research practices task force report: part 1—eliciting concepts for a new PRO instrument. Value Health. 2011;14:967–77.
Furuta GT, Liacouras CA, Collins MH, Gupta SK, Justinich C, Putnam PE, et al. Eosinophilic esophagitis in children and adults: a systematic review and consensus recommendations for diagnosis and treatment. Gastroenterology. 2007;133:1342–63.
Liacouras CA, Furuta GT, Hirano I, Atkins D, Attwood SE, Bonis PA, et al. Eosinophilic esophagitis: updated consensus recommendations for children and adults. J Allergy Clin Immunol. 2011;128:3–20.
Straumann A. The natural history and complications of eosinophilic esophagitis. Gastrointest Endosc Clin N Am. 2008;18:99–118.
Posner S, Boyd A, Patel A. Dysphagia in a 34-year-old woman. JAMA. 2020;323:660–1.
U.S. Department of Health and Human Services. Food and Drug Administration. Center for Drug Evaluation and Research (CDER). Eosinophilic esophagitis: developing drugs for treatment. Guidance for industry. DRAFT GUIDANCE. https://www.regulations.gov/document?D=FDA-2019-D-0177–0002. Accessed 30 Jan 2020.
Franciosi JP, Hommel KA, Bendo CB, King EC, Collins MH, Eby MD, et al. PedsQL eosinophilic esophagitis module: feasibility, reliability, and validity. J Pediatr Gastroenterol Nutr. 2013;57:57–66.
Taft TH, Kern E, Kwiatek MA, Hirano I, Gonsalves N, Keefer L. The adult eosinophilic oesophagitis quality of life questionnaire: a new measure of health-related quality of life. Aliment Pharmacol Ther. 2011;34:790–8.
Hudgens S, Evans C, Philips E, Hill M. Psychometric validation of the Dysphagia Symptom Questionnaire in patients with eosinophilic esophagitis treated with oral budesonide suspension. J Patient Rep Outcomes. 2017;1:3.
Varni JW, Seid M, Kurtin PS. PedsQL 4.0: reliability and validity of the Pediatric Quality of Life Inventory version 4.0 generic core scales in healthy and patient populations. Med Care. 2001;39:800–12.
Brazier JE, Harper R, Jones NM, O'Cathain A, Thomas KJ, Usherwood T, et al. Validating the SF-36 health survey questionnaire: new outcome measure for primary care. BMJ. 1992;305:160–4.
Acknowledgements
The authors would like to thank the participants of this study, and also Lora Todorova for her involvement in the design of the study.
Funding
The study was funded by Shire International GmbH, a member of the Takeda group of companies. The study sponsor also funded the journal’s Rapid Service and Open Access fees.
Medical Writing Assistance
Medical writing and editorial assistance were provided by Phillip Leventhal, PhD of Evidera, and Luci Witcomb, PhD of PharmaGenesis London, London, UK, funded by Shire International GmbH, a member of the Takeda group of companies.
Authorship
All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article, take responsibility for the integrity of the work as a whole, and have given their approval for this version to be published.
Authorship Contributions
RMP, GH, and JW participated in designing and conducting the study, in the interpretation of results, and drafting the manuscript. AB participated in conducting the study, analyzing and interpreting the results, and drafting the manuscript. BG participated in the interpretation of results and drafting the manuscript. All authors provided substantive comments, approved the final version of the manuscript for submission, and agreed to be accountable for its contents.
Prior Presentation
This study was accepted as an abstract: Pokrzywinski R et al. Am J Gastroenterol 2018;113:S195 (https://journals.lww.com/ajg/Fulltext/2018/10001/Documenting_the_Journey_of_Patients_with.345.aspx). This study was presented as a poster at the American College of Gastroenterology (ACG) Annual Scientific Meeting, October 5–10, 2018, Philadelphia, PA, USA. Poster title: “Documenting the journey of patients with eosinophilic esophagitis and its impact on their caregivers.”
Disclosures
Robin M. Pokrzywinski, Gale Harding, Anne Brooks are an employee of Evidera. James Williams is an employee of Shire, a member of the Takeda group of companies and a stockholder of Takeda.
Bridgett Goodwin is an employee of Shire, a member of the Takeda group of companies and a stockholder of Takeda. Shire, a member of the Takeda group of companies, is currently developing budesonide oral suspension (BOS), a topical corticosteroid formulation designed specifically for patients with eosinophilic esophagitis (EoE), and is funding the phase 3 research program for BOS (ClinicalTrials.gov identifiers: NCT02605837; NCT02736409 and NCT03245840).
Compliance with Ethics Guidelines
All procedures performed in this study were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. The study protocol and research materials were approved by an IRB (Ethical & Independent Review Services, Independence, MO, USA); study number 16102-01 (approved September 2, 2016). Once the central IRB review/approval was received (Ethical & Independent Review Services, Independence, MO, USA), Evidera designed, managed, collected, analyzed, and reported the study. Informed consent was obtained from all individual participants included in the study; children signed informed assent forms.
Data Availability
The datasets aggregated and/or analyzed during the current study are available from the corresponding authors in de-identified form on reasonable request.
Author information
Authors and Affiliations
Corresponding author
Additional information
Digital Features
To view digital features for this article go to https://doi.org/10.6084/m9.figshare.12746657.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Pokrzywinski, R.M., Harding, G., Brooks, A. et al. Documenting the Journey of Patients with Eosinophilic Esophagitis and the Impact of the Disease on Patients and their Caregivers: A Cross-sectional, Qualitative Research Study. Adv Ther 37, 4458–4478 (2020). https://doi.org/10.1007/s12325-020-01463-2
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12325-020-01463-2