Abstract
Introduction
Surgical site infection (SSI) following spinal surgery is a major source of postoperative morbidity. Although studies have demonstrated perioperative antimicrobial prophylaxis (AMP) to be beneficial in the prevention of SSI among spinal surgery patients, consensus is lacking over whether preoperative or extended postoperative AMP is most efficacious. To date, no meta-analysis has investigated the comparative efficacy of these two temporally variable AMP protocols in spinal surgery. We undertook a systemic review and meta-analysis to determine whether extended postoperative AMP is associated with a difference in the rate of SSI occurrence among adult patients undergoing spinal surgery.
Methods
Embase and MEDLINE databases were systematically searched for clinical trials and cohort studies directly comparing SSI rates among adult spinal surgery patients receiving either preoperative or extended postoperative AMP. Quality of evidence of the overall study population was evaluated using the Grading of Recommendations Assessment, Development and Evaluation (GRADE) Working Group approach. Random effects meta-analyses were performed utilizing both pooled and stratified data based on instrumentation use.
Results
Five studies met inclusion criteria. No individual study demonstrated a significant difference in the rate of SSI occurrence between preoperative and extended postoperative AMP protocols. The GRADE quality of evidence was low. Among the overall cohort of 2824 patients, 96% underwent lumbar spinal surgery. Pooled SSI rates were 1.38% (26/1887) for patients receiving extended postoperative AMP and 1.28% (12/937) for patients only receiving preoperative AMP. The risk of SSI development among patients receiving extended postoperative AMP was not significantly different from the risk of SSI development among patients only receiving preoperative AMP [RR (risk ratio), 1.11; 95% CI (confidence interval) 0.53–2.36; p = 0.78]. The difference in risk of SSI development when comparing extended postoperative AMP to preoperative AMP was also not significant for both instrumented (RR, 0.92; 95% CI 0.15–5.75; p = 0.93) and non-instrumented spinal surgery (RR, 1.25; 95% CI 0.49–3.17; p = 0.65). There was no evidence of heterogeneity of treatment effects for all meta-analyses.
Conclusion
Preoperative AMP appears to provide equivalent protection against SSI development when compared to extended postoperative AMP. Prudent antibiotic use is also known to decrease hospital length of stay, healthcare expenditure, and risk of complications. However, until higher-quality evidence becomes available regarding AMP in spinal surgery, surgeons should continue to exercise discretion and clinical judgment when weighing the effects of patient comorbidities and complications before determining the optimal duration of perioperative AMP.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Surgical site infection (SSI) following spinal surgery is a major source of operative morbidity |
Studies have demonstrated perioperative antimicrobial prophylaxis (AMP) to be beneficial in the prevention of SSI among spinal surgery patients |
However, consensus is lacking over whether preoperative or extended postoperative AMP is most efficacious |
Our meta-analysis investigated whether extended postoperative AMP is associated with a difference in the rate of SSI occurrence compared to preoperative AMP among adult patients undergoing spinal surgery |
Preoperative AMP appears to provide equivalent protection against SSI development when compared to extended postoperative AMP in patients undergoing spine surgery |
Introduction
Surgical site infection (SSI) following spinal surgery is a major source of operative morbidity. Its incidence ranges from 0.22% to 16.4%, demonstrating substantial variation conditional upon numerous patient, disease, and perioperative factors [1,2,3,4,5,6,7,8,9,10,11,12,13,14,15,16,17,18,19]. Treatment requires prolonged antibiotic therapy and often requires reoperation. Although perioperative antimicrobial prophylaxis (AMP) has proven effective in the prevention of SSI following spinal surgery [18, 19], uncertainty remains regarding its optimal duration.
Preoperative and extended postoperative AMP have been shown to be clinically equivalent in major surgery [20, 21]. Accordingly, the Centers for Disease Control and Prevention (CDC) recommends maintaining AMP throughout the operation until, at most, a few hours after incision closure [22]. Although no spinal surgery study to date has presented significant evidence that extended postoperative AMP is superior to preoperative AMP, spinal surgeons still hesitate to assimilate this evidence in the provision of care [22,23,24,25,26,27,28,29,30,31] and often continue AMP into the postoperative period [32, 33].
The rationale for this skepticism remains unclear, but proposed explanations suggest that the relative quality and number of available studies investigating the efficacy of temporally variable perioperative AMP protocols in spinal surgery have left surgeons reluctant to generalize broad results to their subspecialty before additional high-quality evidence becomes available [27, 28]. Another possible explanation could come from disagreement among major surgery guidelines, discrepancies between subspecialty practices [34,35,36], and the lack of recommendations specific to spinal surgery. In particular, performance measures endorsed by the Centers for Medicare and Medicaid Services are contingent upon discontinuation of AMP within 24 h after the end of major surgery [36,37,38,39]. Meanwhile, only recently have formalized guidelines specific to spinal surgery been provided by the North American Spine Society (NASS), suggesting that AMP be discontinued with intraoperative dosing for adult patients undergoing typical, uncomplicated spinal surgery [23,24,25].
If extended postoperative AMP does not provide a concomitant benefit in the prevention of SSI after spinal surgery, continuation of AMP postoperatively may unnecessarily expose patients to adverse drug reactions, additional costs, and prolonged hospital stays while simultaneously promoting development of antimicrobial resistant bacterial (AMR) strains [19, 26] and Clostridium difficile infection. Therefore, this systematic review and meta-analysis aims to assess the comparative efficacy of extended postoperative AMP versus preoperative AMP in the prevention of SSI among adult patients undergoing spinal surgery.
Methods
Study Selection
We conducted a systematic review of Embase and MEDLINE databases through February 1, 2019. The study selection process was tailored to identify published randomized controlled trials (RCT) and cohort studies comparing the relative efficacy of preoperative and extended postoperative AMP protocols in the prevention of SSI among adult patients undergoing spinal surgery. Only those studies that used SSI occurrence as their primary outcome of interest were considered. Our search terms included “spine surgery” or “spine injury” or “foraminotomy” or “laminectomy” or “spine fusion” or “antibacterial agent” and “antibiotics”, or “cefazolin” or “clindamycin” or “cefuroxime” or “vancomycin” and “postoperative period” or “perioperative care” or “postoperative care” or “intraoperative period” or “preoperative period” or “intraoperative period”. Supplemental Table 1 provides more details on the search methodology in the present investigation. Since identification of SSI involves interpretation of clinical and laboratory findings, included studies were required to provide evidence that diagnosis was at least partially commensurate with current CDC guidelines to ensure consistency [22, 40]. Given that retrospective studies were included in this review, the only parameter that was afforded exception in meeting SSI defining criteria was the length of the observational window in which diagnosis could be made. Postoperative systemic infections and remote coexisting and complicating infections did not meet SSI criteria [40] and were not included. This method of study selection is similar to that implemented by McDonald et al. [20]. Secondary outcomes included the incisional type of SSI and the microbiology and resistance patterns of positive wound cultures.
Included studies needed to directly compare rates of spinal SSI between preoperative and extended postoperative AMP protocols. Studies that compared one perioperative AMP protocol with placebo and those that solely assessed variations of extended postoperative AMP were excluded. Studies contrasting different treatment arms of preoperative and extended postoperative AMP regimens were included but were manipulated a priori in this analysis so that there was only one preoperative AMP group, one extended postoperative AMP group, and one risk ratio (RR) for each study. Pharmacokinetic studies evaluating the comparative efficacy and role of two or more different antibiotics in spinal SSI prevention were excluded. There were no inclusion criteria conditional upon the dosage and type of antibiotics administered, and perioperative changes made to these parameters for clinical reasons were permitted.
The different AMP regimens utilized by included studies were obligated to fulfill specific criteria. Preoperative AMP regimens required administration of a single antibiotic dose prior to surgery and allowed for intraoperative redosing; however, antibiotic administration at the conclusion of the procedure or any time postoperatively was not permitted [20]. Meanwhile, criteria for extended postoperative AMP regimens were identical to those for preoperative AMP protocols, but also allowed for continued antibiotic administration into the postoperative period for an indefinite length of time. Intraoperative redosing and its timing for both prophylactic regimens needed to be contingent upon several factors, including operative duration; half-life of the antimicrobial agents administered; patient characteristics such as renal function, body habitus, and age; amount of blood loss and volume of subsequent transfusions and fluid therapy; and rate of diffusion into the wound, in order to maintain bactericidal concentrations of the antibiotic [20, 22, 30, 41,42,43]. The terms preoperative and extended postoperative AMP were used in this study instead of single-dose and multiple-dose prophylaxis in an attempt to avoid confusion. Single-dose prophylaxis can, by convention, include multiple doses of antibiotics if administered intraoperatively [20], and can easily be misinterpreted as AMP characterized by multiple doses.
Included studies were required to have sample sizes greater than or equal to 20 patients with at least 10 patients in each treatment arm. Case–control studies were also excluded to avoid including a potentially large source of bias. All studies that were not accessible in English were excluded.
There were no specific exclusion criteria placed upon adult age and comorbidity status since this would eliminate the majority of studies without access to individual patient data for pooled analysis. However, studies primarily assessing spinal SSI among pediatric patient populations were excluded. There were also no exclusion criteria surrounding details of the spinal operation performed, including the type of procedure and its elective or emergent nature, surgical complexity and duration, instrumentation use, anatomic level of surgery and whether surgeons operated at multiple spinal levels, revision surgery, and use of surgical site irrigation with antibiotics.
Data Extraction
Using a standardized data extraction form, information was independently collected by two teams of investigators. Discrepancies were resolved for each team by a dedicated third party through review of original articles and group discussions. Information collected from each study included the study period, overall sample size and the number of patients in preoperative and extended postoperative AMP treatment arms, percentage of male patients, descriptive statistics for cohort age, patient comorbidities, antibiotics administered and their perioperative timing and duration, anatomic level and type of spinal surgery, instrumentation status, number of SSIs along with their incisional type and microbiology, comparable SSI prevalence estimates, and sources of confounding. The antibiotics recorded were the predominant form of pharmacologic therapy administered in each study. Antibiotics utilized for exceptional cases, such as patients with penicillin or cephalosporin allergies, were not documented.
Information regarding quality was also extracted and summarized for individual studies and for the combined study population as a whole. Included RCTs were determined to have a high, unclear, or low risk of bias through use of the Cochrane Collaboration’s tool for assessing risk of bias. Meanwhile, the quality of included cohort studies was assessed using the Newcastle–Ottawa Scale (NOS) in which they could receive anywhere from 0 to 9 stars based on their performance in certain measurable areas. The Grading of Recommendations, Assessment, Development, and Evaluations (GRADE) Working Group approach was then used to determine the overall quality of evidence for the entire study population as a whole. The GRADE score was calculated for each study using a composite measure based on type of evidence, quality, consistency, directness, and effect size [44, 45].
Statistical Analysis
Studies with multiple treatment arms for different variations of either preoperative AMP or extended postoperative AMP were combined to form one group for each study. Basic percentage calculations were made without p values and confidence intervals (CI) during the systematic review. Risk ratios and 95% CIs were calculated for each study and then combined using a random effects model to determine whether extended postoperative AMP reduced SSI development when compared to preoperative AMP protocols through a summary risk ratio. A cumulative meta-analysis was performed and visualized through a forest plot to determine if a significant difference existed at any point in time and, if appropriate, to see when conclusions of statistical significance could have first been reached. Based on the availability of data, post hoc subgroup meta-analyses were conducted to determine whether the effect of preoperative AMP and extended postoperative AMP on the rate of SSI was conditional upon other variables through confounding or effect modification. Stratified analysis was only conducted on patients undergoing instrumented and non-instrumented spinal surgery. All analyses were performed using Stata 12.0 software (StataCorp LP, College Station, TX, USA). All tests were two-sided with an alpha value of 0.05.
Heterogeneity, Bias, and Quality Assessment
The degree of heterogeneity among the risk ratios was evaluated using the I2 statistic. The probability of publication bias and other such study effects were visually assessed through observation of funnel plot asymmetry and Egger’s test of linear regression between the precision of included studies and the standardized effect. Selection bias and information bias were qualitatively evaluated.
The Cochrane Collaboration’s tool for assessing risk of bias, the NOS, and the GRADE Working Group scoring system were utilized to assess quality of evidence for each included study and for the overall study population. The Cochrane Collaboration’s tool for assessing risk of bias decides whether RCTs have a higher, unclear, or low risk of bias based on a study’s performance in key domain areas [46]. The NOS awards up to 9 stars for quality features of observational studies [47, 48]. Meanwhile, the GRADE approach collectively evaluates quality of evidence by awarding points in five different categories: type of evidence, quality of study methodology, consistency among and within studies, directness or generalizability of the study population and outcome to the research question, and effect size among all studies. Potential quality of evidence scores can range from 7 to − 4 and are categorized as high (≥ 4), moderate (3), low (2), and very low (≤ 1). Meanwhile, the GRADE approach appraises strength of recommendation by collectively assessing the quality of evidence (GRADE score), desirability of effects, patient values and preferences, and resourcefulness of intervention. GRADE strength of recommendation scores are determined as either strong or weak. The nominal group technique will be utilized within our group dynamic to reach conclusions of these subjective decisions for quality and risk of bias assessment. Finally, we used the nominal group technique [49, 50] to achieve consensus regarding judgments made for risk of bias and quality assessment.
Compliance with Ethics Guidelines
This article is based on previously conducted studies and does not contain any studies with human participants or animals performed by any of the authors.
Results
Systematic Review
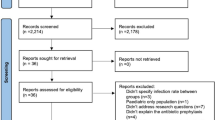
The primary search identified 388 potential studies. After removing 61 duplicate reports and screening the titles and abstracts of the 327 studies that remained, 21 full-text articles were reviewed in detail (Fig. 1). Five studies met inclusion criteria: one RCT [27] and four retrospective cohort studies [28, 33, 51, 52].
Pertinent qualitative and quantitative details from each of these five studies are presented and summarized. Included studies followed CDC guidelines for SSI diagnosis to varying degrees. Each study identified SSI through the clinical observation of the surgeon or attending physician, while some studies also utilized positive wound cultures and other diagnostic measures to supplement diagnosis. The most notable inconsistency across included studies in meeting CDC criteria was the time period in which SSI diagnosis was made. The observational period ranged from within 30 days [28] to more than 6 months [33] after the operation. Hellbusch et al. [27] was the only study that did not elucidate their observational window for diagnosing SSI. The one included RCT was found to have a high risk of bias [27] after evaluation with the Cochrane Collaboration’s tool for assessing risk of bias [46]. Meanwhile, the NOS [47] quality scores of the four remaining cohort studies ranged from 6 stars for two studies [33, 52] to 7 stars for two other studies [28, 51].
Hellbusch et al. [27] conducted an RCT assessing the rate of SSI among 269 consecutive, uninfected patients undergoing clean, instrumented lumbar fusion (97 posterior long segment fusions [PLSFs], 94 transforaminal lumbar interbody fusion and posterior long segment fusions [TLIF/PLSFs], 21 posterior lumbar interbody fusion and posterior long segment fusions [PLIF/PLSFs], 16 minimally invasive sextant fusions, and 5 anterior lumbar interbody fusions [ALIFs]) for degenerative disease, with or without spinal stenosis, from 2000 to 2003. Potential study participants with a cephalosporin or severe penicillin allergy were excluded. Included patients were randomized to one of two AMP protocols: a single preoperative dose of cefazolin or an extended 10-day postoperative regimen of cefazolin and cephalexin. Both AMP protocols received intraoperative dosing. Only 233 patients (male [M] = 102, female [F] = 131; age range = 21–82 years) completed the entire study: 6 patients withdrew secondary to complications or adverse reactions and 30 patients were eliminated from the study after deviation from their assigned AMP protocol. From the 30 eliminated patients, 27 received additional postoperative antibiotic doses, primarily for infectious reasons such as fever of unknown origin and urinary tract infection, while 3 had missed antibiotic doses. Information regarding the length of the observational window for SSI diagnosis was not provided, but all SSIs occurred within 21 days of surgery.
The overall rate of SSI was 3.0% (= 7/233). Two superficial SSIs were reported among the 116 patients receiving extended postoperative AMP, and 5 superficial SSIs were observed among the 117 patients receiving preoperative AMP. Difference testing failed to establish a statistically significant difference in the rate of SSI between patients who received extended postoperative AMP and those who received preoperative AMP (2/116 = 1.72% versus 5/117 = 4.27%; p > 0.25). Two one-sided test (TOST) equivalence testing also failed to produce a statistically significant result. Multivariate analysis included a variety of patient and operative characteristics thought to be associated with SSI, but no statistically significant relationships were reported. Hellbusch et al. hesitated to put forth a clinical recommendation for or against the administration of extended postoperative AMP as a result of insufficient study power. However, they indicated that it seemed unlikely that extended postoperative AMP regimens lasting less than the 10 days would provide additional protection against SSI development given their null result. The Cochrane Collaboration’s risk of bias summary assessment for the outcome of SSI from Hellbusch et al. was high. Meanwhile, Hellbusch et al.’s GRADE evidence quality score was moderate, while its strength of clinical recommendation was found to be strong.
Regarding their study’s low power, Hellbusch et al. noted that 1400 patients would be required to detect a statistically significant difference in SSI as a result of the small effect size difference in proportions (ES = 0.074) and the very low overall rate of infection. As surgical practices improve and SSI becomes less common, the low rate of occurrence undermines the feasibility of conducting adequately powered trials. This is an artifact of surgical practice, and we do not believe that this study by Hellbusch et al. should be overly penalized for encountering this difficulty. However, it is worth noting that eliminating patients who received additional postoperative antibiotics may introduce bias in either direction. The distribution of these patients between treatment arms was not reported, making it difficult to account for potential systematic differences between eliminated patients and adherent patients without individual level data. The authors removed these subjects rather than conducting an as-treated analysis or otherwise statistically addressing this possible source of bias.
The designs of the other four included studies were retrospective. Dobzyniak et al. [52], Kanayama et al. [51], and Kakimaru et al. [28] each took advantage of changes to AMP protocols within their respective institutions to evaluate the effects of different dosing regimens on the rate of SSI in spinal surgery. These institutional changes were motivated by the null result of an in-house animal study assessing extended postoperative AMP on spinal infectious [29] for Dobzyniak et al. and CDC recommendations [22] for Kanayama et al. and Kakimaru et al. In each case, existing AMP protocols were reduced from extended postoperative AMP to preoperative AMP. These temporal variations in prophylaxis could introduce bias, as other uncontrolled factors in the institutional setting and patient population also may have changed over the study period. Nonetheless, the purpose of this paper is to formulate recommendations for institutions, and studies that review the effects of institutional protocol adjustments provide valuable insight to the current review.
Dobzyniak et al. [52] conducted a retrospective cohort study evaluating the rate of SSI among 636 patients (M = 393, F = 243; mean age = 43 years) undergoing hemilaminotomy and discectomy for herniated lumbar disc from 1993 to March 1999. Patients in the preoperative AMP group only received a single dose of AMP. Information regarding the duration of the extended postoperative AMP regimen or whether intraoperative dosing was administered for either group was not provided. Cefazolin was the primary antibiotic administered in both groups. There were 26 patients who did not receive preoperative AMP during this time period that were excluded from the final statistical analysis of the remaining 610 patients. None of these eliminated patients developed SSI. No patients were lost to follow-up during the 6-week period of postoperative observation.
The overall rate of SSI was 1.3% (= 8/610). Five SSIs were reported among the 418 patients receiving extended postoperative AMP, while 3 SSIs were reported among the 192 patients receiving preoperative AMP. Information regarding the incisional type of SSI was not provided. The rate of SSI for the extended postoperative AMP group was not significantly different from the preoperative AMP group (3/192 = 1.20% versus 5/418 = 1.56%; p = 0.71). Dobzyniak et al. concluded that a single preoperative dose of AMP is equally effective in the prevention of SSI following lumbar discectomy as extended postoperative AMP. The NOS quality of evidence score for Dobzyniak et al. accumulated six stars.
Kanayama et al. [51] conducted a retrospective cohort study of 1597 consecutive, uninfected patients (male = 912, female = 685; mean age = 55.4 years) undergoing instrumented and non-instrumented posterior decompression lumbar surgeries for numerous diagnoses (686 disc herniations, 340 degenerative spondylolistheses, 259 spinal stenoses, 73 failed lumbar surgeries, 52 degenerative scolioses, 48 isthmic spondylolistheses, 34 spinal traumas, 27 foraminal stenoses, 27 spinal tumors, and 51 miscellaneous cases) from January 1999 to September 2004. Patients in the preoperative AMP group received a single dose of antibiotics, while those in the extended postoperative AMP group received an extended 5- to 7-day postoperative regimen. Both protocols provided intraoperative dosing and surgical site irrigation with first-generation cephalosporins. Only SSIs occurring within 6 months of surgery were included and no subjects were lost during the observational period.
The overall rate of SSI was 0.7% (= 11/1597). Two deep SSIs were observed among the 464 patients receiving preoperative AMP, while 9 SSIs (2 superficial and 7 deep) were reported among the 1133 patients receiving extended postoperative AMP. Among the 665 cases that utilized instrumentation, there was 1 SSI among the 182 patients receiving preoperative AMP and 7 SSIs among the 483 patients receiving extended postoperative AMP. Meanwhile, among the 932 patients undergoing non-instrumented, posterior decompression, there was 1 SSI reported among the 282 patients receiving preoperative AMP and 2 SSIs observed among the 650 patients receiving extended postoperative AMP. Information regarding SSI incisional type for stratified analyses based on instrumentation was not provided. The rate of SSI did not differ significantly between the extended postoperative AMP group and the preoperative AMP group (9/1113 = 0.8% versus 2/464 = 0.4%, p > 0.05). Only deep SSIs were observed in the preoperative AMP group (2/464 = 0.4%); the rate of deep and superficial SSI was 0.62% (= 7/1133) and 0.18% (= 2/1113), respectively, in the extended postoperative AMP group. Among cases utilizing instrumentation, the difference in rate of SSI between the two study arms was again insignificant (7/483 = 1.4% versus 1/182 = 0.5%, p > 0.05). Kanayama et al. concluded that a single preoperative dose of AMP, with intraoperative dosing, is effective in the prevention of SSI during both instrumented and non-instrumented lumbar spine surgeries. The NOS quality of evidence score for Kanayama et al. had 7 stars.
Kakimaru et al. [28] conducted a retrospective cohort study of 284 patients (M = 183, F = 101; mean age = 63 years, age range = 16–84 years) undergoing microscopic, non-instrumented spinal decompression surgery (115 lumbar fenestrations, 88 cervical laminoplasties, 60 lumbar discectomies, and 21 other procedures) from October 2003 to August 2009. All patients with infectious spondylitis were excluded. There were three different treatment arms in this study: one preoperative AMP group with 143 patients and two temporally variable extended postoperative AMP groups with a collective total of 141 patients. The patients in the preoperative AMP arm received a single preoperative dose of antibiotics and intraoperative dosing. Meanwhile, patients in the first extended postoperative AMP group received a single preoperative and postoperative dose on the operative day, with further AMP dependent upon the attending surgeon’s discretion (average duration = 2.7 postoperative days), but they did not receive intraoperative dosing. In contrast, patients in the second extended postoperative AMP group received a single preoperative dose, intraoperative dosing, and a single postoperative dose after skin closure on the operative day, but did not receive additional postoperative AMP. These two extended postoperative AMP protocols were combined by the study’s investigators to permit statistical analysis between the 143 patients receiving preoperative AMP (M = 94, F = 49, mean age = 64 years) and the 141 patients receiving extended postoperative AMP (M = 89, F = 52, mean age = 62 years). Cefazolin was the primary antibiotic administered in both groups. There was no mention of patients being lost to follow-up during the 30-day period of postoperative observation.
The overall rate of SSI was 2.1% (= 6/284). There were 4 SSIs (3 superficial and 1 deep) in the extended postoperative group and 2 superficial SSIs in the preoperative AMP group. There was no significant difference in rate of SSI between patients receiving extended postoperative AMP and those receiving preoperative AMP (4/141 = 2.8% versus 2/143 = 1.4%, p = 0.335). The rate of superficial and deep SSI was 2.13% (= 3/141) and 0.71% (= 1/141), respectively, in the extended postoperative AMP group; only superficial SSIs were observed in the preoperative AMP group (2/143 = 1.4%). Kakimaru et al. concluded that the SSI rate among their cohort of patients while undergoing microscopic non-instrumented, spinal decompression surgery was not related to AMP duration. The NOS quality of evidence score for Kakimaru et al. had 7 stars.
Khan et al. [33] conducted a retrospective cohort study of 100 patients (M = 40, F = 60; age range = 18–70 years) undergoing clean, elective, and non-instrumented lumbar decompression surgery (66 fenestrations, 28 laminectomies, and 6 combined operations) for disc prolapse, spinal stenosis, or both diagnoses from January 2006 to March 2008. Patients with fractures, trauma, and Pott’s disease were excluded. Three different perioperative AMP regimens were evaluated in separate treatment arms: one preoperative AMP group and two temporally variable AMP groups. The 21 patients in the preoperative AMP group only received a single preoperative dose. The first extended postoperative AMP group had 59 patients and received a single preoperative dose, intraoperative dosing, and two postoperative doses. Meanwhile, the second extended postoperative AMP group had 20 patients and received a single preoperative dose and at least three postoperative doses until the patient demonstrated improvement. Information regarding the overall duration of procedures and the specific duration for each of the extended AMP protocols was not provided. First- or third-generation cephalosporins were the primary antibiotics administered. There was no information provided regarding patients being lost to follow-up during the observational period for SSI diagnosis, which lasted for more than 6 months after surgery.
The overall rate of SSI was 6% (= 6/100). There were no SSIs in the preoperative AMP group, 1 superficial SSI in the first extended postoperative AMP group with intraoperative dosing and only two postoperative doses, and 5 superficial SSIs in the second extended postoperative AMP group with at least three postoperative doses. There were no significant differences in rate of SSI when comparing the preoperative AMP group to either the extended postoperative AMP group receiving exactly two postoperative doses or to the extended postoperative AMP group receiving at least three postoperative doses (0/21 = 0% versus 1/59 = 1.7%, p = 1.00; 0/21 = 0% versus 5/20 = 25%, p = 0.54). Meanwhile, the difference in the rate of SSI between the two extended postoperative AMP groups was found to be statistically significant (1/59 = 1.7% versus 5/20 = 25%, p = 0.003). However, the reported SSI rate of 25% among patients in the extended postoperative AMP groups receiving three or more postoperative doses is considerably higher than other published rates of SSI following non-instrumented spinal surgery [13, 28, 52]. This study is retrospective, and patients were not randomized. Therefore, patients may have received more than three postoperative doses if they were perceived by their attending surgeon to be at a greater risk of developing SSI. They also may have been more likely to undergo operations that carry a higher risk of complication, such as multilevel laminectomy, as the distribution of procedures among patient groups was not specified. Khan et al. also noted that patients in this extended postoperative AMP group receiving at least three postoperative doses were continued on AMP until they improved postoperatively. However, they did not further specify whether improvement referred to mobility and function or whether this group of patients experienced postoperative complications. Since there were no deep SSIs, the rate of superficial SSI was equal to the overall SSI rate of 6%. Khan et al. concluded that a single preoperative dose of AMP was equally effective as extended postoperative AMP in preventing SSI among clean, elective lumbar surgeries. The NOS quality of evidence score for Khan et al. was 6 stars.
Combined Study Characteristics
Data on patient demographics and SSI development from the five included studies was collected (Table 1). Male patients comprised 55% of the 2824 patients making up the overall cohort. All included studies utilized sample populations that were either completely or predominantly composed of adult patients. Average patient age was reported by Dobzyniak et al., Kakimaru et al., and Kanayama et al. and ranged from 43 [52] to 63 years [28]. Meanwhile, age ranges were provided by Kakimaru et al., Hellbusch et al., and Khan et al. and extended from 16 to 84 years [28]. Studies were inconsistent in their reporting and analysis of medical comorbidities, including tobacco use, chronic steroid use, and body mass index (BMI) as a measure of obesity.
There were 2717 (= 96.2%) patients who underwent lumbar spinal surgery, 109 (3.8%) patients who underwent thoracic spinal surgery, and 5 (< 0.1%) patients who underwent cervical spinal procedures. The distribution of the two AMP protocols across spinal surgery level was not provided. Overall, there were 26 SSIs observed in 1887 patients who receive extended postoperative AMP (crude rate = 1.4%), and there were 12 SSIs observed in 937 patients who received preoperative AMP (crude rate = 1.3%). Studies were inconsistent in their reporting and analysis of operative variables, such as procedural duration, the specific procedure performed, and complications, but meaningful stratified analysis was conducted on instrumentation use and a crude analysis was performed for SSI incisional type.
Hellbusch et al. and Kanayama et al. were the only studies that included patients undergoing instrumented spinal fusion, but only the former detailed the specific fusion procedures performed. The mean number of spinal levels fused was not reported in either study. After stratifying the overall cohort by instrumentation, there were 15 SSIs among 888 patients in the instrumented group and 17 SSIs among 1926 patients in the non-instrumented group. The rate of SSI in patients undergoing instrumented spinal surgery was nearly twice that observed in non-instrumented spinal surgery (15/888 = 1.69% versus 17/1926 = 0.9%). Among patients undergoing instrumented spinal surgery, there were 9 SSIs among 599 patients receiving extended postoperative AMP and 6 SSIs among 299 patients receiving preoperative AMP (1.5% versus 2%, respectively). In contrast, among patients undergoing non-instrumented spinal surgery, there were 17 SSIs among 791 patients receiving extended postoperative AMP and 6 SSIs among 638 patients receiving preoperative AMP (1.3% versus 0.94%, respectively).
Rates of superficial and deep incisional SSIs were differentiated in each study except for Dobzyniak et al. After stratifying the four remaining studies by SSI incisional type, there were 20 superficial and 10 deep SSIs among 2214 patients. Among 1469 patients receiving extended postoperative AMP, there were 13 superficial and 8 deep SSIs (0.89% and 0.54%, respectively). Meanwhile, among 745 patients receiving preoperative AMP, there were 7 superficial and 2 deep SSIs (0.94% and 0.27%, respectively). There was no discernable difference in rate of SSI when stratified by the incisional type secondary to crude analysis.
Although each included study reported on the microbiology of positive cultures to varying degrees, inconsistencies in reaching diagnosis through wound culture and the reporting of these findings, both across and within studies, prevented us from ascertaining the true percentage of patients from the overall cohort with positive wound cultures and their individual microbiology. We were also unable to reliably determine what causative organisms were most commonly encountered among the two different AMP protocols and whether any associations existed between instrumentation use or the incisional type of SSI with specific types of microorganisms. In particular, Dobzyniak et al. [52] did not specify the absolute number SSIs attributable to identified causative bacteria, but noted that the majority of positive cultures grew Staphylococcus species. Since most patients either had negative cultures or were diagnosed solely through clinical observation, only 20 individual patients from the overall cohort were reported to have positive wound cultures [27, 28, 33, 51]. Causative organisms included Staphylococcus aureus (n = 10) [28, 33, 51], coagulase-negative Staphylococcus species (n = 5) [27, 28, 51], methicillin-resistant S. aureus (MRSA) (n = 3) [28, 51], Enterococcus faecalis (n = 1) [51], and gram-positive bacillus (n = 1) [51].
There were also inconsistencies in the reporting of AMR strains and bacteria-induced postoperative complications. Kanayama et al., Kakimaru et al., and Khan et al. commented on antibiotic resistance patterns of cultured organisms. Among the 19 patients with positive cultures in these three studies, resistant organisms were cultured in 6 of 15 patients receiving extended postoperative AMP (40%), and in 0 of 4 patients receiving preoperative AMP [28, 33, 51]. In addition, two cases of pseudomembranous colitis secondary to C. difficile were noted in the extended postoperative AMP group while no cases were noted in the preoperative AMP group [28].
The overall combined GRADE quality of evidence score was low. Additionally, the included studies had an average GRADE quality of evidence score of low when this evaluation criteria was applied to them individually.
Meta-Analysis
The overall risk of SSI development in patients who received extended postoperative AMP was 1.11 times the risk of SSI development in patients who only received preoperative AMP, though this effect was not significant (RR = 1.11, 95% CI 0.53–2.36, p = 0.78) (Fig. 2a). There was no evidence of heterogeneity of treatment effects among the included studies (I2 = 0%; p = 0.50), and cumulative meta-analysis failed to reveal a significant risk ratio at any time.
a Forest plot and pooled relative risk of SSI development in patients who received both preoperative and extended postoperative AMP compared to a patients who only received preoperative AMP. b Funnel plot of all studies with non-zero rates of SSI demonstrating a symmetric distribution of study estimates around the estimated true effect size, suggesting no evidence for publication bias. RR relative risk, CI confidence interval, SSI surgical site infection, AMP antimicrobial prophylaxis
Publication bias was assessed using funnel plots (Fig. 2b), and the study estimates appear symmetric about the estimated true effect size. However, the small number of studies limits the interpretation of a funnel plot. Egger’s test for small-study effects was also found to be statistically insignificant (p = 0.81). Taken together, these results support the hypothesis that the SSI rate after spine surgery is not different between extended postoperative AMP and preoperative AMP.
Stratified meta-analysis was performed by whether instrumentation was implanted during surgery. Among patients who underwent instrumented spinal surgery, no significant difference in risk of SSI development was observed between patients who received extended postoperative AMP and those who received preoperative AMP (RR = 0.92, 95% CI 0.15–5.75, p = 0.93) (Fig. 3a). No notable benefit or harm was observed from extended postoperative AMP in terms of occurrence of SSI. Only two studies, Hellbusch et al. and Kanayama et al., contributed to the instrumented cohort in this stratified meta-analysis; the results of neither study were statistically significant independently, and their estimated effects actually trended in opposite directions (RR = 0.40 and RR = 2.64, respectively). Therefore, some heterogeneity could be expected and is indicated by an I2 of 49.1%, but this was found not to be statistically significant (p = 0.16). The interpretation of the funnel plot is extremely limited because of a very small sample size (Fig. 3b).
a Forest plot and pooled relative risk of SSI development in patients undergoing instrumented spinal surgery who received both preoperative and extended postoperative AMP compared to patients who only received preoperative AMP. b Funnel plot of all studies with non-zero rates of SSI demonstrating a symmetric distribution of study estimates around the estimated true effect size. The small sample size limits the interpretation of this plot. RR relative risk, CI confidence interval, SSI surgical site infection, AMP antimicrobial prophylaxis
Four studies reported data on outcomes following non-instrumented spinal surgery. The pooled risk of SSI following non-instrumented spinal surgery in patients who received extended postoperative AMP was not significantly different from the risk of SSI in patients who received preoperative AMP (RR = 1.25, 95% CI 0.49–3.17, p = 0.65) (Fig. 4a). There was no evidence of heterogeneity of treatment effects among the included studies (I2 = 0%; p = 0.70). Again, the study estimates appear symmetrically dispersed about the estimated true effect size, suggesting absence of publication bias, although any conclusions drawn are limited by small study numbers (Fig. 4b).
a Forest plot and pooled relative risk of SSI development in patients undergoing non-instrumented spinal surgery who received both preoperative and extended postoperative AMP compared to patients who only received preoperative AMP. b Funnel plot of all studies with non-zero rates of SSI demonstrating a symmetric distribution of study estimates around the estimated true effect size. The small sample size limits the interpretation of this plot. RR relative risk, CI confidence interval, SSI surgical site infection, AMP antimicrobial prophylaxis
Discussion
Guidelines over the optimal timing of perioperative AMP in spinal surgery have been subject to clinical debate for over half a century. Prior to publication of Burke’s [53] landmark bovine study in 1961, the prophylactic efficacy of perioperative antibiotic utilization was poor secondary to administration that was commonly confined to the postoperative window [54, 55]. Succeeding publications, including Classen et al.’s [30] seminal 1992 observational study, substantiated Burke’s findings over the next three decades [29, 56,57,58,59,60,61,62,63] and highlighted the importance of administering preoperative AMP just prior to first incision. Horwitz and Curtin [6] are often credited with first demonstrating the efficacy of initiating AMP preoperatively in spinal surgery in 1975 and, ultimately, these authors recommended extended prophylaxis. Despite this evidence, RCTs [64,65,66,67,68,69] had so far failed to demonstrate clinical efficacy of perioperative AMP in preventing spinal SSI. Barker [19] performed a meta-analysis of these RCTs in 2002 and found results that robustly reinforced administration of AMP with at least a single preoperative dose during spinal surgery, even when anticipated rates of SSI without AMP are low.
Although research has heavily focused on when to start AMP in spinal surgery, questions over its overall duration remain. In 2003, Dobzyniak et al. [52] and Beiner et al. [70] were the first to call attention to the lack of consistent recommendations over how long, if at all, to continue AMP postoperatively. Clinical studies have since not only assessed the efficacy of extended postoperative therapy to preoperative AMP alone [27, 28, 33, 51, 52] but have also compared the therapeutic benefit of extended AMP regimens to others that differ in postoperative duration [32]. The NASS [24, 25] has made recommendations for optimal antibiotic duration for both uncomplicated and complicated spinal surgeries after qualitatively reviewing these studies in 2013, but a meta-analysis of comparative clinical trials has still not yet been conducted.
The main meta-analysis performed in this study combined the results of one RCT and four retrospective cohort studies. We did not find evidence that the administration of an extended AMP regimen was favorable over the administration of a single preoperative dose as indicated by an overall risk ratio of 1.11 and 95% confidence interval of 0.53–2.36 (p = 0.78). Heterogeneity analysis did not reveal variability among the studies and did not provide any evidence that the studies could not be combined. Funnel plots did not reveal any clear publication bias, and Egger’s test for small-study effects was found to be non-significant. These findings support the hypothesis that combined preoperative and extended postoperative antimicrobial prophylaxis does not reduce SSI rates in adult patients undergoing spinal surgery as compared to a single preoperative dose.
The broad confidence interval of the risk ratio and the insignificant test statistic imply that the meta-analysis is likely underpowered to detect small differences in the risk of SSI after spinal surgery between these two AMP regimens. However, the above finding is consistent with previously published studies. As mentioned previously, Barker performed a meta-analysis of six RCTs in order to evaluate the role of AMP in spinal surgery. A subgroup analysis of these studies failed to demonstrate a greater benefit of prophylaxis when postoperative doses were added to a single preoperative dose [19]. Furthermore, a large meta-analysis of 28 RCTs comparing preoperative and extended AMP in the setting of major surgery also failed to demonstrate superiority of extended AMP over preoperative only regimens [20]. Although the findings of this meta-analysis and the previous literature cannot definitively answer the question of whether extended postoperative AMP provides additional benefit over preoperative only AMP during spinal surgery, it contributes significantly to the existing state of knowledge by providing more evidence that preoperative AMP alone may be sufficient to decrease the risk of SSI.
This is a valuable observation related to the high costs of SSI, the association of antibiotic use with increasing antimicrobial resistance, and related morbidity such as C. difficile colitis. Attributable costs per spinal SSI range from US$15,000 to upwards of US$100,000 [7, 11, 71,72,73,74,75,76,77,78]. Notably, the costs incurred from SSI after spinal surgery are greater than the reported additional expenditures required for the average surgical patient who experiences SSI [79, 80].
In terms of SSI and the potential for AMR, most infections reported in the included studies were, unsurprisingly, due to organisms related to skin flora, and included infections with S. aureus and coagulase-negative Staphylococcus species. Infections with E. faecalis and gram-positive bacillus were also observed. Interestingly, among those studies that commented on antimicrobial patterns, resistant organisms, such as MRSA, were cultured in six of fifteen patients treated with extended AMP and in zero of four patients who only received preoperative AMP. Clearly, the numbers of patients in these two groups are low, and, thus, meaningful statistical inference is impossible. Furthermore, the patients were not randomized and it is possible that patients with increased risk for developing infection from resistant organisms may have also been more likely to receive extended AMP. However, despite these limitations, this observation is concerning given emerging antibiotic resistance patterns. The potential to reduce AMR infections by minimizing the duration of perioperative AMP should continue to encourage additional, high-quality research.
It is incumbent upon all surgeons to endeavor to prevent SSI and AMR emergence by exercising conservative caution when using perioperative AMP. To date, surgeon adherence to recommended guidelines often deviates significantly in practice, especially with regard to how long AMP is extended into the postoperative window [81,82,83]. This variation in prophylactic care is secondary to numerous factors, including instrumentation use, surgical complexity, and patient comorbidity status [9, 16, 52], but can also result from institutional guidelines or surgeon discretion [28, 52]. This lack of homogeneity in AMP in spinal surgery is unsurprising given the historically contradicting recommendations made regarding extension of AMP into the postoperative window [6, 16, 29, 52, 53, 67], and highlights the need for this systematic review and meta-analysis. It is important to weigh the real risk of antimicrobial resistance against a perceived, yet scientifically unsupported, benefit of continuing antibiotics postoperatively. These considerations aided the authors in developing the recommendation that AMP should not be continued postoperatively in uncomplicated, adult patients during spinal surgery.
However, it is difficult to generalize the results of this meta-analysis to all spinal surgery patients because of the complex interaction between patient-related risk factors, surgical complexity, and other unmeasured factors, particularly in complicated cases. In order to help address this uncertainty, we performed stratified meta-analyses. Most of the studies did not report the rates of SSI among various related medical comorbidities; therefore, the types of stratified analyses that could be carried out were limited. However, stratification was possible according to whether instrumentation was implanted. Patients undergoing instrumented spinal fusion are subjected to a higher risk of postoperative infection due to longer operative time, increased procedure complexity, and the implantation of a foreign body [15, 16, 61]. As expected, the rate of SSI in patients following instrumented spinal surgery was nearly twice that observed in patients following non-instrumented spinal surgery. However, we did not find a significant benefit of extended AMP over preoperative only AMP in patients either undergoing instrumented spinal surgery or in those undergoing non-instrumented spinal surgery.
Overall, the strengths of this study make its findings noteworthy for spine surgeons. First, the inclusion criteria concerning the study design were strict. In particular, each study was required to have a near equivalent comparison group which allowed for more accurate effect estimates between the two AMP protocols. This is highlighted in Kanayama et al.’s [51] cohort study in which the investigators identified a potential self-imposed limitation to their study: the definition of SSI required surgical treatment of the wound. Since the majority of SSIs were superficial and managed with conservative therapy, the reported rate of SSI for both treatment arms may be underestimated. However, since the same definition was utilized for both treatment arms, the relative rate of SSI between the two groups may actually be accurately reflected, despite the conservative nature of the definition itself. But, this may not necessarily be the case. Preoperative AMP may have a different effect on SSI requiring surgical intervention when compared to extended postoperative AMP, and this presents an opportunity for bias in this meta-analysis. Also, since Kanayama et al. provided data for the primary analysis and both subgroup meta-analyses, this confounding could extend throughout the study.
The internal validity of this review was high secondary to assessment with funnel plots and Egger’s test, which both failed to identify publication bias. Since this meta-analysis found a non-significant result, the publication of further non-significant studies on this topic would only lend greater support to our current findings, making publication bias of little concern to the authors. Additionally, there was no heterogeneity present among the five studies. Taken together, these results support the internal validity of the results reached in this meta-analysis. With that said, however, the external validity of this study may be called into question. It is important to note that weaknesses of this meta-analysis and its methodology are mutually exclusive to the limitations of the included studies. One weakness of the present study is the generalizability of results. This study aimed to report on the efficacy of temporally variable perioperative AMP in spinal surgery at any level. However, 96.2% (2717/2824) of patients underwent spinal operations confined to the lumbar spine. Stratification of the 107 patients undergoing either cervical or thoracic operations was not possible because of the small sample size. Although unlikely, it is possible that cervical, thoracic, or coccygeal procedures experience different SSI rates under different perioperative AMP protocols. Additionally, there was not enough data provided to conduct further subgroup analyses on patient comorbidity status, which may impact SSI outcomes concerning AMP duration. We were able to stratify based on instrumentation use, but these subgroup analyses were hindered by the same limitations detracting from the primary meta-analysis. There were also differences between studies in the length of AMP prescribed based on individual surgeon preference. Thus, although there was no evidence supporting the extensions of AMP postoperatively for either instrumented or non-instrumented patient subgroups, decisions regarding a patient’s risk of SSI and whether antibiotic prophylaxis is needed following surgery must be made on an individual basis until more data becomes available.
There were several limitations of the studies included in this review and meta-analysis that should be considered separately from the meta-analysis itself. The GRADE quality of evidence score for the overall included data ranged from low [48] to moderate [46] quality, which was primarily related to their observational and often retrospective nature [10, 19,20,21], as the authors were unable to control for unmeasured confounding and bias. Since the RCT [27] was not blinded, investigators were aware of the AMP protocol administered to each patient and bias could arise from increased observation and surgical performance. The majority of included studies were underpowered with relatively small sample sizes, increasing the likelihood of type II error and limiting the ability to detect significant differences in SSI rates between the different AMP protocols. Most studies failed to include explicit and objective definitions of SSI, including those for superficial and deep SSIs; the criteria in which SSI was diagnosed; or who was making these decisions. One study followed guidelines set forth by the CDC [28]. Another study required positive wound cultures and/or the attending surgeon’s clinical impression of typical infectious signs that led to additional surgical intervention [51]. Meanwhile, others did not mention their criteria for defining and diagnosing SSI [33]. Although some studies stated what individuals were making these clinical decisions, possible sources of bias, especially for the prospectively collected databases and RCT, could arise. In particular, if the prospective studies failed to have a third-party record their data, observer bias could occur if the investigators knew which patients received which AMP protocol. At the same time, the retrospective studies could be biased since the authors had no control over data collection. Similarly, the follow-up observational period for SSI identification demonstrated considerable variation among studies and ranged from within 30 days [47] to 6 months [50] and longer [48] to unspecified follow-up times [50]. This review did not exclude studies based on antibiotic type used or changes in antibiotic administered during treatment. The timing of initial preoperative AMP administration and the length of postoperative AMP duration in the extended postoperative groups also demonstrated significant temporal heterogeneity both within and between studies, ranging from within 30 min prior to skin incision to 10 days postoperatively [27, 28, 51]. The timing of perioperative AMP was either unknown or not reported by Dobzyniak et al. and Khan et al. [33, 52]. Hellbusch et al. [27] statistically assessed the impact of demographic factors, comorbidities and past surgical history, and operative variables on the outcome of SSI development, finding no associations of statistical significance. No multivariate analyses were conducted in any of the included studies. The considerable variability in defining, diagnosing, and reporting SSI; the inconsistency in the postoperative observational period; and the inconsistency in preoperative and extended postoperative extended AMP somewhat diminish the validity of this meta-analysis.
These strengths and weaknesses were succinctly enumerated and tallied by the GRADE Working Group scoring system in the results section. As a result of the relative novelty of the GRADE approach and the very low to moderate quality of evidence scores awarded to the included studies, the GRADE scoring system deserves further mention here. The GRADE approach is currently regarded as one of the most effective and unequivocal scoring methods to systematically connect evidence-quality evaluations to degree of efficacy for clinical recommendations [44]. This underlies one of the primary reasons why the GRADE approach is valuable in improving clinical practice: its scoring system transparently separates decisions regarding evidence quality from decisions evaluating recommendation strength [45]. In other words, studies with high quality evidence do not always indicate strong clinical recommendations, and strong clinical recommendations can sometimes be drawn from studies with low quality evidence [45]. Therefore, despite the average low quality of evidence scoring assigned to the included studies through the GRADE system in this review, the statistically null result reported in the present analysis and its associated clinical recommendation to provide spinal surgery patients with preoperative AMP only should give pause to spinal surgeons, nonetheless.
The authors also believe that certain qualities of the GRADE scoring system led to artificially low scores among the included articles. Firstly, the GRADE scoring approach is partial towards studies that demonstrate statistical significance. All included studies failed to demonstrate statistically significant results, and this contributed to their overall low quality of evidence scores through poor scoring in two of the five categories of the GRADE scoring system: effect size and consistency. Effect size awards increasing points when every included study reports large, unidirectional magnitudes of effect. Meanwhile, consistency assigns points to studies that are characterized by dose response patterns, either between or within studies. Magnitude of effect and dose response are two of Hill’s criteria for causality [84] and are desirable if, in reality, there is a difference in effect between treatment arms. However, their inclusion in the GRADE scoring methodology assumes that the true and desirable outcome is always statistically significant. It is pertinent to remember that statistical significance and clinical significance are distinguishable, and statistically insignificant results can impact care in clinically meaningful ways. Given the clinical significance of our null result and its potential to reduce unnecessary medical expenditures and decrease risk of AMR development, it is appropriate here to highlight these shortcomings.
An additional issue brought on by the GRADE approach that contributes to the overall low quality of evidence scores is the explicit value placement on study design. Specifically, the GRADE scoring system allocates four points to RCTs and two points to observational studies, reflecting the old adage that RCTs are the “gold standard” of research. However, the sample size necessary for an RCT to establish statistical significance is prohibitively large in SSI studies in spinal surgery. For example, the investigators [27] who conducted the only RCT included in this meta-analysis hesitated to make clinical recommendations secondary to their small sample size of 233 patients. Utilizing the low SSI rates experienced by patients in their study and the small effect size difference in proportions (ES = 0.074), they performed a power calculation to determine the sample size that would have been required to obtain statistically significant results. They found that 1400 patients, a study population six times greater than their original sample size, would be required to establish statistical significance in comparing SSI rates in patients receiving preoperative AMP to those receiving extended postoperative therapy at the desired power. Additionally, Dimick et al. [26] further justified the impracticality of conducting adequately powered RCTs on SSI development and perioperative AMP in spinal surgery and the notable risk of making a type II error when not enough patients are enrolled. They demonstrated that 474, 988, and 2518 patients would be required to detect baseline SSI rates of 10%, 5%, and 2%, in each treatment group, respectively, of an RCT with 80% power to detect a 50% reduction in SSI through perioperative AMP in spinal surgery. Therefore, it was highly unlikely that RCTs would have been available for this meta-analysis because of the exorbitant costs required to enroll enough patients to ensure adequate power. Thus, the relative ease of identifying patients for a large observational study may, in the case of investigating SSI rates in spinal surgery, make observational data more meaningful than data from small RCTs. However, this is not reflected in the GRADE approach, which broadly penalizes observational studies when, in this clinical application, they are actually the best option for finding statistically significant results, especially given the low incidence of SSI in spinal surgery [85]. This meta-analysis benefited from the relatively large sample sizes provided by the majority of the included observational studies. The GRADE scoring system aims to objectively evaluate a research process in which investigators must invariably make subjective decisions. Researchers and practitioners must consider the need for flexibility in the interpretation of such evaluative methods in order to adapt to the unique challenges of conducting research in particular medical and surgical specialties.
In light of these deficiencies, the authors of this review endorse the validity of the present analysis despite the low GRADE evidence quality scores assigned to the included studies. This is evidenced by the strong strength of recommendation GRADE scores awarded to four of the five included studies, emphasizing the importance of the research context and the discrepancy between statistical and clinical significance. Therefore, the evidence utilized for this review and analysis, although evaluated to be of moderate to poor overall quality, still provides useful, evidence-based clinical recommendations that have the potential to impact current practices as spinal surgeons still commonly extend perioperative AMP based on personal discretion. Therefore, the recommendation set forth by this systematic review and meta-analysis is strong according to the GRADE scoring system.
Conclusion
A meta-analysis of comparative clinical trials investigating the development of SSI in 2824 adult spinal surgery patients, receiving either preoperative or extended postoperative AMP, revealed that SSI occurred in 1.4% of all cases. Analysis of the collective data demonstrated no statistical difference between the temporally variable AMP protocols, suggesting that preoperative AMP without extended postoperative therapy is clinically equivalent to extended postoperative AMP. Subgroup analyses also failed to demonstrate statistical significance in SSI development between the different perioperative AMP protocols between instrumented and non-instrumented spinal surgery and in the dermal level of SSI origin. Postoperative AMP appears unnecessary for spinal surgery given its increased medical costs and risk for AMR development from gratuitous prophylaxis. However, higher-quality data are needed to determine the theoretically optimal perioperative AMP regimen. As a result of the low incidence of SSIs, the high percentage of patients who underwent lumbar region surgery, the inability to stratify results by patient comorbidity, and heterogeneity between study protocols regarding AMP use, the results found in these studies may not be generalizable to a larger patient population.
Surgeons should use their clinical judgment in prescribing the correct prophylaxis for their patients while keeping in mind the real medical, financial, and societal costs of unnecessary antibiotic use. Related to the low incidence of SSI in spinal surgery, future studies of perioperative AMP duration in this surgical subspecialty should either be adequately powered RCTs or large prospective observational studies.
References
Blam OG, Vaccaro AR, Vanichkachorn JS, et al. Risk factors for surgical site infection in the patient with spinal injury. Spine (Phila Pa 1976). 2003;28(13):1475–80.
Browne JA, Cook C, Pietrobon R, Bethel MA, Richardson WJ. Diabetes and early postoperative outcomes following lumbar fusion. Spine (Phila Pa 1976). 2007;32(20):2214–9.
Carreon LY, Puno RM, Lenke LG, et al. Non-neurologic complications following surgery for adolescent idiopathic scoliosis. J Bone Jt Surg Am Vol. 2007;89(11):2427–32.
Culver DH, Horan TC, Gaynes RP, et al. Surgical wound infection rates by wound class, operative procedure, and patient risk index. National Nosocomial Infections Surveillance System. Am J Med. 1991;91(3b):152s–7s.
Fang A, Hu SS, Endres N, Bradford DS. Risk factors for infection after spinal surgery. Spine (Phila Pa 1976). 2005;30(12):1460–5.
Horwitz NH, Curtin JA. Prophylactic antibiotics and wound infections following laminectomy for lumber disc herniation. J Neurosurg. 1975;43(6):727–31.
McGirt MJ, Parker SL, Lerner J, Engelhart L, Knight T, Wang MY. Comparative analysis of perioperative surgical site infection after minimally invasive versus open posterior/transforaminal lumbar interbody fusion: analysis of hospital billing and discharge data from 5170 patients. J Neurosurg Spine. 2011;14(6):771.
National Nosocomial Infections Surveillance System. National Nosocomial Infections Surveillance (NNIS) System Report, data summary from January 1992 through June 2004, issued October 2004. Am J Infect Control. 2004;32(8):470–85.
Olsen MA, Nepple JJ, Riew KD, et al. Risk factors for surgical site infection following orthopaedic spinal operations. J Bone Jt Surg Am Vol. 2008;90(1):62–9.
Omeis IA, Dhir M, Sciubba DM, et al. Postoperative surgical site infections in patients undergoing spinal tumor surgery: incidence and risk factors. Spine (Phila Pa 1976). 2011;36(17):1410–9.
Parker SL, Adogwa O, Witham TF, Aaronson OS, Cheng J, McGirt MJ. Post-operative infection after minimally invasive versus open transforaminal lumbar interbody fusion (TLIF): literature review and cost analysis. Minim Invasive Neurosurg MIN. 2011;54(1):33–7.
Pull ter Gunne AF, Cohen DB. Incidence, prevalence, and analysis of risk factors for surgical site infection following adult spinal surgery. Spine (Phila Pa 1976). 2009;34(13):1422–8.
Smith JS, Shaffrey CI, Sansur CA, et al. Rates of infection after spine surgery based on 108,419 procedures: a report from the Scoliosis Research Society Morbidity and Mortality Committee. Spine (Phila Pa 1976). 2011;36(7):556–63.
Watanabe M, Sakai D, Matsuyama D, Yamamoto Y, Sato M, Mochida J. Risk factors for surgical site infection following spine surgery: efficacy of intraoperative saline irrigation. J Neurosurg Spine. 2010;12(5):540–6.
Weinstein MA, McCabe JP, Cammisa FP Jr. Postoperative spinal wound infection: a review of 2,391 consecutive index procedures. J Spinal Disord. 2000;13(5):422–6.
Wimmer C, Gluch H, Franzreb M, Ogon M. Predisposing factors for infection in spine surgery: a survey of 850 spinal procedures. J Spinal Disord. 1998;11(2):124–8.
Abdul-Jabbar A, Takemoto S, Weber MH, et al. Surgical site infection in spinal surgery: description of surgical and patient-based risk factors for postoperative infection using administrative claims data. Spine (Phila Pa 1976). 2012;37(15):1340–5.
Schnoring M, Brock M. Prophylactic antibiotics in lumbar disc surgery: analysis of 1,030 procedures. Zentralbl Neurochir. 2003;64(1):24–9.
Barker FG 2nd. Efficacy of prophylactic antibiotic therapy in spinal surgery: a meta-analysis. Neurosurgery. 2002;51(2):391–400 (discussion -1).
McDonald M, Grabsch E, Marshall C, Forbes A. Single- versus multiple-dose antimicrobial prophylaxis for major surgery: a systematic review. Aust N Z J Surg. 1998;68(6):388–96.
DiPiro JT, Cheung RP, Bowden TA Jr, Mansberger JA. Single dose systemic antibiotic prophylaxis of surgical wound infections. Am J Surg. 1986;152(5):552–9.
Mangram AJ, Horan TC, Pearson ML, Silver LC, Jarvis WR. Guideline for prevention of surgical site infection, 1999. Hospital Infection Control Practices Advisory Committee. Infect Control Hosp Epidemiol. 1999;20(4):250–78 (quiz 79–80).
Watters WC 3rd, Baisden J, Bono CM, et al. Antibiotic prophylaxis in spine surgery: an evidence-based clinical guideline for the use of prophylactic antibiotics in spine surgery. Spine J. 2009;9(2):142–6.
Shaffer WO, Baisden JL, Fernand R, Matz PG. An evidence-based clinical guideline for antibiotic prophylaxis in spine surgery. Spine J. 2013;13(10):1387–92.
Shaffer WO, Baisden J, Fernand R, Matz PG. Evidence-based clinical guidelines for multidisciplinary spine care: antibiotic prophylaxis in spine surgery. USA: North American Spine Society; 2013.
Dimick JB, Lipsett PA, Kostuik JP. Spine update: antimicrobial prophylaxis in spine surgery: basic principles and recent advances. Spine (Phila Pa 1976). 2000;25(19):2544–8.
Hellbusch LC, Helzer-Julin M, Doran SE, et al. Single-dose vs multiple-dose antibiotic prophylaxis in instrumented lumbar fusion—a prospective study. Surg Neurol. 2008;70(6):622–7 (discussion 7).
Kakimaru H, Kono M, Matsusaki M, Iwata A, Uchio Y. Postoperative antimicrobial prophylaxis following spinal decompression surgery: is it necessary? J Orthop Sci. 2010;15(3):305–9.
Guiboux JP, Cantor JB, Small SD, Zervos M, Herkowitz HN. The effect of prophylactic antibiotics on iatrogenic intervertebral disc infections. A rabbit model. Spine (Phila Pa 1976). 1995;20(6):685–8.
Classen DC, Evans RS, Pestotnik SL, Horn SD, Menlove RL, Burke JP. The timing of prophylactic administration of antibiotics and the risk of surgical-wound infection. N Engl J Med. 1992;326(5):281–6.
Page CP, Bohnen JM, Fletcher JR, McManus AT, Solomkin JS, Wittmann DH. Antimicrobial prophylaxis for surgical wounds. Guidelines for clinical care. Arch Surg. 1993;128(1):79–88.
Takahashi H, Wada A, Iida Y, et al. Antimicrobial prophylaxis for spinal surgery. J Orthop Sci. 2009;14(1):40–4.
Khan IU, Janjua MB, Hasan S, Shah S. Surgical site infection in lumbar surgeries, pre and postoperative antibiotics and length of stay: a case study. J Ayub Med Coll Abbottabad. 2009;21(3):135–8.
Steinberg JP, Braun BI, Hellinger WC, et al. Timing of antimicrobial prophylaxis and the risk of surgical site infections: results from the Trial to Reduce Antimicrobial Prophylaxis Errors. Ann Surg. 2009;250(1):10–6.
Bratzler DW, Houck PM, Richards C, et al. Use of antimicrobial prophylaxis for major surgery: baseline results from the National Surgical Infection Prevention Project. Arch Surg. 2005;140(2):174–82.
Bratzler DW, Houck PM. Antimicrobial prophylaxis for surgery: an advisory statement from the National Surgical Infection Prevention Project. Clin Infect Dis. 2004;38(12):1706–15.
Berenguer CM, Ochsner MG Jr, Lord SA, Senkowski CK. Improving surgical site infections: using National Surgical Quality Improvement Program data to institute Surgical Care Improvement Project protocols in improving surgical outcomes. J Am Coll Surg. 2010;210(5):737–41, 41–3.
Bratzler DW, Hunt DR. The surgical infection prevention and surgical care improvement projects: national initiatives to improve outcomes for patients having surgery. Clin Infect Dis. 2006;43(3):322–30.
Stulberg JJ, Delaney CP, Neuhauser DV, Aron DC, Fu P, Koroukian SM. Adherence to surgical care improvement project measures and the association with postoperative infections. JAMA. 2010;303(24):2479–85.
Horan TC, Gaynes RP, Martone WJ, Jarvis WR, Emori TG. CDC definitions of nosocomial surgical site infections, 1992: a modification of CDC definitions of surgical wound infections. Infect Control Hosp Epidemiol. 1992;13(10):606–8.
Swoboda SM, Merz C, Kostuik J, Trentler B, Lipsett PA. Does intraoperative blood loss affect antibiotic serum and tissue concentrations? Arch Surg. 1996;131(11):1165–71 (discussion 71–72).
Polly DW Jr, Meter JJ, Brueckner R, Asplund L, van Dam BE. The effect of intraoperative blood loss on serum cefazolin level in patients undergoing instrumented spinal fusion. A prospective, controlled study. Spine (Phila Pa 1976). 1996;21(20):2363–7.
Alexander JW, Solomkin JS, Edwards MJ. Updated recommendations for control of surgical site infections. Ann Surg. 2011;253(6):1082–93.
BMJ Group. What is GRADE? BMJ 2014; 2012 [updated Sept 20, 2012]. https://clinicalevidence.bmj.com/x/set/static/ebm/learn/665072.html. Accessed Mar 14, 2014.
Guyatt GH, Oxman AD, Vist GE, et al. GRADE: an emerging consensus on rating quality of evidence and strength of recommendations. BMJ. 2008;336(7650):924–6.
Higgins JPT, Altman DG, Stern JAC. Chapter 8: assessing risk of bias in included studies. In: Higgins JPT, Green S, editors. Cochrane Handbook for Systematic Reviews of Intervenions Version 510 (updated March 2011): The Cochrane Collaboration; 2011.
Wells GA, Brodsky L, O’Connell D, et al. An evaluation of the Newcastle Ottawa Scale: an assessment tool for evaluating the quality of non-randomized studies. In: Abstract, XI International Cochrane Colloquium Book of Abstracts. 2003;O-63.
Wells GA, Brodsky L, O'Connell D, et al. The Newcastle-Ottawa Scale (NOS) for assessing the quality of nonrandomised studies in meta-analyses. 2014. https://www.ohri.ca/programs/clinical_epidemiology/oxford.asp. Accessed Nov 15, 2014.
Delbecq AL, van de Ven AH. A group process model for problem identification and program planning. J Appl Behav Sci. 1971;7:466–92.
Delbecq AL, van de Ven AH, Gustafson DH. Group techniques for program planning: a guide to nominal group and Delphi processes. Glenview: Scott Foresman; 1975.
Kanayama M, Hashimoto T, Shigenobu K, Oha F, Togawa D. Effective prevention of surgical site infection using a Centers for Disease Control and Prevention guideline-based antimicrobial prophylaxis in lumbar spine surgery. J Neurosurg Spine. 2007;6(4):327–9.
Dobzyniak MA, Fischgrund JS, Hankins S, Herkowitz HN. Single versus multiple dose antibiotic prophylaxis in lumbar disc surgery. Spine (Phila Pa 1976). 2003;28(21):E453–E455455.
Burke JF. The effective period of preventive antibiotic action in experimental incisions and dermal lesions. Surgery. 1961;50:161–8.
Hong HS, Chang MC, Liu CL, Chen TH. Is aggressive surgery necessary for acute postoperative deep spinal wound infection? Spine (Phila Pa 1976). 2008;33(22):2473–8.
Sanchez-Ubeda R, Fernand E, Rousselot LM. Complication rate in general surgical cases; the value of penicillin and streptomycin as postoperative prophylaxis; a study of 511 cases. N Engl J Med. 1958;259(22):1045–50.
Bernard HR, Cole WR. The prophylaxis of surgical infection: the effect of prophylactic antimicrobial drugs on the incidence of infection following potentially contaminated operations. Surgery. 1964;56:151–7.
Polk HC Jr, Lopez-Mayor JF. Postoperative wound infection: a prospective study of determinant factors and prevention. Surgery. 1969;66(1):97–103.
Stone HH, Hooper CA, Kolb LD, Geheber CE, Dawkins EJ. Antibiotic prophylaxis in gastric, biliary and colonic surgery. Ann Surg. 1976;184(4):443–52.
Burdon DW. Principles of antimicrobial prophylaxis. World J Surg. 1982;6(3):262–7.
Kaiser AB. Antimicrobial prophylaxis in surgery. N Engl J Med. 1986;315(18):1129–38.
Massie JB, Heller JG, Abitbol JJ, McPherson D, Garfin SR. Postoperative posterior spinal wound infections. Clin Orthop Relat Res. 1992;284:99–108.
Savitz MH, Malis LI, Meyers BR. Prophylactic antibiotics in neurosurgery. Surg Neurol. 1974;2(2):95–100.
Pavel A, Smith RL, Ballard A, Larsen IJ. Prophylactic antibiotics in clean orthopaedic surgery. J Bone Jt Surg Am Vol. 1974;56(4):777–82.
Rubinstein E, Findler G, Amit P, Shaked I. Perioperative prophylactic cephazolin in spinal surgery. A double-blind placebo-controlled trial. J Bone Jt Surg Br Vol. 1994;76(1):99–102.
Pavel A, Smith RL, Ballard A, Larson IJ. Prophylactic antibiotics in elective orthopedic surgery: a prospective study of 1,591 cases. South Med J. 1977;70(Suppl 1):50–5.
Geraghty J, Feely M. Antibiotic prophylaxis in neurosurgery. A randomized controlled trial. J Neurosurg. 1984;60(4):724–6.
Young RF, Lawner PM. Perioperative antibiotic prophylaxis for prevention of postoperative neurosurgical infections. A randomized clinical trial. J Neurosurg. 1987;66(5):701–5.
Bullock R, van Dellen JR, Ketelbey W, Reinach SG. A double-blind placebo-controlled trial of perioperative prophylactic antibiotics for elective neurosurgery. J Neurosurg. 1988;69(5):687–91.
Djindjian M, Lepresle E, Homs JB. Antibiotic prophylaxis during prolonged clean neurosurgery. Results of a randomized double-blind study using oxacillin. J Neurosurg. 1990;73(3):383–6.
Beiner JM, Grauer J, Kwon BK, Vaccaro AR. Postoperative wound infections of the spine. Neurosurg Focus. 2003;15(3):E14.
Emohare O, Ledonio CG, Hill BW, Davis RA, Polly DW Jr, Kang MM. Cost savings analysis of intrawound vancomycin powder in posterior spinal surgery. Spine J. 2014;14(11):2710–5.
Levi AD, Dickman CA, Sonntag VK. Management of postoperative infections after spinal instrumentation. J Neurosurg. 1997;86(6):975–80.
Parker SL, Shau DN, Mendenhall SK, McGirt MJ. Factors influencing 2-year health care costs in patients undergoing revision lumbar fusion procedures. J Neurosurg Spine. 2012;16(4):323–8.
Godil SS, Parker SL, O'Neill KR, Devin CJ, McGirt MJ. Comparative effectiveness and cost-benefit analysis of local application of vancomycin powder in posterior spinal fusion for spine trauma: clinical article. J Neurosurg Spine. 2013;19(3):331–5.
Gold HS, Moellering RC Jr. Antimicrobial-drug resistance. N Engl J Med. 1996;335(19):1445–533.
Poelstra KA, Vaccaro AR, Albert TA, et al. Gentamicin microspheres for infection prophylaxis in the rabbit spine. In: 53rd orthopaedic research society (ORS) meeting; February 10–14; San Diego, 2007.
Stall AC, Becker E, Ludwig SC, Gelb D, Poelstra KA. Reduction of postoperative spinal implant infection using gentamicin microspheres. Spine (Phila Pa 1976). 2009;34(5):479–83.
O'Neill KR, Smith JG, Abtahi AM, et al. Reduced surgical site infections in patients undergoing posterior spinal stabilization of traumatic injuries using vancomycin powder. Spine J. 2011;11(7):641–6.
Kirkland KB, Briggs JP, Trivette SL, Wilkinson WE, Sexton DJ. The impact of surgical-site infections in the 1990s: attributable mortality, excess length of hospitalization, and extra costs. Infect Control Hosp Epidemiol. 1990s;20(11):725–30.
de Lissovoy G, Fraeman K, Hutchins V, Murphy D, Song D, Vaughn BB. Surgical site infection: incidence and impact on hospital utilization and treatment costs. Am J Infect Control. 2009;37(5):387–97.
Maki DG, Schuna AA. A study of antimicrobial misuse in a university hospital. Am J Med Sci. 1978;275(3):271–82.
Larsen RA, Evans RS, Burke JP, Pestotnik SL, Gardner RM, Classen DC. Improved perioperative antibiotic use and reduced surgical wound infections through use of computer decision analysis. Infect Control Hosp Epidemiol. 1989;10(7):316–20.
Shapiro M, Townsend TR, Rosner B, Kass EH. Use of antimicrobial drugs in general hospitals: patterns of prophylaxis. N Engl J Med. 1979;301(7):351–5.
Hill AB. The environment and disease: association of causation? Proc R Soc Med. 1965;58:295–300.
Platt R. Methodologic aspects of clinical studies of perioperative antibiotic prophylaxis. Rev Infect Dis. 1991;13(Suppl 10):S810–S814814.
Acknowledgements
Funding
No funding or sponsorship was received for this study or publication of this article.
Authorship
All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article, take responsibility for the integrity of the work as a whole, and have given their approval for this version to be published.
Disclosures
Blaine T. Phillips, Emma S. Sheldon, Vwaire Orhurhu, Robert A. Ravinsky, Monika E. Freiser, Morteza Asgarzadeh, Omar Viswanath, and Marie Roguski have nothing to disclose. Alan D. Kaye is a member of the journal’s editorial board.
Compliance with Ethics Guidelines
This article is based on previously conducted studies and does not contain any studies with human participants or animals performed by any of the authors.
Data Availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Author information
Authors and Affiliations
Corresponding author
Additional information
Digital Features
To view digital features for this article go to https://doi.org/10.6084/m9.figshare.12205415.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Phillips, B.T., Sheldon, E.S., Orhurhu, V. et al. Preoperative Versus Extended Postoperative Antimicrobial Prophylaxis of Surgical Site Infection During Spinal Surgery: A Comprehensive Systematic Review and Meta-Analysis. Adv Ther 37, 2710–2733 (2020). https://doi.org/10.1007/s12325-020-01371-5
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12325-020-01371-5