Abstract
Chronic venous disease (CVD) is a prevalent condition that tends to worsen with age. Patients initially seek treatment to relieve symptoms of leg pain, discomfort, heaviness and swelling, all of which impact their quality of life. As the disease increases in severity to include varicose veins, skin changes, and venous ulcer, the demand for treatment increases while the quality of life further diminishes. The prevalence of CVD is highest in Western countries where it already consumes up to 2% of healthcare budgets. With the aging of the global population, the prevalences of CVD and severe CVD are projected to increase substantially, foretelling unsustainably large increases in the healthcare resources and costs needed to treat CVD patients in the coming decades. Effective venoactive drug treatments and ablation procedures are available that provide symptom relief, improve quality of life, slow disease progression, and promote ulcer healing. In addition, venoactive drug treatments may be highly cost-effective. However, there is evidence that physician awareness of CVD is suboptimal and that many patients with CVD are not being treated or referred to specialists according to established guidelines. To decrease this treatment gap and prevent unnecessary disease progression, international guidelines are available to help physicians consider CVD treatment options and refer patients when warranted. Improved disease awareness and appropriate early treatment may help reduce the coming burden of CVD.
Funding: Servier.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Patients with chronic venous disease (CVD) seek treatment for a variety of symptoms and signs that may substantially impact their quality of life (QoL). Symptoms include leg pain, discomfort, and heaviness, whereas the clinical signs of CVD are varicose veins (VVs), oedema, skin discolouration, lipodermatosclerosis, and, in severe cases, venous ulceration. Based on the presence of specific clinical signs, which may or may not be symptomatic but are associated with increasing clinical severity, CVD can be classified from C0 (no signs) to C6 (venous ulceration) (Table 1). Some of the principal issues facing the medical community are: why CVD develops, why it progresses in certain individuals but not in others, and how many people have CVD. In addition, despite the existence of national referral guidelines in the UK, and international guidelines for the diagnosis and treatment of CVD, the management of CVD is variable. A 2013 survey of general practitioners in the UK indicated that less than 50% were aware of referral guidelines for CVD patients and would refer patients with moderate to severe disease (C4 or C5) to a vein specialist, and only about 10% were aware of clinical venous scoring systems [1]. These findings indicate that a substantial knowledge gap needs to be bridged to ensure that CVD patients of all severities receive appropriate treatment according to international guidelines [2]. In addition, the National Institute for Health and Care Excellence (NICE) in the UK also provides comprehensive guidance for the diagnosis and management of varicose veins [3].
This article is based on previously conducted studies and does not contain any studies with human participants or animals performed by the author.
Epidemiology of CVD
Thirty years ago in an analysis of 3822 adults in the Framingham Study cohort, it was recognised that the incidence of VVs was higher in women than in men [4]. New VV case rates in the cohort over 2 years were 51.9 per 1000 women and 39.4 per 1000 men. More recent evidence supports these trends but also shows that the prevalence of CVD and VV vary widely by region, though they are highest in Western countries [5]. Risk factors confirmed in subsequent studies were: excess body mass index (BMI) and obesity, low physical activity, and high systolic blood pressure for both sexes; older age at menopause, and sedentary occupations for women; and smoking for men [4]. Following a systematic review performed 20 years after the Framingham Study, Robertson et al. proposed a variety of risk factors for primary CVD, including increasing age, positive family history, gender, pregnancy, obesity, reduced mobility at work, smoking, low fibre intake, and constipation [6].
Venous reflux is a known precursor for CVD. In the Edinburgh Vein Study cohort, which is a prospective random population-based cohort of 1566 adult patients, a follow-up at 13 years after baseline found the annual incidence of venous reflux to be 1% [7]. Risk factors associated with venous reflux were being overweight [odds ratio (OR) 2.1, 95% confidence interval (CI) 1.0–4.4] and a history of deep vein thrombosis (OR 11.3, 95% CI 1.0–132.3), whereas pregnancy and smoking were not significant risk factors. The 13-year incidence of VV was two-fold higher in participants who had venous reflux at baseline than in those who did not.
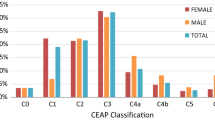
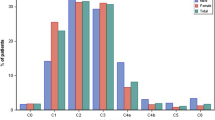
An epidemiological survey of 6009 adult patients (mean age 53.4 years) conducted by general practitioners in Belgium and Luxembourg found the overall prevalence of CVD (class C1–C6) to be high (61.3%) and underestimated by general practitioners, who considered that only 45.9% of these patients had CVD [8]. This study also reported the C-class to be significantly higher in women than in men, and that CVD severity was associated with high BMI, absence of regular exercise, number of pregnancies, a family history of CVD, and increasing age. QoL decreased with the appearance of the first symptoms of CVD and further decreased as the cumulative number of symptoms increased. CVD symptoms also led to losses in productivity as 10.4% of patients with CVD reported a loss of working days. This study was conducted as part of the Vein Consult Program, which is an extensive international prospective epidemiological survey supported by the Union Internationale de Phlebologie with the goal to improve understanding of the worldwide prevalence of CVD. The epidemiology of CVD has also been reported for a much larger international population of 91,545 adult patients in the Vein Consult Program cohort across diverse geographic regions, including Europe, Latin America, the Middle East, and the Far East [9]. Overall, 41.4% of these patients had early stage CVD (C0s–C1) and 84% had any class of CVD greater than C0s. These results confirm that CVD is a widespread global problem. It is also widely under-recognised because general practitioners considered that only 51% of the patients diagnosed with C0s–C1 patients had CVD. Although referrals to specialists tended to increase with CVD severity, referral rates by general practitioners varied by region and were lowest in the Far East.
As the age of the global population is increasing due to medical advances and longer lifespans, the numbers of patients with CVD are also growing. Barnes et al. reported that US outpatient visits for venous disease, including deep vein thrombosis, increased significantly from 4 million visits in 1997 to nearly 6 million visits in 2006, providing some evidence of the growing burden of venous disease, although some of this increase might be due to improvements in CVD patient management [10]. Disease severity and treatment requirements also increase with age. In a retrospective study of 38,750 CVD patients in the US, the initial presenting symptoms of pain, heaviness, fatigue, and aching were more common in patients under 65 years of age. The symptoms and signs of more severe CVD, such as swelling, skin discolouration, and venous ulceration, as well as venous ablations, were more common patients older than 65 [11].
A systematic review of epidemiological studies showed that the burden of VVs (57.1–90.4%) and treatment practices varies across European countries [12]. Comparisons of the predicted numbers of CVD patients (with CVD > C2) to the actual number of patients being treated for venous disease indicated that some countries are not treating all patients with C2-level disease, while others may be treating excessive numbers of patients. The numbers of procedures performed for VVs per million people across these countries differed by up to four-fold, indicating that there are marked treatment gaps in some countries.
The prevalence of CVD and other venous diseases are predicted to increase because the prevalence of the main risk factors for them, age and obesity, are increasing rapidly. In the UK, which is contributing to the current global obesity epidemic, the population of adults over age 85 has been predicted to grow to over 1.4 million men and 1.94 million women by 2033. In the US, the number of venous thromboembolism cases has been projected to increase from 1 million in 2010 to 1.8 million in 2050 [13], while VV procedures are projected to increase by over 60% in the US and Europe between 2013 and 2021 [14].
With the rising prevalence of venous diseases, QoL in an increasing number of patients can also be expected to decline. The QoL of patients with C6/venous ulceration has been reported to be comparable to that of patients with congestive heart failure or chronic lung disease, suggesting that, if CVD is allowed to progress to severe stages, the QoL of CVD patients can become very poor [15]. Indeed, QoL is lower in patients with recurrent VVs than in the general population, and patients with recurrent VVs scored lower than normal in all eight scales of the short-form health survey (SF-36) [16]. However, interventions to treat VVs have been shown to significantly and substantially improve QoL in these patients [17,18,19].
CVD Progression
Patients with CVD are prone to disease progression, in which patients progress to a higher clinical class over time or develop venous reflux and/or VVs in veins not previously affected. Disease progression is distinct from treatment failure, neovascularisation following surgical removal of VVs, or incomplete removal of varicosities. Progression can occur despite treatment or surgical removal of afflicted veins [20].
Annual progression rates of approximately 4% have been reported for the Edinburgh Vein Study, the Bonn Vein Study, and from reviews of other epidemiological studies [21,22,23]. In the Edinburgh Vein Study, the overall progression rate was 58% after a follow-up of 13 years [21]. The main risk factors for progression in patients with VVs at baseline were an age greater than 55 years (OR 3.9, 95% CI 1.1–14.3), being overweight/obese (BMI ≥ 25; OR 1.9, 95% CI 1.1–3.1), and a family history of VV (OR 1.9, 95% CI 1.20–3.04) [21]. Additional risk factors included female gender and superficial venous reflux.
Following endovenous ablation of VVs, 15–47% of patients experience clinical recurrence, with a new area of truncal reflux occurring in 16% of patients as a result of disease progression [24]. In another study, new truncal reflux developed in 16–24% of patients after thermal ablation [25]. Recurrence can arise from neovascularisation, disease progression and incomplete ablation, either alone or in combination [20].
Treatments
Pharmacological treatment can improve CVD symptoms and QoL [2, 26, 27]. Among the venoactive drugs available, Daflon® (micronised purified flavonoid fraction, MPFF) has been the most widely studied and has accumulated the most evidence for its efficacy [28,29,30,31,32,33,34]. In one recent study, MPFF treatment reduced the frequency of night cramps, pain, leg heaviness, and situational (evening) reflux in the great saphenous vein [34]. Other venoactive drugs may also be effective for specific applications [26]. Pentoxifylline is a useful adjunct to compression therapy for patients with venous ulcers; oxerutins and calcium dobesilate may help reduce oedema; and rutosides may provide symptom relief for varicose veins in pregnancy. However, in a recent update of treatment guidelines, Daflon/MPFF was the only venoactive drug to receive strong recommendations for relief of CVD symptoms and for venous ulcer healing [2]. Physicians should refer to these guidelines to identify the most appropriate pharmacological treatments for their CVD patients.
Clearly, CVD is a complex chronic disease that will progress and substantially impact patient QoL if improperly treated. For this reason, perhaps the key recommendation for general practitioners is to refer to a vascular service specialist those patients who present with symptomatic or recurrent varicose veins or with lower limb skin changes (e.g. pigmentation or eczema) assumed to be caused by chronic venous insufficiency. Additionally, the CEAP classification is not recommended for referral guidance in the UK by NICE, as patients may not be referred despite having symptoms due to C2-level disease.
Treatment options have improved in recent years, and include venoactive drug therapy, surgery, and endovenous ablation of VVs using lasers, radiofrequency, or sclerotherapy. There is general agreement among physicians in the US and Europe that all of these strategies are more effective than compression alone [3, 35, 36]. In addition, virtually all of these strategies have been shown to be cost-effective [37,38,39,40].
CVD is a Growing Economic Burden
The Millenium Research Group has estimated that, by 2021, VV treatments will have increased twofold over 2011 levels [41]. At US$2000 per treatment, total costs have been projected to reach US$8 trillion by 2021. Further, 1 in 65 people have a VV treatment every year and, with life expectancies increasing, this means that virtually every person who attains the age of 80–85 years will have had at least one vein treatment.
The global varicose vein treatment market, which was US$290.59 million in 2016, has been estimated to reach over US$396 million by 2021. Currently, care and treatment for CVD patients consumes 2% of national healthcare budgets [42, 43]. Treatments and care strategies that can prevent or slow disease progression, or VV recurrence, are likely to reduce these substantial costs.
Venous Leg Ulcers
Currently, venous leg ulcers affect 1% of the adult population [44]. In the US, accumulated annual costs of prescription drugs, emergency department visits, home healthcare, outpatient visits, and hospitalisations in the 12 months after initial ulceration have been estimated to be US$2611 for Medicare-insured patients and US$3066 for privately-insured patients [44]. Total costs were estimated to reach US$15 billion annually [44].
Treatments that speed ulcer healing could help reduce these costs. As an example, adding pentoxifylline treatment to compression therapy significantly improves venous ulcer healing rates versus compression therapy alone or if combined with placebo, and also reduces treatment costs [45]. A cost-effectiveness analysis of this adjunct pentoxifylline treatment, which costs GB£6 per month, suggested that the combined treatment costs only GB£98 (95% CI − 49.2 to 245.0) per quality-adjusted life year (QALY) gained [45]. As a point of reference, the cost-effectiveness threshold in the UK is considered to be GB£20,000 per QALY gained, so adjunct pentoxifylline therapy is likely to be extremely cost-effective. Even with rising costs of pharmaceuticals, any venoactive drug that significantly reduces ulcer healing time is likely to be cost-effective.
In a contrasting example, surgical treatments for post-thrombotic syndrome, such as deep vein angioplasty and stents, are quite expensive but there is little evidence from randomised controlled clinical trials to show that they are effective. Thus, the use of such treatments should be evaluated with caution.
Conclusions
The burdens of VV, CVD, and venous ulcers are great for patients and healthcare systems. Because these conditions are complex, are prone to worsening, and can substantially impact patient QoL, it is critical that patients receive appropriate treatment to prevent disease progression and recurrence. Thus, referral of these patients to a specialist may be warranted. Further, it is important for physicians to consider the options and costs of available CVD treatments. Pharmacological treatments with venoactive drugs alone, or as adjuncts to compression therapy, offer patient benefits that are likely to be cost-effective. However, more cost–benefit studies are needed, as treatments not proven to be cost-effective will not be available for reimbursement.
References
Lane TR, Sritharan K, Herbert JR, Franklin IJ, Davies AH. Management of chronic venous disease by primary care. Phlebology. 2013;28(6):299–304 (Epub 2012/09/20).
Nicolaides A, Kakkos S, Baekgaard N, Comerota A, de Maesenner M, Eklof B, et al. Management of chronic venous disorders of the lower limbs. Guidelines According to Scientific Evidence. Part I. Int Angiol. 2018;37(3):181–254 (Epub 2018/06/07).
Marsden G, Perry M, Kelley K, Davies AH. Diagnosis and management of varicose veins in the legs: summary of NICE guidance. BMJ. 2013;347. https://doi.org/10.1136/bmj.f4279.
Brand FN, Dannenberg AL, Abbott RD, Kannel WB. The epidemiology of varicose veins: the Framingham Study. Am J Prev Med. 1988;4(2):96–101 (Epub 1988/03/01).
Beebe-Dimmer JL, Pfeifer JR, Engle JS, Schottenfeld D. The epidemiology of chronic venous insufficiency and varicose veins. Ann Epidemiol. 2005;15(3):175–84 (Epub 2005/02/23).
Robertson L, Evans C, Fowkes FG. Epidemiology of chronic venous disease. Phlebology. 2008;23(3):103–11 (Epub 2008/05/10).
Robertson LA, Evans CJ, Lee AJ, Allan PL, Ruckley CV, Fowkes FG. Incidence and risk factors for venous reflux in the general population: Edinburgh Vein Study. Eur J Vasc Endovasc Surg. 2014;48(2):208–14 (Epub 2014/06/22).
Vuylsteke ME, Thomis S, Guillaume G, Modliszewski ML, Weides N, Staelens I. Epidemiological study on chronic venous disease in Belgium and Luxembourg: prevalence, risk factors, and symptomatology. Eur J Vasc Endovasc Surg. 2015;49(4):432–9 (Epub 2015/02/24).
Rabe E, Guex JJ, Puskas A, Scuderi A, Fernandez Quesada F. Epidemiology of chronic venous disorders in geographically diverse populations: results from the Vein Consult Program. Int Angiol. 2012;31(2):105–15 (Epub 2012/04/03).
Barnes GD, Gafoor S, Wakefield T, Upchurch GR Jr, Henke P, Froehlich JB. National trends in venous disease. J Vasc Surg. 2010;51(6):1467–73 (Epub 2010/03/23).
Pappas PJ, Lakhanpal S, Nguyen KQ, Vanjara R. The Center for Vein Restoration Study on presenting symptoms, treatment modalities, and outcomes in Medicare-eligible patients with chronic venous disorders. J Vasc Surg Venous Lymphat Disord. 2018;6(1):13–24 (Epub 2017/12/19).
Moore HM, Lane TR, Thapar A, Franklin IJ, Davies AH. The European burden of primary varicose veins. Phlebology. 2013;28(Suppl 1):141–7 (Epub 2013/03/27).
Deitelzweig SB, Johnson BH, Lin J, Schulman KL. Prevalence of clinical venous thromboembolism in the USA: current trends and future projections. Am J Hematol. 2011;86(2):217–20 (Epub 2011/01/26).
Onida S, Davies AH. Predicted burden of venous disease. Phlebology. 2016;31(1 Suppl):74–9 (Epub 2016/02/27).
Carradice D, Mazari FA, Samuel N, Allgar V, Hatfield J, Chetter IC. Modelling the effect of venous disease on quality of life. Br J Surg. 2011;98(8):1089–98 (Epub 2011/05/24).
Smith JJ, Garratt AM, Guest M, Greenhalgh RM, Davies AH. Evaluating and improving health-related quality of life in patients with varicose veins. J Vasc Surg. 1999;30(4):710–9 (Epub 1999/10/08).
Shepherd AC, Gohel MS, Lim CS, Davies AH. A study to compare disease-specific quality of life with clinical anatomical and hemodynamic assessments in patients with varicose veins. J Vasc Surg. 2011;53(2):374–82 (Epub 2010/12/07).
Carroll C, Hummel S, Leaviss J, Ren S, Stevens JW, Everson-Hock E, et al. Clinical effectiveness and cost-effectiveness of minimally invasive techniques to manage varicose veins: a systematic review and economic evaluation. Health Technol Assess. 2013;17(48):i–xvi, 1–141 (Epub 2013/11/02).
Brittenden J, Cotton SC, Elders A, Ramsay CR, Norrie J, Burr J, et al. A randomized trial comparing treatments for varicose veins. N Engl J Med. 2014;371(13):1218–27 (Epub 2014/09/25).
Kostas T, Ioannou CV, Touloupakis E, Daskalaki E, Giannoukas AD, Tsetis D, et al. Recurrent varicose veins after surgery: a new appraisal of a common and complex problem in vascular surgery. Eur J Vasc Endovasc Surg. 2004;27(3):275–82 (Epub 2004/02/05).
Lee AJ, Robertson LA, Boghossian SM, Allan PL, Ruckley CV, Fowkes FG, et al. Progression of varicose veins and chronic venous insufficiency in the general population in the Edinburgh Vein Study. J Vasc Surg Venous Lymphat Disord. 2015;3(1):18–26 (Epub 2015/01/01).
Pannier F, Rabe E. The relevance of the natural history of varicose veins and refunded care. Phlebology. 2012;27(Suppl 1):23–6 (Epub 2012/02/15).
Pannier F, Rabe E. Progression in venous pathology. Phlebology. 2015;30(1 Suppl):95–7 (Epub 2015/03/03).
O’Donnell TF, Balk EM, Dermody M, Tangney E, Iafrati MD. Recurrence of varicose veins after endovenous ablation of the great saphenous vein in randomized trials. J Vasc Surg Venous Lymphat Disord. 2016;4(1):97–105 (Epub 2016/03/08).
Bush RG, Bush P, Flanagan J, Fritz R, Gueldner T, Koziarski J, et al. Factors associated with recurrence of varicose veins after thermal ablation: results of the recurrent veins after thermal ablation study. Sci World J. 2014;2014:505843 (Epub 2014/03/05).
Gohel MS, Davies AH. Pharmacological agents in the treatment of venous disease: an update of the available evidence. Curr Vasc Pharmacol. 2009;7(3):303–8 (Epub 2009/07/16).
Martinez-Zapata MJ, Vernooij RW, Uriona Tuma SM, Stein AT, Moreno RM, Vargas E, et al. Phlebotonics for venous insufficiency. Cochrane Database Syst Rev. 2016;4:CD003229 (Epub 2016/04/07).
Bush R, Comerota A, Meissner M, Raffetto JD, Hahn SR, Freeman K. Recommendations for the medical management of chronic venous disease: The role of Micronized Purified Flavanoid Fraction (MPFF). Phlebology. 2017;32(1_suppl):3–19 (Epub 2017/02/18).
Carpentier P, van Bellen B, Karetova D, Hanafiah H, Enriquez-Vega E, Kirienko A, et al. Clinical efficacy and safety of a new 1000-mg suspension versus twice-daily 500-mg tablets of MPFF in patients with symptomatic chronic venous disorders: a randomized controlled trial. Int Angiol. 2017;36(5):402–9 (Epub 2017/02/17).
das Gracas CdSM, Cyrino FZ, de Carvalho JJ, Blanc-Guillemaud V, Bouskela E. Protective effects of micronized purified flavonoid fraction (MPFF) on a novel experimental model of chronic venous hypertension. Eur J Vasc Endovasc Surg. 2018;55(5):694–702 (Epub 2018/03/29).
de Souza M, Cyrino FZ, Mayall MR, Virgini-Magalhaes CE, Sicuro FL, de Carvalho JJ, et al. Beneficial effects of the micronized purified flavonoid fraction (MPFF, Daflon(R) 500 mg) on microvascular damage elicited by sclerotherapy. Phlebology. 2016;31(1):50–6 (Epub 2014/12/18).
Kakkos SK, Nicolaides AN. Efficacy of micronized purified flavonoid fraction (Daflon(R)) on improving individual symptoms, signs and quality of life in patients with chronic venous disease: a systematic review and meta-analysis of randomized double-blind placebo-controlled trials. Int Angiol. 2018;37(2):143–54 (Epub 2018/02/02).
Katsenis K. Micronized purified flavonoid fraction (MPFF): a review of its pharmacological effects, therapeutic efficacy and benefits in the management of chronic venous insufficiency. Curr Vasc Pharmacol. 2005;3(1):1–9 (Epub 2005/01/12).
Tsukanov YT, Tsukanov AY. Diagnosis and treatment of situational great saphenous vein reflux in daily medical practice. Phlebolymphology. 2017;24(3):144–51.
Gloviczki P, Comerota AJ, Dalsing MC, Eklof BG, Gillespie DL, Gloviczki ML, et al. The care of patients with varicose veins and associated chronic venous diseases: clinical practice guidelines of the Society for Vascular Surgery and the American Venous Forum. J Vasc Surg. 2011;53(5 Suppl):2S–48S (Epub 2011/05/06).
Wittens C, Davies AH, Baekgaard N, Broholm R, Cavezzi A, Chastanet S, et al. Editor’s choice—management of chronic venous disease: clinical practice guidelines of the European Society for Vascular Surgery (ESVS). Eur J Vasc Endovasc Surg. 2015;49(6):678–737 (Epub 2015/04/30).
Marsden G, Perry M, Bradbury A, Hickey N, Kelley K, Trender H, et al. A cost-effectiveness analysis of surgery, endothermal ablation, ultrasound-guided foam sclerotherapy and compression stockings for symptomatic varicose veins. Eur J Vasc Endovasc Surg. 2015;50(6):794–801 (Epub 2015/10/05).
Shepherd AC, Ortega-Ortega M, Gohel MS, Epstein D, Brown LC, Davies AH. Cost-effectiveness of radiofrequency ablation versus laser for varicose veins. Int J Technol Assess Health Care. 2015;31(5):289–96 (Epub 2015/12/31).
Gohel MS, Epstein DM, Davies AH. Cost-effectiveness of traditional and endovenous treatments for varicose veins. Br J Surg. 2010;97(12):1815–23 (Epub 2010/10/06).
Tassie E, Scotland G, Brittenden J, Cotton SC, Elders A, Campbell MK, et al. Cost-effectiveness of ultrasound-guided foam sclerotherapy, endovenous laser ablation or surgery as treatment for primary varicose veins from the randomized CLASS trial. Br J Surg. 2014;101(12):1532–40 (Epub 2014/10/03).
Millenium Research Group—DRG. Varicose Vein Treatment Devices|US|2015| Market Analysis. Global Information, Inc.; 2015 [cited 2018]; Available from: https://www.giiresearch.com/report/mrg202059-us-markets-varicose-vein-treatment-devices-2011.html. Accessed 15 Dec 2018.
Laing W. Chronic venous diseases of the leg. London: Office of Health Economics; 1992.
Rabe E, Pannier F. Societal costs of chronic venous disease in CEAP C4, C5, C6 disease. Phlebology. 2010;25(Suppl 1):64–7 (Epub 2010/10/05).
Rice JB, Desai U, Cummings AK, Birnbaum HG, Skornicki M, Parsons N. Burden of venous leg ulcers in the United States. J Med Econ. 2014;17(5):347–56 (Epub 2014/03/15).
Varatharajan L, Thapar A, Lane T, Munster AB, Davies AH. Pharmacological adjuncts for chronic venous ulcer healing: a systematic review. Phlebology. 2016;31(5):356–65 (Epub 2015/06/04).
Acknowledgements
Funding
This supplement has been sponsored by Servier. The article processing charges and the open access fee for this article were funded by Servier. The author had full access to all of the data for this study and takes complete responsibility for the integrity of the data and accuracy of the data analysis.
Medical Writing
Medical writing services were provided by Dr Kurt Liittschwager (4Clinics, France) and funded by Servier.
Authorship
Alun H. Davies meets the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article, takes responsibility for the integrity of the work as a whole, and has approved this version for publication.
Prior Presentation
This article and all of the articles in this supplement are based on the International satellite symposium at European Venous Forum, June 29th, Athens.
Disclosures
Alun H. Davies received honorarium from Servier for the symposium lecture. The symposium was funded by an educational grant from Servier.
Compliance with Ethics Guidelines
This article is based on previously conducted studies and does not contain any studies with human participants or animals performed by the author.
Author information
Authors and Affiliations
Corresponding author
Additional information
Enhanced Digital Features
To view enhanced digital features for this article go to https://doi.org/10.6084/m9.figshare.7599155.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/), which permits any noncommercial use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Davies, A.H. The Seriousness of Chronic Venous Disease: A Review of Real-World Evidence. Adv Ther 36 (Suppl 1), 5–12 (2019). https://doi.org/10.1007/s12325-019-0881-7
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12325-019-0881-7