Abstract
Introduction
Allergic asthma is a chronic inflammatory disease caused by immunoglobulin E (IgE)-mediated allergy. Omalizumab is a monoclonal anti-IgE antibody for the treatment of severe allergic asthma (SAA).
Objectives
The primary objective of the study was to assess asthma-related control in patients with SAA receiving omalizumab therapy. Secondary objectives included quality of life, treatment effectiveness, rate of severe exacerbations, and safety.
Methods
This was a prospective, multi-centre, non-interventional study to assess patient-related long-term outcomes of omalizumab treatment in Germany. This 3-year study enrolled patients aged ≥ 18 years with SAA. Asthma control was assessed using the asthma control questionnaire (ACQ-6) and physician-assessed global evaluation of treatment effectiveness (GETE). Exacerbations were recorded, and quality of life was assessed using the mini-asthma quality of life questionnaire (mini-AQLQ).
Results
Of 161 patients screened, 153 participated in this study. Most patients (92.2%) had been receiving prior omalizumab therapy for mean (SD) 2.9 (2.3) years. Omalizumab slightly decreased mean ACQ-6 score from 2.0 (1.22) at baseline to 1.7 (1.23) at the end of the 3-year treatment period [difference: –0.18 (1.07), P = 0.340]. Post-hoc analyses of ACQ-6 for the small number of treatment-naïve patients showed a decrease in mean (SD) ACQ-6 from 2.7 (1.08) at baseline to 1.4 (1.40) after 3 years of omalizumab treatment. Mini-AQLQ increased from 4.5 (1.26) at baseline to 4.7 (1.48) after 3 years [difference: 0.26 (1.35), P = 0.186]. GETE was reported as excellent or good for most patients (67.46–84.69%) and more than two-thirds had no severe exacerbation. There were no unexpected safety signals during the study period and no tachyphylaxis was observed.
Conclusions
In conclusion, despite most patients receiving prior omalizumab treatment for approximately 3 years, there was no decrease in effectiveness or safety over the subsequent 3 years during this study. This supports the long-term use of omalizumab in maintaining asthma control and quality of life.
Funding
Novartis Pharma GmbH, Germany.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Omalizumab is a monoclonal anti-IgE antibody approved for treatment of severe allergic asthma. |
This real-life observational study showed that effectiveness of omalizumab in maintaining control of asthma and improving quality of life did not lessen over time. |
No new safety issues were observed over 3 years of treatment. |
Introduction
Asthma is one of the most prevalent chronic respiratory diseases, affecting approximately 358 million people globally [1]. According to the Global Initiative for Asthma (GINA) strategy [2], the overarching long-term goal of asthma management is to achieve control of the condition. Reducing future risk of exacerbation and reducing or withholding oral corticosteroid (OCS) dose to avoid or minimise side effects are also important goals in asthma management [2].
Despite the availability of treatment recommendations and standard-of-care therapies [2], it is estimated that approximately 5–10% of asthma patients have severe disease, and over half of patients with severe asthma have uncontrolled disease [3].
Omalizumab is an anti-immunoglobulin E (IgE) monoclonal antibody approved in the European Union (EU) as an add-on therapy to improve asthma control in patients aged ≥ 6 years with severe persistent allergic asthma [4]. The efficacy of omalizumab in improving asthma control has been documented in several studies [5, 6], including the 5-year Evaluating Clinical Effectiveness and Long-term Safety (EXCELS) in patients with moderate-to-severe asthma observational study [7].
Asthma control can be measured by means of the asthma control questionnaire (ACQ)-6, which is a shortened, six-item variant of the ACQ proper, but excludes the lung function assessment domain. Importantly, this shortened version has been fully validated and achieves similar results to the full ACQ [8].
The aim of this observational study was to further investigate the effectiveness and safety of long-term omalizumab therapy on asthma control and quality of life of patients with severe allergic asthma under real-life conditions. This was done using two questionnaires, the ACQ-6 and the mini-asthma quality of life questionnaire (AQLQ), respectively. Owing to the real-life aspect of this study, previous exposure to omalizumab was permitted. Furthermore, exacerbations and safety aspects were documented.
Methods
Study Design
This was a prospective, multi-centre, non-interventional study of omalizumab carried out in Germany between May 2013 and March 2017. The scheduled observation period was up to 3 years. Omalizumab injections were envisaged every 2–4 weeks and medical findings and questionnaire records were to be documented at inclusion, during follow-up visits (every 2–4 weeks, every 3 months, and every year), and after 3 years. The primary objective of the study was to assess asthma-related control using the ACQ-6 in patients with severe allergic asthma. Secondary objectives included assessment of quality of life, treatment effectiveness of omalizumab, rate of severe exacerbations, and safety.
This study received approval from the ethics committee of the Otto von Guericke University of Magdeburg, Germany (Ethics Approval Number: 07/13). All patients provided written informed consent for inclusion in this study. As this was a non-interventional study, trial registration was not required.
Participants
Patients ≥ 18 years with severe allergic asthma were eligible for inclusion in this study if they had been previously treated with high-dose ICS and long-acting β2-agonists (LABA). Both patients who were previously treated with omalizumab and newly initiated patients were eligible. Patients with contraindications to the omalizumab SmPC [4] or a history of lung disease other than allergic asthma were excluded from this study. Further eligibility criteria are outlined in the Supplementary Information.
Study Assessments
Effectiveness of omalizumab was assessed (1) using the ACQ-6, (2) by global evaluation of treatment effectiveness (GETE), and (3) by monitoring exacerbations every 2–4 weeks. ACQ-6 total scores were defined as: < 0.75: asthma controlled adequately; 0.75–1.50: threshold range (undecided); > 1.50: asthma not controlled adequately. A decrease of > 0.5 from baseline ACQ-6 was considered a clinically relevant improvement. An exacerbation was defined as administration of an OCS course, an unscheduled visit to the physician, a hospitalisation, or emergency treatment because of asthma deterioration, or decrease of FEV1 < 60% from the personal best value. Quality of life data were captured every 3 months using the mini-AQLQ, which was completed by the patients.
Safety
Adverse events (AE) and serious AEs (SAE) were recorded during the course of this study. Adverse events were further categorised according to relationship with omalizumab as follows: non-serious AE or serious AE with no causality with omalizumab; non-serious adverse drug reaction or serious adverse drug reaction with causality with omalizumab.
Statistical Analysis
The analyses presented in this manuscript were conducted in the Full Analysis Set (FAS), except for the incidence of AEs, which was reported in the Safety Analysis Set. A Wilcoxon signed rank test was used to test the pre- and post-treatment differences in ACQ-6 and mini-AQLQ scores for differences from zero. For all other analyses, epidemiological and descriptive statistics were employed. For most parameters, mean ± standard deviation (SD) are reported, unless otherwise stated. To assess differences between treatment-naïve and treatment-experienced patients, a post hoc analysis of ACQ-6 and mini-AQLQ was conducted in the FAS. The FAS was stratified into two groups according to duration of omalizumab therapy prior to the start of the study. Of note, the stratification of “treatment-naïve patients with ≤ 4 months of omalizumab therapy prior to study start” versus “treatment-experienced patients with > 4 months of omalizumab therapy prior to study start” was chosen because in clinical practice, the first evaluation of omalizumab treatment efficacy is recommended after 16 weeks (i.e. 4 months) of treatment [9]. Statistical analyses were conducted by Novartis Pharma GmbH (Germany) and Kantar Health GmbH (Germany) using SAS software version 9.4.
Results
Participants
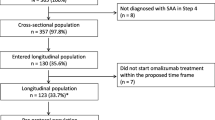
Of 161 severe allergic asthma patients screened, all 161 (100%) patients were included in the SAF. Of these, eight (5%) patients were excluded from the FAS (n = 153) for the reasons listed in Fig. 1. All patients with their last documented visit at the first annual visit or later were included in the FAS1Y (n = 118, 73.3%). Similarly, all patients with their last documented visit at the second annual visit or later were included in the FAS2Y (n = 107, 66.5%). This also demonstrates the remarkably low discontinuation rate in this study. Patients with a premature final visit, no documented final visit, or an observation period ≤ 2.74 years were included in the Safety Dropout Set (SDS; n = 63, 39.1%). The mean observation period (SD) was 901.6 (367.0) days. The n numbers for each individual analysis are inconsistent because not all variables could be collected at all time points for every patient. The median (minimum, maximum) age of patients in the FAS was 50 (18, 76) years, with no patients < 18 years old (Table 1). Most patients were female (n = 93; 60.8%) and lifelong non-smokers (n = 109; 71.24%). Mean (SD) forced expiratory volume in 1 s (FEV1) was 70.1% (20.43) predicted of normal at baseline. The majority of patients (n = 141; 92.2%) had previous exposure to omalizumab and had been receiving omalizumab therapy for a mean (SD) of 2.9 (2.3) years prior to study start. The mean (SD) time elapsed since diagnosis of asthma was 15.5 (12.2) years.
Participant enrolment. All patients who signed informed consent and had at least one visit after the baseline visit were included in the SAF. COPD chronic obstructive pulmonary disease, FAS full analysis set, FAS1Y 1 year complete FAS, FAS2Y 2 years complete FAS, SAF safety analysis set, SDS study dropout set
Primary End Point: Effect of Omalizumab Treatment on Patient-Reported Asthma Control
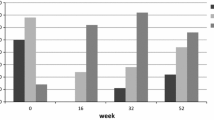
After 3 years of omalizumab treatment, the mean (SD) ACQ-6 total score decreased numerically from 2.0 (1.22) at baseline to 1.7 (1.23) at the end of the observation period, representing a change from baseline of − 0.18 (1.07) (P = 0.340). The greatest improvement in ACQ-6 score was seen at the 0.5-year and 1-year time points, where respective mean decreases of − 0.2 (1.08) (P = 0.087) and − 0.2 (1.20) (P = 0.102) in total score compared with baseline were recorded (Fig. 2, Table E1). An overview of the patient responses to items 1–6 in the ACQ-6 is available in Table E2 of the Supplementary Information.
Secondary End Points
Mini-AQLQ
Mini-AQLQ total score was significantly improved with omalizumab treatment compared with baseline at 0.5 years (P = 0.002), 1 year (P = 0.001), 1.5 years (P = 0.009), and 2 years (P = 0.011) (Table E3). Overall, this measure increased from 4.5 (1.26) at baseline to 4.7 (1.48) after 3 years (difference: 0.26 [1.35], P = 0.186). The effect of omalizumab on individual domains of the mini-AQLQ remained relatively stable across all time points. Symptoms were significantly improved at all time points with omalizumab treatment compared with baseline. Patient-reported emotional function, environmental stimuli, and activity limitation were also improved at various time points in response to omalizumab treatment (Fig. 3).
Mini-AQLQ total and domain scores over time (FAS, n = 153). Data shown are mini-AQLQ total score and domain scores (SD) recorded by patients at baseline and at half-yearly observation windows throughout the study. Statistical significance compared with baseline (P < 0.05) is denoted by *. FAS full analysis set, mini-AQLQ mini-asthma quality of life questionnaire, SD standard deviation
GETE
The majority of patients had GETE assessed as either “excellent” or “good” at all time points (67.46–84.69%). The proportion of patients with excellent GETE (i.e. total control of asthma) slightly increased from 24.56% (n = 28 of 114) at the 0.5-year visit to 28.57% (n = 36 of 126) at 3 years (Table 2). No GETE assessment reported worsening of asthma for any patient at any time point in this study.
Post-hoc analyses revealed a difference in the proportion of people with excellent, good, or moderate GETE at the end of the study between patients who had previous exposure to therapy for at least 4 months prior to the study start (treatment-experienced, n = 106) and those who did not (treatment-naïve, n = 18), respectively. By the end of this study, excellent GETE was reported for 27.36% (n = 29 of 106) and 33.33% (n = 6 of 18) of treatment-experienced and treatment-naïve patients, respectively. Good GETE (i.e. significant improvement in asthma) was reported for 43.40% (n = 46 of 106) and 11.11% (n = 2 of 18) of treatment-experienced and treatment-naïve patients, respectively (Table E4).
Severe exacerbations
The proportion of patients experiencing severe exacerbations remained low and stable throughout this study; 12.42% (n = 19 of 153), 7.87% (n = 10 of 127), and 11.97% (n = 14 of 117) of patients had ≥ 2 severe exacerbations in the first, second, and third years of observation, respectively (Figure E2). The proportion of patients experiencing ≥ 2 exacerbations during the winter months (October–March) decreased from 9.45% (n = 12 of 127) in the first year to 5.13% (n = 6 of 117) and 0.00% (n = 0 of 63) in the second and third year of observation, respectively. The number of hospitalisations due to severe exacerbations remained very low; three patients were hospitalised more than once for this reason during this study.
The shift in proportion of patients experiencing 0, 1, or ≥ 2 exacerbations in the first versus third year of the study is depicted in Fig. 4.
Number of severe exacerbations in first year versus third year of the study (FAS, n = 153). The percentage of patients who experienced 0 (n = 100), 1 (n = 23), or ≥ 2 (n = 19) severe exacerbations in the first year of the study are shown in the pink column. The subset of these patients who went on to experience 0, 1, or ≥ 2 severe exacerbations in the final (third) year of the study are also shown
Importantly, acceptance of omalizumab therapy, assessed by the physician, was high and did not significantly change over the course of the study (Table E5).
Post-Hoc Analysis of the Primary and Secondary End Points
Post-hoc analysis of the ACQ-6 results stratified by previous exposure to omalizumab, smoking history, gender, and age confirmed the results of the full analysis set for all subgroups (Table E6). Subgroup analyses explored ACQ-6 and mini-AQLQ results between treatment-experienced (n = 141 of 153, 92.2%) and treatment-naïve (n = 12 of 153, 7.8%) patients who did and did not receive omalizumab therapy for > 4 months before study entry, respectively.
Post-hoc analyses of the ACQ-6 results showed improvements for treatment-naïve patients with a decrease in mean (SD) ACQ-6 from 2.7 (1.08) at baseline to 1.4 (1.40) after 3 years of omalizumab treatment. Treatment-experienced patients (with more than 4 months’ exposure to omalizumab prior to study start) had better baseline ACQ-6 scores than the treatment-naïve patients and retained good asthma control over the course of the study [mean (SD) ACQ-6: 1.8 (1.20) at baseline and 1.7 (1.22) at 3 years].
As the first evaluation of omalizumab treatment is recommended at 16 weeks [9], we performed a post hoc analysis of ACQ-6 and mini-AQLQ in treatment-naïve patients only at the time point closest to this recommendation, i.e., at 0.5 years. For these patients, improvements from baseline were observed for ACQ-6 and mini-AQLQ at the 0.5-year observation. Mean ACQ-6 reduced from 2.7 (1.08) at baseline to 2.0 (1.56) after 0.5 years of omalizumab treatment. Similarly, mean mini-AQLQ increased from 4.0 (1.03) at baseline to 4.7 (1.25) at 0.5 years.
Smoking status affected the extent of asthma control improvement achieved in this study. Smokers and ex-smokers achieved greater improvements in asthma control over the course of this study than non-smokers. In smokers and ex-smokers, ACQ-6 improved from 2.2 (1.31) at baseline to 1.6 (1.02) at the 3-year observation. In non-smokers, level of asthma control was better than for smokers at the beginning of the study and was retained for the duration of the study: 1.9 (1.19) at baseline to 1.7 (1.31) at the 3-year time point. More discreet differences were observed when the effect of gender and age on asthma control was investigated.
Post-hoc analyses of mini-AQLQ results stratified by previous exposure to omalizumab, smoking history, gender, and age confirmed the results of the full analysis set for all subgroups (Table E7). Similar results were observed for mini-AQLQ. Treatment-naïve patients demonstrated a greater increase from baseline in mini-AQLQ total score compared with treatment-experienced patients. Mini-AQLQ increased from 4.0 (1.03) at baseline to 5.0 (1.51) in treatment-naïve patients at the end of the study. Baseline quality of life was better and remained stable in the treatment-experienced patients: mini-AQLQ score was 4.6 (1.26) at baseline and 4.7 (1.49) at the end of the study. Greater increases from baseline were observed in mini-AQLQ in smokers and ex-smokers, and males compared with non-smokers and females, respectively.
Due to the limited number of patients in these post-hoc analyses, the differences did not reach statistical significance.
Safety
A total of 128 of 161 patients (79.5%) reported at least one adverse event over the course of this study. The most frequent AEs classified by MedDRA terminology were infections and infestations (28.5% of AEs; n = 189 of 663) and respiratory, thoracic, or mediastinal disorders (24.8% of AEs; n = 164 of 662).
Adverse events were further categorised according to relationship to omalizumab. Causality with omalizumab therapy was not suspected in the majority (89.8%; n = 946 of 1053) of adverse events. Overall, of 161 patients, 119 (73.9%) and 60 (37.3%) experienced a non-serious AE or serious AE, respectively, not related to the study drug. This is compared with 34 (21.1%) and 21 (13.0%) patients who experienced a non-serious adverse drug reaction or serious adverse drug reaction related to omalizumab, respectively. A complete list of reported adverse events and their relationship to the study drug is available in the Supplementary Information (Table E8). One patient died during this study, but the fatal event was not deemed by the investigator to be related to the study drug.
Discussion
Despite numerous randomised clinical trials demonstrating the efficacy of omalizumab over the last 2 decades, data supporting the effectiveness of anti-IgE therapy in real-world clinical settings have been relatively lacking. However, recent data from the real-life Prospective Observational Study to Evaluate Predictors of Clinical Effectiveness in Response to Omalizumab (PROSPERO) study have provided some promising results in which omalizumab reduced exacerbation rates and hospitalisations and improved Asthma Control Test scores over a 42-week period compared with pre-treatment [10].
The study presented herein further builds on these results. This was an observational study carried out in Germany over a 3-year period, which assessed the effectiveness of omalizumab on asthma control and quality of life in patients with severe allergic asthma. By means of the ACQ-6, asthma control with omalizumab was shown to be stable, and there was no significant loss of efficacy of this biological therapy over time. Of note, these results are in line with those previously reported by Braunstahl and colleagues, measured over a shorter period of time [11].
While neither statistical significance nor the minimal clinically important difference in ACQ-6 of 0.5 was reached in this study, it is important to consider that the majority of patients were treated with omalizumab before the start of the study, making it difficult to observe meaningful differences from baseline over the study course. Furthermore, according to the SmPC [4], omalizumab is effective within 12–16 weeks of treatment initiation; therefore, significant differences from baseline at half-yearly observations over 3 years were not necessarily expected.
Interestingly, the post-hoc analysis performed on ACQ-6 results between treatment-naïve and treatment-experienced patients indicated that treatment-naïve patients experienced improvements in control as the study progressed, although statistical significance could not be demonstrated because of the low number of participants in this analysis. Similarly, no loss of omalizumab efficacy was observed in treatment-experienced patients (i.e. those who had been exposed to > 4 months of omalizumab therapy before the start of this study). These patients tended to obtain control early and retain similar levels of control throughout the course of the study. Furthermore, we observed no loss of efficacy of omalizumab in patients who smoke.
The mini-AQLQ is a 15-item, self-administered questionnaire [12] and was used in this study to measure patient-reported quality of life. Despite the vast majority of patients already receiving omalizumab before the start of the study, significantly improved quality of life scores were reported by patients over the first 2 years of treatment compared with baseline scores. Omalizumab use improved symptoms and emotional function over the course of this study. However, as this study lacked a control group, we cannot exclude the possibility that this is not a treatment effect. Post-hoc analyses on the mini-AQLQ data also indicated improvement in quality of life for treatment-naïve patients and sustained quality of life improvement for treatment-experienced patients.
Effectiveness variables captured in this observational study also included GETE. Physician’s GETE is a recommended instrument to measure response to treatment and overall asthma control [11]. In this study, the majority of patients were considered “responders” to omalizumab treatment, having either excellent or good physician-evaluated GETE at each half-yearly time point. This is consistent with other real-life effectiveness studies of omalizumab [11, 13].
AEs were reported for a relatively high number of patients. We think that the forced reporting model of AEs in this study (i.e. AE reporting was triggered by certain electronic case report form entries by the physician) may hamper representative conclusions pertaining to the safety profile of omalizumab. Importantly, causality with omalizumab was not suspected for most adverse events. No unexpected safety concerns occurred during this study. Given the observational nature of this study, the discontinuation rate was remarkably low.
This study carries certain limitations. As it captured retrospective data during the year before study commencement, as well as prospective data for the 3 years following study commencement, appropriate comparisons between the two data sets cannot be easily made. The low proportion of patients without previous exposure to omalizumab may have further hampered appropriate comparisons. It is difficult to determine efficacy and safety as there is confounding from the substantial number of patients who were receiving omalizumab before the start of this study. Furthermore, this observational study lacked a control group, which may limit drawing meaningful causal relationships from these data.
Nonetheless, our findings support the persistent effectiveness and safety of omalizumab in maintaining asthma control and quality of life in patients with severe asthma, with no observed tachyphylaxis. All sub-populations analysed in these studies showed similar effects.
References
GBD Chronic Respiratory Disease Collaborators. Global, regional, and national deaths, prevalence, disability-adjusted life years, and years lived with disability for chronic obstructive pulmonary disease and asthma, 1990–2015: a systematic analysis for the Global Burden of Disease Study 2015. Lancet Respir Med. 2017;5(9):691–706.
Global Inititative for Asthma. Global strategy for asthma management and prevention, 2018. http://ginasthma.org/gina-reports/. Accessed on Feb 15, 2019.
Chung KF, Wenzel SE, Brozek JL, Bush A, Castro M, Sterk PJ, et al. International ERS/ATS guidelines on definition, evaluation and treatment of severe asthma. Eur Respir J. 2014;43(2):343–73.
Novartis Pharma GmbH, Xolair Summary of Manufactured Product Characteristics (SMPC). http://www.ema.europa.eu/docs/en_GB/document_library/EPAR_-_Product_Information/human/000606/WC500057298.pdf. Accessed on June 15, 2018.
Ledford D, Busse W, Trzaskoma B, Omachi TA, Rosen K, Chipps BE, et al. A randomized multicenter study evaluating Xolair persistence of response after long-term therapy. J Allergy Clin Immunol. 2017;140(1):162–9.
Sousa AS, Pereira AM, Fonseca JA, Azevedo LF, Abreu C, Arrobas A, et al. Asthma control and exacerbations in patients with severe asthma treated with omalizumab in Portugal. Rev Port Pneumol. 2015;21(6):327–33.
Zazzali JL, Raimundo KP, Trzaskoma B, Rosen KE, Schatz M. Changes in asthma control, work productivity, and impairment with omalizumab: 5-year EXCELS study results. Allergy Asthma Proc. 2015;36(4):283–92.
Juniper EF, Svensson K, Mork AC, Stahl E. Measurement properties and interpretation of three shortened versions of the asthma control questionnaire. Respir Med. 2005;99(5):553–8.
Bousquet J, Brusselle G, Buhl R, Busse WW, Cruz AA, Djukanovic R, et al. Care pathways for the selection of a biologic in severe asthma. Eur Respir J. 2017;50(6):1701782.
Casale TB, Luskin AT, Busse W, Zeiger RS, Trzaskoma B, Yang M, et al. Omalizumab effectiveness by biomarker status in patients with asthma: evidence from PROSPERO, a prospective real-world study. J Allergy Clin Immunol Pract. 2019;7(1):156.e1–164.e1.
Braunstahl GJ, Chen CW, Maykut R, Georgiou P, Peachey G, Bruce J. The eXpeRience registry: the ‘real-world’ effectiveness of omalizumab in allergic asthma. Respir Med. 2013;107(8):1141–51.
Juniper EF, Guyatt GH, Cox FM, Ferrie PJ, King DR. Development and validation of the mini asthma quality of life questionnaire. Eur Respir J. 1999;14(1):32–8.
Brusselle G, Michils A, Louis R, Dupont L, Van de Maele B, Delobbe A, et al. “Real-life” effectiveness of omalizumab in patients with severe persistent allergic asthma: the PERSIST study. Respir Med. 2009;103(11):1633–42.
Acknowledgements
We gratefully acknowledge the support of the patients and participating sites of this study.
Funding
This study was funded by Novartis Pharma GmbH, Germany. The Rapid Service and Open Access Fees for publication in Advances in Therapy were also funded by Novartis Pharma GmbH, Germany.
Medical Writing and/or Editorial Assistance
We also thank Gillian Lavelle, PhD, and Claire Twomey, PhD, of Novartis Product Lifecycle Services, Dublin, Ireland, for providing medical writing support, which was funded by Novartis Pharma GmbH, Germany, in accordance with Good Publication Practice (GPP3) guidelines (http://www.ismpp.org/gpp3).
Authorship
All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article, take responsibility for the integrity of the work as a whole, and have given their approval for this version to be published.
Authorship Contributions
All authors made substantial contributions to the conception and design of the study, or acquisition, analysis, and interpretation of the data. Furthermore, all authors critically revised each draft of this article, approved the final version to be published, and agree to be accountable for all aspects of this work.
Disclosures
Claudia Mailänder is a full-time employee of Novartis Pharma GmbH, Germany. Inessa Schwab Sauerbeck is a full-time employee of Novartis Pharma GmbH, Germany. Jens Schreiber reports grants and personal fees from Novartis.
Compliance with Ethics Guidelines
This study was conducted in accordance with the Directives 2001/83/EC and 2010/84/EU, the guideline on Good Pharmacovigilance Practices, Modules VI and VIII, the code of the Freiwillige Selbstkontrolle für die Arzneimittelindustrie (FSA Code; Voluntary Self-Regulation of the Pharmaceutical Industry), the joint recommendations of the Bundesinstitut für Arzneimittel und Medizinprodukte (BfArM; Federal Institute for Drugs and Medical Devices), the Paul Ehrlich-Institute for planning, implementation, and analysis of an observational study, and the Verband forschender Arzneimittelhersteller (VFA; German Association of Research-based Pharmaceutical Companies). This study received approval from the ethics committee of the Otto-von-Guericke University of Magdeburg, Germany (Ethics Approval Number: 07/13). Informed consent was obtained from all individual participants included in the study.
Data Sharing Statement
Novartis is committed to sharing access to patient-level data and supporting clinical documents from eligible studies with qualified external researchers. These requests are reviewed and approved by an independent review panel on the basis of scientific merit. All data provided are anonymised to respect the privacy of patients who have participated in the trial in line with applicable laws and regulations. This trial data availability is according to the criteria and process described on www.clinicalstudydatarequest.com.
Author information
Authors and Affiliations
Corresponding author
Additional information
Enhanced Digital Features
To view enhanced digital features for this article go to https://doi.org/10.6084/m9.figshare.10011848.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/), which permits any noncommercial use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Schreiber, J., Schwab Sauerbeck, I. & Mailänder, C. The Long-Term Effectiveness and Safety of Omalizumab on Patient- and Physician-Reported Asthma Control: A Three-Year, Real-Life Observational Study. Adv Ther 37, 353–363 (2020). https://doi.org/10.1007/s12325-019-01135-w
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12325-019-01135-w