Abstract
Background
It is recognized that, as a result of variation in tissue anatomy, current auto-injectors may have insufficient needle length to achieve successful intramuscular agent delivery in a number of patients. The Zeneo® auto-injector is a novel prefilled, single-use, needle-free device currently in development for intradermal, subcutaneous, and intramuscular agent delivery across a variety of clinical indications. We aimed to evaluate delivery depth of the device calibrated at pressure appropriate for intramuscular (IM) administration.
Methods
This was a prospective single-center study in healthy adult volunteers, in whom each received a single injection of saline into the anterolateral thigh. Using sequential MRI scans, we measured skin-to-muscle distance (STMD) agent delivery depth, and the success of IM agent penetration. Device dynamic pressure measurements were also recorded.
Results
Results are reported for 37 subjects with evaluable MRI scans; 19 men, 18 women; mean age 38 years (range 20–58); mean BMI 27.0 kg/m2 (range 21.2–30.8 kg/m2). Mean STMD values were 18.6 mm (range 13.4–23.6 mm) in women and 10.0 mm (range 5.0–21.7 mm) in men, with gender differences due primarily to greater subcutaneous thickness in women. A trend for greater STMD in subjects with BMI greater than 25 kg/m2 was seen. Mean injectate penetration depths of 30.1 mm (range 20.2–45.6 mm) were observed with values similar in male and female subjects. Successful IM delivery was reported in 95% of subjects. When failure occurred, this was not due to inadequate injection depth. Device pressure (P max) had the greatest influence on injectate muscle penetration.
Conclusion
Use of the Zeneo® auto-injector achieves delivery depth that ensures intramuscular delivery in both men and women, regardless of BMI. Consistent with other reported data, STMD is greater in women.
Funding
Crossject.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
In potentially life-threatening allergic reactions or anaphylaxis (that may arise in response to a number of different triggers), prompt administration of intramuscular adrenaline/epinephrine is the recommended treatment [1,2,3]. Where possible, intramuscular (IM) injection in the lateral thigh is the recommended route of administration, as this results in more rapid absorption and time to peak plasma concentration compared with IM injections via other sites (e.g., deltoid) and with subcutaneous injections [3, 4]. In clinical practice, use of adrenaline auto-injectors is a standard approach for IM administration. A number of different devices are available, which vary regarding a number of characteristics including needle length and device pressures. However, some concerns have been reported regarding the efficacy of some injector devices to successfully deliver adrenaline to the IM tissues because of inadequate needle length [4,5,6,7,8,9]. Furthermore, with conventional auto-injectors, use when required may be reduced as a result of patient fears of injection site pain or a more generalized needle phobia, and accidental injuries may also occur [10, 11].
As such, alternative devices to improve use, including needle-free technologies, are welcome. Zeneo® (Crossject, Chenôve, France) is a novel prefilled, single-use, needle-free auto-injection device currently in development for a variety of clinical indications. This cartridge-based device, specifically designed for self-administration, works via gas propulsion of the drug through micronozzles (250–300 µm diameter) (Fig. 1). Automated drug delivery is triggered by pushing the device onto skin in a simple “place and push” approach, with the device mechanisms housed within a protective casing. By altering the propulsion pressure parameters of the device, which impacts upon the depth of delivery of the chosen agent, the device can be tailored to deliver fixed doses of agents in a range of volumes via the intradermal, subcutaneous (SC), and intramuscular (IM) routes. The primary objective of this study was to evaluate the functional performance of this device in the setting of IM delivery to the lateral thigh. Outcomes of interest included the skin-to-muscle distance (STMD) in the study population, agent delivery depth, and the success of IM agent penetration. Additional outcomes included the impact of anthropometric characteristics on device performance. Safety data (tolerance and pain assessments) and patient experience data were also collected, and these will be the subject of a subsequent broader report.
Methods
Study Design and Study Population
This was a single-center study in healthy volunteers performed between June and December 2015 at the Centre d’Investigation Clinique INSERM 1432, CHU Dijon-Bourgogne, France. The study (Crossject study identifier: G-E-CJT-XC-150127) was performed in accordance with Good Clinical Practice (GCP) guidelines. All procedures followed were also in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Declaration of Helsinki (1964), as revised in 2013 [12]. The study protocol was reviewed and approved by an independent ethics committee (Comité de Protection des Personnes, CPP) and the French National Agency for Medicines and Health Products Safety (ANSM). All subjects provided written informed consent before study inclusion.
The study recruited male or female healthy volunteers aged between 18 and 60 years and body mass index (BMI) 20–30 kg/m2. Prior to inclusion, participants were screened and excluded if clinical examination and/or laboratory investigations were abnormal. At this visit, a range of physical measurements were recorded, including height, weight, and anthropometric measures (waist, hip, and thigh size).
Intervention and Assessments
Subjects returned within 30 days of screening visit for evaluation of device performance. All subjects were under medical supervision during the procedure and for 1 h after the injection. After skin surface antisepsis (with 60% ethanol) all subjects received an injection of 0.9% saline (0.625 ml) into the thigh with the needle-free injector device. Injections were performed by either a trained nurse or by the investigator and applied perpendicular to the skin. During the injection, device dynamic pressure measurements were recorded via a transducer, mounted on the device, from which pressure parameters were obtained, including P init (pressure at 0.7 ms) and P max (maximum pressure after 2 ms).
For each subject, sequential MRI scanning was performed within 7 min of the injection on a 3.0-T Siemens Trio Tim system (Siemens Medical Solutions, Erlangen, Germany) with high resolution T2-weighted 3D DESS (double echo steady-state) sequences used to acquire high contrast images capable of distinguishing the injectate (saline) from muscle and subcutaneous tissue. Images were analyzed for assessment of qualitative and quantitative parameters to characterize different injection/device functions. Qualitative assessments included the success of IM delivery, as assessed by MRI documented muscle penetration. Quantitative assessments included dermal and subcutis thickness at injection site, STMD, and maximum penetration of injectate. Image analysis was performed using ImageJ open source software (NIH, USA).
Data Analysis
Quantitative categorical and continuous variables were described by the number of data, mean ± standard deviation (SD), and range, as appropriate. Differences between groups (men vs women) were calculated using the two-tailed t test for normally distributed data. Principal component analysis (PCA) was performed to identify those factors impacting upon depth of agent delivery.
Results
Baseline Demographic and Clinical Characteristics
A total of 44 subjects received an IM injection. Of these, four subjects were excluded because of incorrect device technique; in three other subjects, technical difficulties in MRI scans rendered these unsuitable for analysis. As such, a total of 37 subjects with evaluable MRI scans were included in this analysis. Subject characteristics are shown in Table 1. The analysis population included very similar numbers of men and women, with a mean age of 38 years (range 20–58 years). Mean baseline body weight was 77 kg (range 56.5–101.9 kg), with a mean BMI of 27.0 kg/m2 (range 21.2–30.8 kg/m2).
Skin Thickness
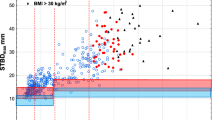
The thickness of different tissue components (dermis and subcutaneous tissues) and the overall STMD are shown in Table 2. The MRI scans showed that the mean dermal thickness for the study population was 1.8 ± 0.3 mm (range 1.1–2.4 mm) with few differences in values between men and women. However, for subcutaneous thicknesses, while for the overall population, the mean thickness was 12.4 ± 5.8 mm (range 3.0–21.9 mm), there were significant differences observed when comparing women and men, with values of 16.9 ± 3.3 mm (range (12.0–21.9 mm) and 8.1 ± 4.4 mm (range 3.0–19.4 mm) respectively (mean difference −8.8; 95% confidence interval −11.4 to −6.2, P < 0.0001). Consequently significant differences in the STMD were also seen, with mean STMD values of 18.6 ± 3.3 mm (range 13.4–23.6 mm) in women, and 10.0 ± 4.7 mm (range 5.0–21.7 mm) reported in men (mean difference −8.6; 95% CI −11.3 to −5.9, P < 0.0001). For both SC thickness and STMD, values were higher in subjects with BMI greater than 25, although these differences were not significant.
Device Pressure and Injection Performance
Across the overall study population, the mean P init was 155 bar (range 140–174 bar) and mean P max was 212 bar (range 187–232 bar). This was within the expected device pressure performance profile. MRI scans were also analyzed to measure delivery and penetration depth of the injectate within muscle (Fig. 2). Out of 37 subjects, 35 had MRI scans that demonstrated IM penetration, while two subjects had no identifiable IM injectate component; this represents a success rate of 94.6%. The MRI scans showed that the mean depth of injectate penetration was 30.1 ± 5.8 mm (range 20.2–45.6 mm) for the overall study population, with minor, non-significant differences observed between male (28.6 ± 6.1 mm, range 20.9–39.2 mm) and female subjects (31.5 ± 5.9 mm, range 20.2–45.6 mm) (Table 2). BMI showed little impact upon injection depth.
To develop an explanatory model to explore the impact of key variables on injection depth, we used principal component analysis (PCA). STMD, P max, BMI, and thigh circumference were all identified as being correlated with injection depth (data not shown) with multivariate linear regression analysis suggesting that P max had the greatest impact on increasing injection depth.
Discussion
In this study, using MRI scanning, we evaluated skin and subcutaneous thickness and STMD at the lateral thigh in healthy volunteers. All measurements were recorded following saline injection via the Zeneo® needle-free auto-injector, and as such were performed on tissue subject to tissue compression as a result of device pressure, which represents the clinical conditions in practice. While a number of studies have evaluated skin measurements in different populations using imaging modalities, most have done so using ultrasound [6, 7, 9, 13,14,15] and less commonly CT [5, 16], relatively few studies have used MRI to evaluate STMDs in the thigh, and primarily in children [16]. MRI was used as the imaging modality, as that also allowed assessment of in vivo agent penetration and IM delivery. We are not aware of studies that have directly compared the sensitivity/accuracy of these different imaging modalities on measuring STMD; a study which investigated such comparisons would of course be of some interest in this respect (although quite possibly challenging to perform). We found that while the mean STMD for the overall population was 14.2 mm, substantial variation exists, with the distance ranging from 5.0–23.6 mm. Furthermore, STMD was greater in women. These data are broadly similar to those reported in previous studies (using ultrasound or CT) [5,6,7, 9, 13, 15] and support the view that STMD is greater in women than in men as a result of a greater SC thickness.
Some studies have discussed thigh STMD in the context of typical needle length and the risk of failure to achieve IM delivery. Using measurements obtained by a retrospective analysis of CT scans, Song et al. reported mean STMDs of 6.6 mm (range 2.1–34.7 mm) in men and significantly greater STMDs of 14.8 mm (range 2.4–37.1 mm; P > 0.0005) in women [5]. In that study, they also evaluated the number of patients with an STMD of greater than 14.3 mm, on the rationale that use of a standard 14.3-mm needle (a frequently used needle length in auto-injector devices) would fail to deliver an IM dose in these patients. They found that while only 2% of men had an STMD greater than 14.3 mm, and were at risk of failure, 42% of women had an STMD exceeding 14.3 mm [5]. Subsequent studies have followed this approach, with greater needle length, and also reported similar concerns. Bhalla et al. evaluated potential risk of IM failure based upon STMD greater than 15.9 mm and reported that 31% were at risk of failure, with women being at far greater risk [7]. More recently, another US study found that 28% of women had an STMD greater than 15.2 mm, while a UK study found that 68% of patients were at risk of IM failure with a needle length of 15.02 mm [6, 15].
These data provide the basis for arguing the need for greater needle lengths in devices for IM delivery. This is supported by our study, where we also found relatively high numbers of subjects with STMDs exceeding the needle length of commonly available auto-injectors. In 17 out of 37 subjects (46%), the STMD was greater than 14.3 mm. While only two men (11%) had STMD greater than 14.3 mm, 15 women (83%) had STMD greater than this value; 43% of subjects had an STMD greater than 15.2 mm, 11% of all men and 78% of all women. It should be recognized that the effect of tissue compression in response to forces required to trigger auto-injectors and also subsequent needle propulsion forces may each contribute to agent delivery depth [17]. Data from a porcine model suggests that such forces make a significant contribution to delivery beyond the 14.3-mm needle length [18]. Nevertheless, it remains that successful IM delivery with many current injectors of this needle length is uncertain. This is reflected in recommendations from Europe where UK guidance is that a 25-mm needle should be used when administering adrenaline/epinephrine in anaphylaxis [19]. Furthermore, albeit in the context of vaccination, the Centers for Disease Control and Prevention (CDC) in the USA also recommend use of a 25-mm needle to ensure IM delivery in the thigh [20].
We also evaluated administration and IM delivery of a test agent (saline) and measured depth of delivery and also delivery success within the thigh muscle. While studies examining this have been performed using animal models [18], we are not aware of similar studies in humans. We found that use of the Zeneo® device, calibrated at pressure appropriate for IM administration, resulted in mean injectate penetration depths of 30.1 mm (range 20.2–45.6 mm) with no meaningful differences between male and female subjects. Additionally, successful IM delivery was reported in 35/37 (95%) of subjects. In those cases of unsuccessful IM delivery, in one subject the injectate traveled the entire STMD and spread on the fascial surface, while in the other case, the injectate traversed the subcutaneous tissue but was directed to a tendon area. This 95% success rate compares favorably with the failure rates extrapolated from the modeling data as reported above [5,6,7, 15]. Although exploratory, our analyses suggested that device pressure (P max) had the greatest influence on injectate penetration. This may be anticipated as the device is designed so as to be capable of delivering to specific tissues for specific indications (i.e., IM or SC) on the basis of pressure settings.
Our study has a number of limitations. Our study population is small, and, because of prespecified inclusion criteria, while over 70% of subjects were overweight (BMI >25 kg/m2), only one had a BMI slightly above 30 kg/m2. As such, there is limited data from the present study regarding device performance, STMD, and IM delivery success in obese patients (BMI >30 kg/m2). This is an important consideration, as it is well recognized that STMD is greater in obese subjects (especially women) [5,6,7]. The mean injectate penetration depths of 30.1 ± 5.8 mm we report would be sufficient for reaching muscle in the great majority of obese patients reported in these studies. Furthermore, an inherent advantage of the Zeneo® system is that device pressure can be modified to deliver agents to different delivery depths—to suit the clinical indication—and so can be altered should greater delivery depths for IM penetration in obese patients with greater STMDs be necessary. Studies evaluating Zeneo® device performance in obese patients are ongoing.
The study was performed in one center in France, and the subjects recruited may not be representative of those from other areas. Injections were administered by healthcare staff with experience in device use, rather than subject self-administration, and while we do not anticipate lower rates, data is required to show whether the injection depths and IM delivery success will be similar following self-administration. In this study we did not formally analyze the volume of saline delivered within the muscle, and so the proportion of injectate deposited in the more superficial dermal and subcutaneous tissues is unclear. This aspect of the device performance is the subject of further investigation. Finally, in the present study, device use and saline injection were performed directly against naked skin. However, in the clinical context of adrenaline injection, successful delivery through clothing (e.g., denim) is an essential consideration. The effect of clothing on Zeneo® device performance has been investigated in allied, as of yet unpublished, studies; these suggest that there is no significant impact of administration through clothing on depth of agent penetration and IM delivery (Crossject, data on file).
Strengths of this study include the use of MRI to document STDM measurements in a realistic clinical setting, i.e., following use of an auto-injector, along with measurement of injectate depths and documented IM penetration. To our knowledge, this is the first such study to do so.
Conclusions
Our study reported STMDs similar to those reported in other studies and with similar gender differences, being significantly greater in women. Use of the Zeneo® needle-free auto-injector achieves penetration depths substantially greater than the needle length of many currently available devices, with successful IM agent delivery in 95% of subjects.
References
Lieberman P, Nicklas RA, Randolph C, et al. Anaphylaxis–a practice parameter update 2015. Ann Allergy Asthma Immunol. 2015;115(5):341–84.
Simons FE, Ebisawa M, Sanchez-Borges M, et al. 2015 update of the evidence base: World Allergy Organization anaphylaxis guidelines. World Allergy Organ J. 2015;8(1):32.
Simons FE, Ardusso LR, Bilò MB, et al. International consensus on (ICON) anaphylaxis. World Allergy Organ J. 2014;7(1):9.
Simons FER, Gu X, Simons KJ. Epinephrine absorption in adults: intramuscular versus subcutaneous injection. J Allergy Clin Immunol. 2001;108(5):871–3.
Song TT, Nelson MR, Chang JH, Engler RJ, Chowdhury BA. Adequacy of the epinephrine autoinjector needle length in delivering epinephrine to the intramuscular tissues. Ann Allergy Asthma Immunol. 2005;94(5):539–42.
Tsai G, Kim L, Nevis IF, et al. Auto-injector needle length may be inadequate to deliver epinephrine intramuscularly in women with confirmed food allergy. Allergy Asthma Clin Immunol. 2014;10(1):39.
Bhalla MC, Gable BD, Frey JA, Reichenbach MR, Wilber ST. Predictors of epinephrine autoinjector needle length inadequacy. Am J Emerg Med. 2013;31(12):1671–6.
Stecher D, Bulloch B, Sales J, Schaefer C, Keahey L. Epinephrine auto-injectors: is needle length adequate for delivery of epinephrine intramuscularly? Pediatrics. 2009;124(1):65–70.
Dreborg S, Wen X, Kim L, et al. Do epinephrine auto-injectors have an unsuitable needle length in children and adolescents at risk for anaphylaxis from food allergy? Allergy Asthma Clin Immunol. 2016;12:11.
Money AG, Barnett J, Kuljis J, Lucas J. Patient perceptions of epinephrine auto-injectors: exploring barriers to use. Scand J Caring Sci. 2013;27(2):335–44.
Simons FE, Lieberman PL, Read Jr EJ, Edwards ES. Hazards of unintentional injection of epinephrine from autoinjectors: a systematic review. Ann Allergy Asthma Immunol. 2009;102(4):282–7.
World Medical Association. World Medical Association Declaration of Helsinki: ethical principles for medical research involving human subjects. JAMA. 2013;310(20):2191–4.
Gibney M, Arce CH, Byron KJ, Hirsch LJ. Skin and subcutaneous adipose layer thickness in adults with diabetes at sites used for insulin injections: implications for needle length recommendations. Curr Med Res Opin. 2010;26(6):1519–30.
Hirsch L, Byron K, Gibney M. Intramuscular risk at insulin injection sites–measurement of the distance from skin to muscle and rationale for shorter-length needles for subcutaneous insulin therapy. Diabet Technol Ther. 2014;16(12):867–73.
Johnstone J, Hobbins S, Parekh D, O’Hickey S. Excess subcutaneous tissue may preclude intramuscular delivery when using adrenaline autoinjectors in patients with anaphylaxis. Allergy. 2015;70(6):703–6.
Lippert WC, Wall EJ. Optimal intramuscular needle-penetration depth. Pediatrics. 2008;122(3):e556–63.
Song TT, Lieberman P. Epinephrine auto-injector needle length: what is the ideal length? Curr Opin Allergy Clin Immunol. 2016;16(4):361–5.
Song TT, Merrill NL, Cole JW. Delivery depth of epinephrine by auto-injector into the subcutaneous tissue of pig. Ann Allergy Asthma Immunol. 2013;111(2):143–5.
Resuscitation Council (UK). Emergency treatment of anaphylactic reactions: guidelines for healthcare providers. London: Resuscitation Council (UK); 2008.
Centers for Disease Control and Prevention. Epidemiology and prevention of vaccine-preventable diseases. In: Hamborsky J, Kroger A, Wolfe S, editors. 13th ed. Washington: Public Health Foundation; 2015.
Acknowledgements
This study, article processing charges and the open access fee for this study were funded by Crossject. Medical writing support was provided by Iain O’Neill on behalf of Newmed Medical Publishing, funded by Crossject. Marc Bardou, Maxime Luu, and Xavière Castano were responsible for the conception and design of the study, study monitoring, and data collection and interpretation. Paul Walker performed the MRI imaging and analyzed the data. Christophe Auriel performed the statistical analyses. All authors had full access to all of the data in this study and take complete responsibility for the integrity of the data and data analysis. All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this manuscript, take responsibility for the integrity of the work as a whole, and have given final approval for the version to be published. The authors wish to thank the subjects and healthcare staff who participated in this study.
Disclosures
Christophe Auriel and Xavière Castano are employees of Crossject, the study sponsor. Marc Bardou is an expert for the French Medicines Agency. Maxime Luu and Paul Walker report no conflicts of interest.
Compliance with Ethics Guidelines
The study (Crossject study identifier: G-E-CJT-XC-150127) was performed in accordance with Good Clinical Practice (GCP) guidelines. All procedures followed were also in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Declaration of Helsinki (1964), as revised in 2013 [12]. The study protocol was reviewed and approved by an independent ethics committee (Comité de Protection des Personnes, CPP) and the French National Agency for Medicines and Health Products Safety (ANSM). All subjects provided written informed consent before study inclusion.
Data Availability
The datasets generated during and/or analyzed during the current study are not publicly available for reasons of commercial confidentiality but are available from the corresponding author on reasonable request.
Open Access
This article is distributed under the terms of the Creative Commons Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/), which permits any noncommercial use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
Author information
Authors and Affiliations
Corresponding author
Additional information
Enhanced content
To view enhanced content for this article go to http://www.medengine.com/Redeem/6427F0606404A394.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0), which permits use, duplication, adaptation, distribution, and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Bardou, M., Luu, M., Walker, P. et al. Efficacy of a Novel Prefilled, Single-Use, Needle-Free Device (Zeneo®) in Achieving Intramuscular Agent Delivery: An Observational Study. Adv Ther 34, 252–260 (2017). https://doi.org/10.1007/s12325-016-0452-0
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12325-016-0452-0