Abstract
Introduction
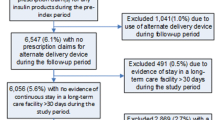
Real-world data comparing outcomes of type 2 diabetes mellitus (T2DM) patients initiating different insulin regimens can help with treatment decisions and patient management. Clinical and economic outcomes following initiation with insulin glargine disposable pen (GLA-P) or insulin detemir disposable pen (DET-P) in T2DM patients were compared over 1-year follow-up.
Methods
This retrospective cohort analysis was conducted on data in a US national managed care claims database (July 2006 to September 2010) from patients initiating insulin treatment with GLA-P or DET-P. Treatment persistence, adherence, glycated hemoglobin (A1C), hypoglycemic events, and healthcare costs during follow-up were compared.
Results
In all, 1682 patients were identified; 1016 (60.4%) started using GLA-P, 666 (39.6%) started using DET-P. After 1:1 propensity score matching, each cohort comprised 640 patients. Patients initiating GLA-P were significantly more likely to persist and adhere to treatment, and used a lower daily consumption dose. Over the last quarter of follow-up, fewer GLA-P users switched to DET-P compared with those switching from DET-P to GLA-P. GLA-P was associated with lower A1C levels and higher reduction of A1C levels from baseline, with no significant difference in the number of patients having hypoglycemic events. Patients in both cohorts had similar total and diabetes-related healthcare costs, but healthcare costs were lower in the GLA-P cohort for each 1% reduction in A1C from baseline.
Conclusion
This real-world study demonstrates that patients initiating GLA-P were more likely to persist with and adhere to treatment, with better glycemic control and similar overall hypoglycemia rate at no increase in healthcare cost.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Shaw JE, Sicree RA, Zimmet PZ. Global estimates of the prevalence of diabetes for 2010 and 2030. Diabetes Res Clin Pract. 2010;87:4–14.
Sturm R. Stemming the global obesity epidemic: what can we learn from data about social and economic trends? Public Health. 2008;122:739–746.
Hill JO, Wyatt HR, Reed GW, Peters JC. Obesity and the environment: where do we go from here? Science. 2003;299:853–855.
Centers for Disease Control and Prevention. National diabetes fact sheet: national estimates and general information on diabetes and prediabetes in the United States, 2011. Atlanta, GA: US Department of Health and Human Services, Centers for Disease Control and Prevention, 2011. Available at: www.cdc.gov/diabetes/pubs/pdf/ndfs_2011.pdf. Last accessed October 13, 2011.
American Diabetes Association. Economic costs of diabetes in the U.S. in 2007. Diabetes Care. 2008;31:596–615.
Mattila TK, de Boer A. Influence of intensive versus conventional glucose control on microvascular and macrovascular complications in type 1 and 2 diabetes mellitus. Drugs. 2010;70:2229–2245.
Kim S. Burden of hospitalizations primarily due to incontrolled diabetes: implications of inadequate primary health care in the United States. Diabetes Care. 2007;30:1281–1282.
Morsanutto A, Berto P, Lopatriello S, et al. Major complications have an impact on total annual medical cost of diabetes: results of a database analysis. J Diabetes Complications. 2006;20:163–169.
The Diabetes Control and Complications Trial Research Group. The effect of intensive treatment of diabetes on the development and progression of long-term complications in insulin-dependent diabetes mellitus. N Engl J Med. 1993;329:977–986.
UK Prospective Diabetes Study (UKPDS) Group Intensive blood-glucose control with sulphonylureas or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes. Lancet. 1998;352:837–853.
American Diabetes Association. Standards of medical care in diabetes—2011. Diabetes Care. 2011;34(Suppl. 1):S11–S61.
Rodbard HW, Jellinger PS, Davidson JA, et al. 2009 AACE/ACE Consensus Statement: Statement by an American Association of Clinical Endocrinologists/American College of Endocrinology Consensus Panel on Type 2 Diabetes Mellitus: An Algorithm for Glycemic Control. Available at: http://alt.aace.com/pub/pdf/GlycemicControlAlgorithm.pdf. Last accessed October 13, 2011.
Department of Health and Human Services, Centers for Disease Control and Prevention. 2010 Diabetes Data & Trends. Treating Diabetes (insulin and oral medication use). Available at: www.cdc.gov/diabetes/statistics/treating_national.htm. Last accessed October 13, 2011.
Rosenstock J, Davies M, Home PD, Larsen J, Koenen C, Schernthaner G. A randomised, 52-week, treat-to-target trial comparing insulin detemir with insulin glargine when administered as add-on to glucose-lowering drugs in insulinnaive people with type 2 diabetes. Diabetologia. 2008;51:408–416.
Heller S, Koenen C, Bode B. Comparison of insulin detemir and insulin glargine in a basal-bolus regimen, with insulin aspart as the mealtime insulin, in patients with type 1 diabetes: a 52-week, multinational, randomized, open-label, parallelgroup, treat-to-target noninferiority trial. Clin Ther. 2009;31:2086–2097.
Raskin P, Gylvin T, Weng W, Chaykin L. Comparison of insulin detemir and insulin glargine using a basal-bolus regimen in a randomized, controlled clinical study in patients with type 2 diabetes. Diabetes Metab Res Rev. 2009;25:542–548.
Marks JB. How do detemir and glargine compare when added to oral agents in insulin-naïve patients with type 2 diabetes mellitus? Nat Clin Pract Endocrinol Metab. 2008;4:490–491.
Gordon J, Pockett RD, Tetlow AP, McEwan P, Home PD. A comparison of intermediate and long-acting insulins in people with type 2 diabetes starting insulin: an observational database study. Int J Clin Pract. 2010;64:1609–1618.
Swinnen SG, Dain MP, Aronson R, et al. A 24-week, randomized, treat-to-target trial comparing initiation of insulin glargine once-daily with insulin detemir twice-daily in patients with type 2 diabetes inadequately controlled on oral glucose-lowering drugs. Diabetes Care. 2010;33:1176–1178.
Reynolds LR. Comparing insulins detemir and glargine in type 2 diabetes: more similarities than differences. Commentary. Postgrad Med. 2010;122:201–203.
Coscelli C, Lostia S, Lunetta M, Nosari I, Coronel GA. Safety, efficacy, acceptability of a pre-filled insulin pen in diabetic patients over 60 years old. Diabetes Res Clin Pract. 1995;28:173–177.
Kadiri A, Chraibi A, Marouan F, et al. Comparison of NovoPen 3 and syringes/vials in the acceptance of insulin therapy in NIDDM patients with secondary failure to oral hypoglycaemic agents. Diabetes Res Clin Pract. 1998;41:15–23.
Korytkowski M, Bell D, Jacobsen C, Suwannasari R; FlexPen Study Team. A multicenter, randomized, open-label, comparative, two-period crossover trial of preference, efficacy, and safety profiles of a prefilled, disposable pen and conventional vial/syringe for insulin injection in patients with type 1 or 2 diabetes mellitus. Clin Ther. 2003;25:2836–2848.
Keith K, Nicholson D, Rogers D. Accuracy and precision of low-dose insulin administration using syringes, pen injectors, and a pump. Clin Pediatr (Phila). 2004;43:69–74.
Baser O, Bouchard J, DeLuzio T, Henk H, Aagren M. Assessment of adherence and healthcare costs of insulin device (FlexPen) versus conventional vial/syringe. Adv Ther. 2010;27:94–104.
Rosenbaum PR, Rubin DB. The central role of the propensity score in observational studies for causal effects. Biometrika. 1983;70:41–55.
Garg SK. The role of basal insulin and glucagonlike peptide-1 agonists in the therapeutic management of type 2 diabetes—a comprehensive review. Diabetes Technol Ther. 2010;12:11–24.
Pscherer S, Dietrich ES, Dippel FW, Neilson AR. Comparison of one-year costs of type 2 diabetes treatment with insulin glargine or insulin detemir in a basal supported oral therapy (BOT) in Germany. Int J Clin Pharmacol Ther. 2010;48:129–137.
Guillermin AL, Samyshkin Y, Wright D, Nguyen T, Villeneuve J. Modeling the lifetime costs of insulin glargine and insulin detemir in type 1 and type 2 diabetes patients in Canada: a meta-analysis and a cost-minimization analysis. J Med Econ. 2011;14:207–216.
Swinnen SG, Simon AC, Holleman F, Hoekstra JB, Devries JH. Insulin detemir versus insulin glargine for type 2 diabetes mellitus. Cochrane Database Syst Rev. 2011;7:CD006383.
Pearl J. Understanding propensity scores. In: Causality: Models, Reasoning, and Inference. 2nd Edition. New York, NY: Cambridge University Press; 2009:348–351.
Baser O. Too much ado about instrumental variable approach: Is the cure worse than the disease? Value Health. 2009;12:1201–1209.
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is published with open access at Springerlink.com
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Xie, L., Wei, W., Pan, C. et al. A real-world study of patients with type 2 diabetes initiating basal insulins via disposable pens. Adv Therapy 28, 1000–1011 (2011). https://doi.org/10.1007/s12325-011-0074-5
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12325-011-0074-5