Abstract
The Achilles tendon (AT) is the most frequently ruptured tendon in the human body yet the aetiology remains poorly understood. Despite the extensively published literature, controversy still surrounds the optimum treatment of complete rupture. Both non-operative management and percutaneous repair are attractive alternatives to open surgery, which carries the highest complication and cost profile. However, the lack of a universally accepted scoring system has limited any evaluation of treatment options. A typical UK district general hospital treats approximately 3 cases of AT rupture a month. It is therefore important for orthopaedic surgeons to correctly diagnose and treat these injuries with respect to the best current evidence-based practice. In this review article, we discuss the relevant pathophysiology and diagnosis of the ruptured AT and summarize the current evidence for treatment.
Similar content being viewed by others
Introduction
The Achilles tendon (AT) is the largest and strongest tendon in the human body [1]. Spontaneous rupture has become more common recently due to an increase in the elderly population and recreational sporting activity by the middle aged [2]. Although most AT ruptures (44–83 %) occur during sport, intrinsic structural, biochemical and biomechanical changes may be important [3]. Until the beginning of the twentieth century, treatment was mainly non-operative with various methods of immobilization. Following reports in the 1920s by Abrahamsen [4], operative repair has gained popularity. However, when Lea and Smith [5] published encouraging non-operative outcomes, both options seemed noteworthy.
Controversy surrounding best treatment exists because outcomes are determined by the repair method and also post-operative functional rehabilitation. Specifically, the risks and benefits of open versus closed treatment continue to be debated and the safest, cost-effective method remains undecided. This paper reviews the pathophysiology and diagnosis of acute AT rupture and summarizes the current evidence base for treatment.
Epidemiology
AT ruptures have become more common in the last two decades with an annual incidence of 18 per 100,000 [6]. The male to female ratio is approximately 1.7:1–30:1 [7] perhaps reflecting a higher prevalence of males in sports. Most studies demonstrate a bimodal age distribution with peaks in the fourth decade and a second lesser peak between the sixth and eight decades, probably representing two different aetiologies [8]. In younger patients, sports are most often implicated, most commonly football (in Europe) [9] and racquet sports (in Scandinavia) [6].
The Achilles tendon
Anatomy
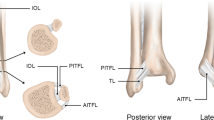
The tendinous contributions of the gastrocnemius and soleus muscles merge to form the AT. The gastrocnemii contribution begins as a wide aponeurosis at the lower end of these muscular bellies. The soleus tendon contribution is thicker and shorter. Both tendons converge approximately 15 cm proximal to the insertion site, which is a specialized region composed of a tendon attachment, a layer of hyaline cartilage and a bone area uncovered by periosteum. A subcutaneous bursa lies between the skin and the AT and a retrocalcaneal bursa between the AT and the calcaneus. In cross-section, the AT spirals internally with the right one rotating counter-clockwise 120° towards its insertion (left clockwise) [10].
The vascular supply to the AT originates from three sources: the surrounding connective tissue, the musculotendinous junction and the bone–tendon junction. The latter derives from the long and short vincula, via the paratenon and mainly from the ventral mesotendon [11]. There is no consensus on the distribution of the blood vessels. Whilst cadaveric studies show low blood vessel density in the mid portion [12], supporting the outdated theory of a hypovascular ‘watershed’ zone, recent laser Doppler flowmetry studies show an even distribution which may vary with age, gender and loading conditions [13]. The sensory nerve supply to the AT is derived from the attaching muscles and overlying cutaneous nerves, particularly, the sural nerve [14].
Histologically, 90–95 % of the cellular elements in the AT comprise of tenocytes and tenoblasts. Collagen accounts for approximately 70 % of the dry weight of a tendon, of which almost 95 % is type-1 collagen with a small amount of elastin [15]. The epitenon and paratenon layers are separated by a thin film of fluid which aid tendon excursion with reduced friction. In the ruptured AT, tenocytes produce a larger proportion of type III collagen in comparison with tenocytes from a healthy tendon [16]. Type III collagen is less resistant to tensile forces and therefore renders it more susceptible to rupture [17].
Biomechanics
With the presence of actin and myosin in tenocytes, the AT has ideal mechanical properties for transmission of force from muscle to bone. It is stiff but resilient, with a high tensile strength and the capacity to stretch up to 4 % of the original length thereby accommodating most physiological loads. Fukashiro et al. [18] measured a peak force of 2,233 N within the human AT in vivo before heel strike. Arndt et al. [19] showed the AT may be subjected to non-uniform stresses as a result of asynchronous contraction of the different components of triceps surae or uncoordinated agonist–antagonist muscle contraction.
At rest, the AT has a wavy configuration from crimping of the collagen fibrils [20]. When stretched more than 2 %, this is lost and it responds linearly to increasing loads [21]. If, however, the strain applied exceeds 4 % of its normal length, the tendon begins to disrupt with tensile failure at levels greater than 8 % [21].
The Aetiology of rupture
Multiple attributing factors and associations with other medical disorders have been described (Table 1) in AT rupture. Two main explanations exist: the degenerative and mechanical theories. Risk factors for rupture include corticosteroids, fluoroquinolone antibiotics and hyperthermia.
Theories of rupture
Degenerative theory
Early experiments supported the degeneration theory and Langergren and Lindholm [22] showed that ruptures were usually limited to a segment about 2–6 cm proximal to the calcaneal insertion. They concluded that hypovascularity coupled with repeated trauma, together preventing regeneration, can lead to rupture. Another study reported degenerative changes in all 74 patients, also implicating poor blood supply [23]. The intra-tendinous vessels and the relative area occupied by these blood vessels are known to be lowest at this zone. Whether this is due to the torsional trajectory of the tendon compressing on the transverse vincula, or simply a lack of vascularized tissue is unknown [23]. Kannus and Jozsa [24] showed histological evidence of degeneration in a third of ruptured ATs and noted most patients were asymptomatic prior to rupture. Alternating sports with periods of inactivity could result in cumulative microtrauma, which, although below the threshold of complete rupture, may result in secondary intra-tendinous degenerative changes [25].
Ageing affects all collagenous structures in the body including the AT [26]. Tendons from skeletally mature individuals are stronger and more resilient than those from older ones [27]. The changes in microstructure associated with ageing include the following: (1) increase in non-reducible collagen cross-linking, (2) increase in elastin content, (3) reduction in collagen fibril ‘crimp’ angle, (4) smaller collagen fibril diameter, (5) decreased extracellular water and mucopolysacharides and (6) increase in type V collagen [27]. These changes could lower the threshold for microscopic collagen fibril tears and increase the likelihood of damage. Chronic tendinosis may sometimes manifest itself as calcification within the Achilles tendon. This may be either insertional or non-insertional in distribution and is likely a reflection of microtears and degeneration within the intra-tendinous substance. As such, the patient should be warned that he or she may be at increased risk for a complete rupture. Likewise, removal of a calcific mass may resolve the symptoms of tendinosis. Collins and Raleigh [28] recently showed an association between sequence variants in the TNC gene and molecular mechanisms resulting in rupture, suggesting a genetic predisposition.
Mechanical theory
The mechanical theory relates to the peak incidence occurring in the middle aged rather than in the elderly [29]. Inglis and Sculco [30] suggested that a malfunction in the inhibitory mechanism protecting against excessive or uncoordinated muscle contractions could cause rupture at the site of maximum stress and torsion. Athletes returning after inactivity may be most susceptible to this mechanism. Barfred [31] showed that complete rupture can occur in healthy tendons, if obliquely loaded at a short initial length with maximal muscle contraction typical of rapid push-offs necessary in many sports. Therefore, a violent muscular force could cause rupture from incomplete synergism of agonist muscle contractions, inefficient plantaris action or a difference in the muscle–tendon thickness quotient. In a study of 109 runners, Clement et al. [32] demonstrated that AT injury may be due to structural or dynamic disturbances such as overtraining, functional over-pronation and gastrocnemius-soleus insufficiency. Again they concluded that repeated microtrauma from eccentric loading of fatigued muscle led to multiple microruptures and eventual failure beyond a critical point.
Risk factors
Corticosteroids
Both systemic and local corticosteroids have been implicated in AT rupture. Indeed, their anti-inflammatory properties may initially mask underlying tendinopathy symptoms [33]. Corticosteroids delay healing and an intra-tendinous injection for tendonitis may weaken it for up to 2 weeks [34]. Histologically, there is collagen necrosis with reformation of an amorphous collagen mass. Though historically popular, a recent meta-analysis demonstrated corticosteroid injections have no benefit in Achilles tendinopathy [35].
Fluoroquinolones
Fluoroquinolone antibiotics, such as ciprofloxacin, rarely cause Achilles tendinopathy. Such treatment resulted in a spectrum of tendon disorders including 31 ruptures in a study of 100 patients [36] and a 5.8 % tendinopathy rate in a study by Barge-Caballero et al. [37] in 149 heart transplant patients. The pathophysiology remains unclear. Whilst the chelating properties of fluoroquinolones may disturb tendon integrity, mitochondria could be a biological target [38].
Vascular influence
Petersen et al. [39] investigated the expression of the important angiogenic factors, vascular endothelial growth factor receptors 1 and 2 (VEGFR-1 and VEGFR-2), in the AT. Neovascularization was shown to influence the aetiology of degeneration and rupture. Moreover, inhibiting angiogenesis may be a new therapeutic approach to degenerative AT disease [39].
Hyperthermia
Temperature rises of up to 45 °C have been detected in vivo in the central core of equine superficial digital flexor tendons (SDFT) during high-speed locomotion [40]. Mathematical models show a similar rise in human ATs during vigorous exercise [41]. Hyperthermic tendon damage compromises the integrity of the extracellular matrix. Moreover, the central core is also the site of degeneration and subsequent injury in both the equine SDFT [40] and the human AT [23].
Presentation and diagnosis
The diagnosis of AT rupture is based on a good history and examination. The typical patient is a middle-aged man playing a sport entailing sudden starting and stopping, such as tennis, basketball or badminton. Patients characteristically report sudden onset of pain in the affected leg or the sensation of being struck at the back of their lower leg. They are often unable to bear weight with ankle weakness, although plantar flexion may be preserved. The patient with a chronically ruptured tendon may only describe minor trauma and difficulty with daily functional activities such as stairs [42].
Arner and Lindholm [43] described 3 main mechanisms of indirect injury in 92 patients with AT ruptures. Fifty-three per cent occurred when pushing off with the weight-bearing forefoot whilst extending the knee; 17 % from sudden unexpected dorsiflexion of the ankle (slipping into a hole) and 10 % from violent dorsiflexion of a plantar flexed foot (fall from height).
Clinical examination may reveal oedema and bruising with a palpable gap along the course of the tendon. In their series of 303 patients, Krueger-Franke et al. [44] measured the mean location of rupture intra-operatively to be 4.78 cm proximal to the calcaneal insertion. Despite this, over 20 % of ruptures are missed by the first examining doctor [45]. To avoid misdiagnosis, there are several diagnostic signs and tests, both clinical and radiographic that are useful.
Clinical tests
A summary of the commonly performed clinical tests is tabulated in Table 2. Maffulli et al. [46] analysed various clinical tests used in 174 patients with complete AT rupture and 28 with suspected tears. Both Simmonds (Fig. 1) and Matles tests were significantly more sensitive (0.96 and 0.88) than the Copeland and O’Brien tests (0.80). All tests showed a high positive predictive value and ability to exclude tear with specificities of 0.93 (Simmonds), 0.85 (Matles) and 0.89 (gap palpation). In our institution, the senior author emphasizes the diagnostic value of inspection of both ankles hanging off the examination couch in the prone position. This reliably demonstrates a comparable loss of plantar flexion attitude in the affected limb, due to absence of the resting tension of the intact AT (Fig. 2).
Radiological tests
Plain radiography
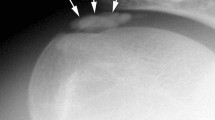
Most authors regard radiographs of secondary importance to physical examination. Kager et al. [47] described a triangle seen on the lateral radiograph that is fat filled and bound by the margins of the anterior border of the AT, the posterior tibia and the superior calcaneus. Following rupture, the anterior border of the AT approaches the posterior aspect of the tibia and the triangle loses its regular configuration. In addition, the Toygar sign [48] involves measuring the angle of the posterior skin surface on the lateral radiograph, given the ruptured tendon ends displace anteriorly.
Ultrasonography
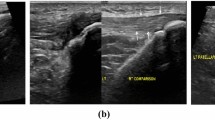
Although operator dependent, ultrasonography remains favoured by musculoskeletal radiologists in diagnosis [49]. It is relatively inexpensive, fast, repeatable and enables dynamic examination. Linear ultrasonography produces both a dynamic and panoramic image with the normal AT appearing as a hypoechogenic, ribbon-like image contained within 2 hyperechogenic bands [50]. Rupture appears as an acoustic vacuum with thick irregular edges [50]. Ultrasonography is important to diagnose partial (often subclinical) ruptures [51] and exclude injury thus preventing unnecessary treatment.
Magnetic resonance imaging (MRI)
Despite the MRI being relatively expensive, it delineates the AT against the fat pad of Kager’s triangle well. It is the imaging modality of choice as it is superior in detecting incomplete ruptures and various chronic degenerative changes. In T1-weighted images, complete rupture is identified as disruptions of the signal within the tendon, whilst in T2-weighted images, there is a generalized signal intensity increase with oedema and haemorrhage (Fig. 3).
MRI is most useful in defining AT quality and whether it needs, or indeed can be repaired. For example, with gross degeneration tendon transfer is a better option. Furthermore, MRI identifies the presence of plantaris which may be used as an adjunct to repair or reconstruction. It also provides information in regard to the height of the rupture, and whether there has been any bony avulsion requiring bone fixation (Fig. 3).
Management of acute rupture
Management is dependent on surgeon preference and patient choice. The goals of treatment are to restore length and tension, therefore optimizing ultimate strength and function. Options can be broadly categorized into operative (open or percutaneous) and non-operative (cast immobilization or functional bracing).
Non-operative management
Cast immobilization has been shown to have equally good outcome to open repair but with lower complications, morbidity and cost. Although rupture outside the synovial sheath has a tendency to spontaneously repair, the tendon ends need to be held closely opposed to avoid lengthening and loss of power. Lea and Smith [5] successfully described non-operative management claiming that the ruptured AT ‘will regenerate itself’. They reported satisfactory results in 95 % of patients treated in a short leg cast for 8 weeks with a 11 % re-rupture rate. Numerous further studies reported re-rupture rates of 18–39 % [30, 52]. Nistor et al. [53] undertook the first level II prospective randomized series in 105 patients comparing cast immobilization with surgery and showed no difference in the range of motion or plantar flexion strength. Re-rupture rates were 8 and 4 %, respectively, with 29 complications in the operative group only. Carden et al. [7] also showed a higher re-rupture rate if non-operative management was not initiated within 48 h of injury.
Functional bracing
Functional bracing (Fig. 4) may result in increased range of motion, an earlier return to the pre-injury activity level and greater comfort [54]. A level 1 study of 40 patients using the ‘Sheffield brace’ reported that ankle dorsiflexion improved more rapidly than without a brace with a re-rupture rate of 10 % [54]. Wallace et al. [55] recommended an equinus cast for 4 weeks followed by a functional removable brace with a 2 % re-rupture and 3.5 % partial re-rupture rate (mainly non-compliant patients). Similar encouraging results have been reported [56, 57], although in contrast they initiated the regime within 24 h of injury.
Operative management
Open repair
The advantage of open repair is the possibility of exerting early tension on the repaired tendon. There are many repair methods with variations in suture technique (Figs. 5, 6, 7), suture type [Vicryl, Dexon, polydioxanone (PDS)], external fixation and augmented repairs.
The argument against operative treatment remains the complications. In a review of 4,477 patients by Lemaire et al. [58], the complication rate was 12.5 %, the commonest being minor wound problems (6.5 %) with a re-rupture rate of 1.5 %. Cetti et al. [59] reported that patients in the operative group were more likely to resume sporting activities (57 vs. 29 %) and at 12-month follow-up, less likely to have difficulty walking and wearing shoes (29 vs. 49 %). The same authors also reviewed studies measuring plantar flexion strength (relative to normal) with values of 87.3 and 78.2 % treated operatively and non-operatively, respectively [59].
Several authors have reported augmented open repair of acutely ruptured ATs, primarily with gastrocnemius-soleus fascia [60], polypropylene braid [61], polyethylene mesh and the plantaris tendon (Fig. 8) [62]. There is, however, insufficient evidence to advocate augmented repairs over simple end-to-end suturing techniques.
Functional rehabilitation
Lately, early functional rehabilitation and the theoretical accelerated return of tendon strength after open repair have gained much attention. In a Level 1 study of 71 patients, Mortensen et al. [52] examined conventional casting for 8 weeks versus early restricted motion in a below knee brace for 6 weeks post-operatively. The early motion group had a smaller initial loss of range of motion and returned to both work and sports sooner. More recently, Maffulli et al. [63] compared immediate and delayed (in a cast) weight-bearing post-operatively. The former showed early independent ambulation (mean 2.5 weeks) with greater satisfaction levels and no difference in tendon thickness or isometric strength. A randomized controlled trial by Costa et al. [64] showed improved functional outcome for fully weight-bearing patients post-operatively without a higher complication rate.
Percutaneous repair
Percutaneous repair was introduced to reduce wound complications and was first developed by Ma and Griffith [65]. It involves passing a Bunnell stitch through a series of stab incisions along the medial and lateral aspects of the AT without exposing the rupture site. They reported no re-ruptures or infections. On the contrary, subsequent studies have demonstrated re-rupture and nerve injury rates of up to 8 % [66] and 13 % [67, 68], respectively.
Biomechanically, percutaneous repair may deter optimal suture technique thereby compromising on repair strength. Krackow et al. [69] described a non-strangulating locking loop technique (Fig. 7) with superior pull-out strength. Watson et al. [70] showed this was stronger than the Bunnell and Kessler methods in cadavers. In a biomechanical analysis using the Bunnell stitch, Hockenbury and Johns [71] showed that percutaneous repair provided 50 % of the initial strength of open repair. However, more recently, Carmont and Maffulli [72] described a modification of the Webb and Bannister [73] technique (Fig. 9) with promising results.
Cretnik et al. [74] compared 132 percutaneous with 105 open repair patients using a Kessler method with fascial reinforcement. The percutaneous group had no wound complications compared to 5.7 % of the open group. Re-rupture rates were similar at 3.7 and 2.8 %, respectively. In a Level 1 study of 66 patients, the open group had 21 % wound complication and 6 % re-rupture rates, compared to 0 % infection, 3 % re-rupture and 3 % nerve injury rates in the percutaneous group [66]. More recently, in a study examining isokinetic strength and endurance, Goren et al. [75] showed no difference between the two techniques. Whilst percutaneous repair may have lower wound complications, concern remains regarding nerve injury and re-rupture rates. This prompted Calder and Saxby [76] to report a previously described a mini-open technique (Achillon suture system) in 46 patients. They used a supervised active rehabilitation programme from 2 weeks post-operatively. There were no re-ruptures at 1 year and most resumed sporting activities at 6 months. Other authors have been able to reproduce similar results with the Achillon technique whilst reporting none of the surgical complications above [77].
Discussion
Despite the vast amount of available evidence, there is no clear consensus on how best to manage these common injuries. In 2010, the American Academy of Orthopaedic Surgeons’ AT workgroup issued guidelines confirming there was no strong evidence to substantiate any of the recommendations in the diagnosis and treatment of acute AT rupture. There was moderate strength recommendations favouring early post-operative protected weight-bearing and protective devices that allow for early ambulation [78].
In 2002, Wong et al. [79] selected 125 studies on 5,370 patients and reported the rate of wound complications of 14.6 % in operative and 0.5 % in non-operative patients. Re-rupture rates in both groups were 1.4 % and 10.7 %, respectively. They concluded that patients treated with open repair had the best functional recovery with a decreasing trend of reported complications. In 2005, a Cochrane review by Khan et al. [80] on 12 selected trials concluded that open repair had a lower risk of re-rupture (3.5 %) than non-operative repair (12.6 %). Comparing open and percutaneous repairs, the re-rupture rates were 4.3 and 2.1 % with overall complication rates (excluding re-rupture) of 26.1 and 8.3 %, respectively. Most recently, however, Soroceanu et al. [81], in a meta-analysis of ten carefully selected trials, recognized the evolving role of functional rehabilitation programmes in our treatment armamentarium. They reported re-rupture rates that were comparable in both the operative and non-operative groups at centres that use functional rehabilitation and early range of motion protocols (Table 3).
Until recently, the lack of a universal consistent scoring protocol for evaluating outcomes of AT rupture treatment makes direct comparison of different studies difficult and therefore unreliable. Nilsson-Helander et al. [82] designed the Achilles tendon Total Rupture Score (ATRS), a patient reported tool based on symptoms and physical activity for measuring treatment outcomes. This is the first reported validated score with a high reliability (intra-class correlation coefficient of 0.98), responsiveness and sensitivity [83]. In 2010, the same group showed in a Level 1 study that there was no difference between operative and non-operative treatment of rupture when evaluated using the ATRS score [83].
Further to the outcome parameters discussed above, there is fair evidence in the literature to suggest improved plantar flexion strength and a tendency towards a higher sporting activity resumption rate post-operatively. Cost analysis favours non-operative treatment, but percutaneous repair may be equivalent to open repair [84]. There is a clear trend towards a more functional rehabilitation programme with early return to activity and good clinical results irrespective of the treatment method.
Conclusion
The incidence of AT ruptures has increased in recent years. This may be due to increased participation in sporting activities coupled with greater awareness amongst hospital doctors and family practitioners. Whilst the exact aetiology of rupture is unknown, diagnosis is often obvious after a detailed history and examination. Clinical examination remains the gold standard, whilst the imaging modality of choice is MRI. There is inconclusive evidence in the literature to recommend one treatment option against another. In centres offering functional rehabilitation, non-operative treatment and early mobilization achieve excellent results. Currently, open repairs may still be preferable in younger and more physically demanding individuals requiring greater push-off strength, although there are risks of wound complications. Modern percutaneous techniques followed by early functional rehabilitation are becoming increasingly popular with good early results.
References
O’Brien M (1992) Functional anatomy and physiology of tendons. Clin Sports Med 11(3):505–520
Chalmers J (2000) Review article: treatment of Achilles tendon ruptures. J OrthopSurg (Hong Kong) 8(1):97–99
Landvater SJ, Renström PA (1992) Complete Achilles tendon ruptures. Clin Sports Med 11(4):741–758
Abrahamsen K (1923) Ruptura tendinis Achillis. UgesskrLaeger 85:279–285
Lea RB, Smith L (1972) Non-surgical treatment of tendo Achilles rupture. J Bone Joint Surg Am 54(7):1398–1407
Moller A, Anstrom M, Westlin NE (1996) Increasing incidence of Achilles tendon rupture. Acta Orthop Scand 67:277–279
Carden DG, Noble J, Chalmers J et al (1987) Rupture of the calcaneal tendon: the early and late management. J Bone Joint Surg Br 69(3):416–420
Boyden EM, Kitaoka HB, Cahalan TD et al (1995) Late versus early repair of Achilles tendon rupture: clinical and biomechanical evaluation. Clin Orthop Relat Res 317:150–158
Jozsa L, Kvist M, Balint JB et al (1989) The role of recreational sports activity in Achilles tendon rupture: a clinical, pathoanatomical and sociological study of 292 cases. Am J Sports Med 17:338–343
Rufai A, Ralphs JR, Benjamin M (1995) Structure and histopathology of the insertional region of the human Achilles tendon. J Orthop Res 13(4):585–593
Mayer L (1916) The physiological method of tendon transplantation. Surg Gynecol Obstet 22:182–197
Carr AJ, Norris SH (1989) The blood supply of the calcaneal tendon. J Bone Joint Surg Br 71(1):100–101
Astrom M, Westlin N (1994) Blood flow in the human Achilles tendon assessed by Doppler flowmetry. J Orthop Res 12(2):246–252
Stilwell DL Jr (1957) The innervation of tendons and aponeuroses. Am J Anat 100(3):289–317
Coombs RRH, Klenerman L, Narcisi P et al (1980) Collagen typing in Achilles tendon rupture. In: Proceedings of the British orthopaedic research society. J Bone Joint Surg Am 62B(2):258
Maffulli N, Ewen SW, Waterston SW et al (2000) Tenocytes from ruptured and tendinopathic Achilles tendons produce greater quantities of type III collagen than tenocytes from normal Achilles tendons. An in vitro model of human tendon healing. Am J Sports Med 28(4):499–505
Kannus P, Josza L, Jarvinnen M (2000) Basic science of tendons. In: Garrett WJ, Speer K, Kirkendall DT (eds) Principles and practice of orthopaedic sports medicine. Lipincott Williams & Wilkins, Philadelphia
Fukashiro S, Itoh M, Ichinose Y et al (1995) Ultrasonography gives directly but not noninvasively elastic characteristic of human tendon in vivo. Eur J Appl Physiol Occup Physiol 71(6):555–557
Arndt AN, Komi PV, Bruggeman GP et al (1998) Individual muscle contribution to the in vivo Achilles tendon force. ClinBiomech 13:532–541
O’Brien T (1984) The needle test for complete rupture of the Achilles tendon. J Bone Joint Surg Am 66(7):1099–1101
Maffulli N (1999) Rupture of the Achilles tendon. J Bone Joint Surg Am 81(7):1019–1036
Langergren C, Lindholm A (1958) Vascular distribution in the Achilles tendon; an angiographic and microangiographic study. Acta Chir Scand 116(5–6):491–495
Arner O, Lindholm A, Orell SR (1959) Histological changes in subcutaneous rupture of the Achilles tendon; a study of 74 cases. Acta Chir Scand 116(5–6):484–490
Jozsa L, Kannus P (1997) Histopathological findings in spontaneous tendon ruptures. Scand J Med Sci Sports 7(2):113–118
Fox JM, Blazina ME, Jobe FW et al (1975) Degeneration and rupture of the Achilles tendon. Clin Orthop Relat Res 107:221–224
Tuite DJ, Renstrom PA, O’Brien M (1997) The aging tendon. Scand J Med Sci Sports 7(2):72–77
Blevins FT, Hecker AT, Bigler GT et al (1994) The effects of donor age and strain rate on the biomechanical properties of bone-patellar tendon-bone allografts. Am J Sports Med 22(3):328–333
Collins M, Raleigh SM (2009) Genetic risk factors for musculoskeletal soft tissue injuries. Med Sport Sci 54:136–149
Barfred T (1973) Achilles tendon rupture: aetiology and pathogenesis of subcutaneous rupture assessed on the basis of literature and rupture experiments on rats. Acta Orthop Scand 152(S):3–126
Inglis AE, Sculco TP (1981) Surgical repair of ruptures of the tendo Achilles. Clin Orthop Relat Res 156:160–169
Barfred T (1971) Experimental rupture of the Achilles tendon. Comparison of various types of experimental rupture in rats. Acta Orthop Scand 42(6):528–543
Clement DB, Taunton JE, Smart GW (1984) Achilles tendinitis and peritendinitis: etiology and treatment. Am J Sports Med 12(3):179–184
DiStefano VJ, Nixon JE (1973) Ruptures of the Achilles tendon. J Sports Med 1(2):34–37
Kennedy JC, Willis RB (1976) The effects of local steroid injections on tendons: a biomechanical and microscopic correlative study. Am J Sports Med 4(1):11–21
Shrier I, Matheson GO, Kohl HW III (1996) Achilles tendonitis: are corticosteroid injections useful or harmful? Clin J Sports Med 6(4):245–250
Royer RJ, Pierfitte C, Netter P (1994) Features of tendon disorders with fluroquinolones. Therapie 49(1):75–76
Barge-Caballero E, Crespo-Leiro M, Paniagua-Martin M et al (2008) Quinolone-related Achilles tendonpathy in heart transplant patients: incidence and risk factors. J Heart Lung Transpl 27(1):46–51
Melhus A (2005) Fluoroquinolones and tendon disorders. Expert Opin Drug Saf 4(2):299–309
Petersen W, Pufe T, Zantop T et al (2004) Expression of VEGFR-1 and VEGFR-2 in degenerative Achilles tendons. Clin Orthop Relat Res 420:286–291
Webbon PM (1977) A post-mortem study of equine digital flexor tendons. Equine Vet J 9(2):61–67
Wilson A, Goodship AE (1994) Exercise-induced hyperthermia as a possible mechanism for tendon degeneration. J Biomech 27(7):899–905
Hattrup SJ, Johnson KA (1985) A review of ruptures of the Achilles tendon. Foot Ankle 6(1):34–38
Arner O, Lindholm A (1959) Subcutaneous rupture of the Achilles tendon: a study of 92 cases. Acta Chir Scand Suppl 116(S239):1–51
Krueger-Franke M, Siebert CH, Scherzer S (1995) Surgical treatment of ruptures of the Achilles tendon: a review of long term results. Br J Sports Med 29(2):121–125
Inglis AE, Scott WN, Sculco TP et al (1976) Ruptures of the tendo Achilles: an objective assessment of surgical and non-surgical treatment. J Bone Joint Surg Am 58(7):990–993
Maffulli N (1998) The clinical diagnosis of subcutaneous tear of the Achilles tendon. Am J Sports Med 26(2):266–270
Kager H (1939) Zur Klinik und Diagnostik des Achillesshnenrisses. Chirurgie 11:691–695
Toygar O (1947) Subkutane rupture der Achillesschne (Diagnostik und Behandlungsergebnisse). Helvet Chir Acta 14:209–231
Bleakney RR, Tallon C, Wong JK et al (2002) Long term ultrasonographic features of the Achilles tendon after rupture. Clin J Sports Med 12(5):273–278
Barbolini G, Monetti G, Montorsi A et al (1988) Results with high definition sonography in the evaluation of Achilles tendon conditions. Int J Sports Traumatol 10:225–234
Kalebo P, Allenmark C, Peterson L et al (1992) Diagnostic value of ultrasonography in partial ruptures of the Achilles tendon. Am J Sports Med 20(4):378–381
Mortensen HM, Skov O, Jensen PE (1999) Early motion of the ankle after operative treatment of a rupture of the Achilles tendon. A prospective, randomized clinical and radiographic study. J Bone Joint Surg Am 81(7):983–990
Nistor L (1981) Surgical and non-surgical treatment of Achilles tendon rupture: a prospective randomised study. J Bone Joint Surg Am 63(3):394–399
Saleh M, Marshall PD, Senior R et al (1992) The Sheffield splint for controlled early mobilization after rupture of the calcaneal tendon: a prospective randomized comparison with plaster treatment. J Bone Joint Surg Br 74(2):206–209
Wallace RG, Traynor IE, Kernohan WG et al (2004) Combined conservative and orthotic management of acute ruptures of the Achilles tendon. J Bone Joint Surg Am 86-A(6):1198–1202
Ingvar J, Tagil M, Eneroth M (2005) Non-operative treatment of Achilles tendon rupture: 196 consecutive patients with a 7 % re-rupture rate. Acta Orthop 76(4):597–601
Twaddle BC, Poon P (2007) Early motion for Achilles tendon ruptures: is surgery important? A randomized prospective study. Am J Sports Med 35(12):2033–2038
Lemaire R, Popovic N (1999) Diagnosis and treatment of acute ruptures of the Achilles tendon: current concepts review. Acta Orthop Belg 65(4):458–471
Cetti R, Henriksen LO, Jacobsen KS (1994) A new treatment of ruptured Achilles tendons: a prospective randomised study. Clin Orthop Relat Res 308:155–165
Zell RA, Santoro VM (2000) Augmented repair of acute Achilles tendon ruptures. Foot Ankle Int 21(6):469–474
Giannini S, Girolami M, Ceccarelli F et al (1994) Surgical repair of Achilles tendon ruptures using polypropylene braid augmentation. Foot Ankle Int 15(7):372–375
Lynn TA (1966) Repair of the torn Achilles tendon using the plantaris tendon as a reinforcing membrane. J Bone J Surg Am 48(2):268–272
Maffulli N, Tallon C, Wong J et al (2003) Early weight bearing and ankle mobilization after open repair of acute midsubstance tears of the Achilles tendon. Am J Sports Med 31(5):692–700
Costa ML, MacMillan K, Halliday D et al (2006) Randomised controlled trials of immediate weight-bearing mobilisation for rupture of tendo Achilles. J Bone Joint Surg Br 88(1):69–77
Ma GW, Griffith TG (1977) Percutaneous repair of acute closed ruptured Achilles tendon: a new technique. Clin Orthop Relat Res 128:247–255
Haji A, Sahai A, Symes A et al (2004) Percutaneous versus open tendo Achilles repair. Foot Ankle Int 25(4):215–218
Bradley JP, Tibone JE (1990) Percutaneous and open surgical repairs of Achilles tendon ruptures: a comparative study. Am J Sports Med 18(2):188–195
Gorschewsky O, Pitzl M, Putz A et al (2004) Percutaneous repair of acute Achilles tendon rupture. Foot Ankle Int 25(4):219–224
Krackow KA, Thomas SC, Jones LC (1986) A new stitch for ligament tendon fixation: brief note. J Bone Joint Surg Am 68(5):764–766
Watson TW, Jurist KA, Yang KH et al (1995) The strength of Achilles tendon repair: an in vitro study of the biomechanical behaviour in human cadaver tendons. Foot Ankle Int 16(4):191–195
Hockenbury RT, Johns JC (1990) A biomechanical in vitro comparison of open versus percutaneous repair of tendon Achilles. Foot Ankle 11(2):67–72
Carmont MR, Maffulli N (2008) Modified percutaneous repair of ruptured Achilles tendon. Knee Surg Sports Traumatol Arthrosc 16(2):199–203
Bhandari M, Guyatt GH, Siddiqui F et al (2002) Treatment of acute Achilles tendon ruptures: a systematic overview and meta-analysis. Clin Orthop Relat Res 400:190–200
Cretnik A, Kosanovic M, Smrkolj V (2005) Percutaneous versus open repair of the ruptured Achilles tendon. A comparative study. Am J Sports Med 33(9):1369–1379
Goren D, Ayalon M, Nyska M (2005) Isokinetic strength and endurance after percutaneous and open surgical repair of Achilles tendon ruptures. Foot Ankle Int 26(4):286–290
Calder JD, Saxby TS (2005) Early, active rehabilitation following mini-open repair of Achilles tendon rupture: a prospective study. Br J Sports Med 39(11):857–859
Valente M, Crucil M, Alecci V et al (2012) Minimally invasive repair of acute Achilles tendon ruptures with Achillon device. Musculoskelet Surg 96(1):35–39
Chiodo CP, Glazebrook M, Bluman EM et al (2010) Diagnosis and treatment of acute Achilles tendon rupture. J Am Acad Orthop Surg 18(8):503–510
Wong J, Barrass V, Maffulli N (2002) Quantitative review of operative and nonoperative management of Achilles tendon ruptures. Am J Sports Med 30(4):565–575
Khan RJ, Fick D, Keogh A et al (2005) Treatment of acute Achilles tendon ruptures. A meta-analysis of randomized, controlled trials. J Bone Joint Surg Am 87(10):2202–2210
Soroceanu A, Sidhwa F, Aarabi S et al (2012) Surgical versus nonsurgical treatment of acute Achilles tendon rupture: a meta-analysis of randomized trials. J Bone Joint Surg Am 94(23):2136–2143
Nilsson-Helander K, Thomee R, Gravare-Silbernagel K et al (2007) The Achilles tendon total rupture score (ATRS). Am J Sports Med 35(3):421–426
Nilsson-Helander K, Silbernagel KG, Thomeé R, Faxén E, Olsson N, Eriksson BI, Karlsson J (2010) Acute achilles tendon rupture: a randomized, controlled study comparing surgical and nonsurgical treatments using validated outcome measures. Am J Sports Med 38(11):2186–2193
Ebinesan AD, Sarai BS, Walley GD et al (2008) Conservative, open or percutaneous repair for acute rupture of the Achilles tendon. Disabil Rehabil 30(20–22):1721–1725
Conflict of interest
The authors declare that they have no competing interests.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Thevendran, G., Sarraf, K.M., Patel, N.K. et al. The ruptured Achilles tendon: a current overview from biology of rupture to treatment. Musculoskelet Surg 97, 9–20 (2013). https://doi.org/10.1007/s12306-013-0251-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12306-013-0251-6