Abstract
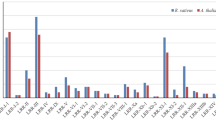
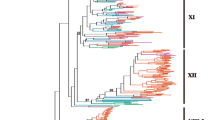
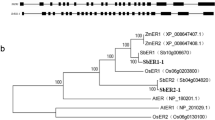
Leucine-rich repeat receptor-like kinases (LRR-RLKs) represent the largest subgroup of receptor-like kinases (RLKs) in plants. While some LRR-RLK members play a role in regulating various plant growth processes related to morphogenesis, disease resistance, and stress response, the functions of most LRR-RLK genes remain unclear. In this study, we identified 397 LRR-RLK genes from the genome of Camellia sinensis and categorized them into 16 subfamilies. Approximately 62% of CsLRR-RLK genes are situated in regions resulting from segmental duplications, suggesting that the expansion of CsLRR-RLK genes is due to segmental duplications. Analysis of gene expression patterns revealed differential expression of CsLRR-RLK genes across different tissues and in response to stress. Furthermore, we demonstrated that CssEMS1 localizes to the cell membrane and can complement Arabidopsis ems1 mutant. This study is the initial in-depth evolutionary examination of LRR-RLKs in tea and provides a basis for future investigations into their functionality.











Similar content being viewed by others
Data availability
The data supporting the findings of this study are available in the supplementary material.
References
Albrecht C, Russinova E, Hecht V, Baaijens E, de Vries S (2005) The Arabidopsis thaliana SOMATIC embryogenesis receptor-like kinases1 and 2 control male sporogenesis. Plant Cell 17(12):3337–3349. https://doi.org/10.1105/tpc.105.036814
Bailey TL, Boden M, Buske FA et al (2009) MEME SUITE: tools for motif discovery and searching. Nucleic Acids Res 37(Web Server issue):W202-208. https://doi.org/10.1093/nar/gkp335
Brandt B, Hothorn M (2016) SERK co-receptor kinases. Curr Biol 26(6):R225-226. https://doi.org/10.1016/j.cub.2015.12.014
Canales C, Bhatt AM, Scott R, Dickinson H (2002) EXS, a putative LRR receptor kinase, regulates male germline cell number and tapetal identity and promotes seed development in Arabidopsis. Curr Biol 12(20):1718–1727. https://doi.org/10.1016/s0960-9822(02)01151-x
Chen C, Chen H, Zhang Y et al (2020a) TBtools: an integrative toolkit developed for interactive analyses of big biological data. Mol Plant 13(8):1194–1202. https://doi.org/10.1016/j.molp.2020.06.009
Chen JD, Zheng C, Ma JQ et al (2020b) The chromosome-scale genome reveals the evolution and diversification after the recent tetraploidization event in tea plant. Hortic Res 7:63. https://doi.org/10.1038/s41438-020-0288-2
Chinchilla D, Shan L, He P, de Vries S, Kemmerling B (2009) One for all: the receptor-associated kinase BAK1. Trends Plant Sci 14(10):535–541. https://doi.org/10.1016/j.tplants.2009.08.002
DeYoung BJ, Bickle KL, Schrage KJ, Muskett P, Patel K, Clark SE (2006) The CLAVATA1-related BAM1, BAM2 and BAM3 receptor kinase-like proteins are required for meristem function in Arabidopsis. Plant J 45(1):1–16. https://doi.org/10.1111/j.1365-313X.2005.02592.x
Eyüboglu B, Pfister K, Haberer G et al (2007) Molecular characterisation of the STRUBBELIG-RECEPTOR FAMILY of genes encoding putative leucine-rich repeat receptor-like kinases in Arabidopsis thaliana. BMC Plant Biol 7:16. https://doi.org/10.1186/1471-2229-7-16
Finn RD, Coggill P, Eberhardt RY et al (2016) The Pfam protein families database: towards a more sustainable future. Nucleic Acids Res 44(D1):D279-285. https://doi.org/10.1093/nar/gkv1344
Gómez-Gómez L, Boller T (2000) FLS2: an LRR receptor-like kinase involved in the perception of the bacterial elicitor flagellin in Arabidopsis. Mol Cell 5(6):1003–1011. https://doi.org/10.1016/s1097-2765(00)80265-8
He Y, Zhou J, Shan L, Meng X (2018) Plant cell surface receptor-mediated signaling-a common theme amid diversity. J Cell Sci. https://doi.org/10.1242/jcs.209353
Hord CL, Chen C, Deyoung BJ, Clark SE, Ma H (2006) The BAM1/BAM2 receptor-like kinases are important regulators of Arabidopsis early anther development. Plant Cell 18(7):1667–1680. https://doi.org/10.1105/tpc.105.036871
Huang J, Zhang T, Linstroth L et al (2016) Control of anther cell differentiation by the small protein ligand TPD1 and its receptor EMS1 in Arabidopsis. PLoS Genet 12(8):e1006147. https://doi.org/10.1371/journal.pgen.1006147
Katoh K, Standley DM (2013) MAFFT multiple sequence alignment software version 7: improvements in performance and usability. Mol Biol Evol 30(4):772–780. https://doi.org/10.1093/molbev/mst010
Kinoshita T, Caño-Delgado A, Seto H et al (2005) Binding of brassinosteroids to the extracellular domain of plant receptor kinase BRI1. Nature 433(7022):167–171. https://doi.org/10.1038/nature03227
Letunic I, Doerks T, Bork P (2015) SMART: recent updates, new developments and status in 2015. Nucleic Acids Res 43:D257-260. https://doi.org/10.1093/nar/gku949
Lin F, Li S, Wang K et al (2020) A leucine-rich repeat receptor-like kinase, OsSTLK, modulates salt tolerance in rice. Plant Sci 296:110465. https://doi.org/10.1016/j.plantsci.2020.110465
Liu PL, Du L, Huang Y, Gao SM, Yu M (2017) Origin and diversification of leucine-rich repeat receptor-like protein kinase (LRR-RLK) genes in plants. BMC Evol Biol 17(1):47. https://doi.org/10.1186/s12862-017-0891-5
Liu S, Lei J, Zhang J et al (2022) Genome-wide identification and analysis of wheat LRR-RLK family genes following Chinese wheat mosaic virus infection. Front Plant Sci 13:1109845. https://doi.org/10.3389/fpls.2022.1109845
Man J, Gallagher JP, Bartlett M (2020) Structural evolution drives diversification of the large LRR-RLK gene family. New Phytol 226(5):1492–1505. https://doi.org/10.1111/nph.16455
Marchler-Bauer A, Derbyshire MK, Gonzales NR et al (2015) CDD: NCBI’s conserved domain database. Nucleic Acids Res 43:D222-226. https://doi.org/10.1093/nar/gku1221
Mizuno S, Osakabe Y, Maruyama K et al (2007) Receptor-like protein kinase 2 (RPK 2) is a novel factor controlling anther development in Arabidopsis thaliana. Plant J 50(5):751–766. https://doi.org/10.1111/j.1365-313X.2007.03083.x
Moore RC, Purugganan MD (2003) The early stages of duplicate gene evolution. Proc Natl Acad Sci U S A 100(26):15682–15687. https://doi.org/10.1073/pnas.2535513100
Shi CC, Feng CC, Yang MM et al (2014) Overexpression of the receptor-like protein kinase genes AtRPK1 and OsRPK1 reduces the salt tolerance of Arabidopsis thaliana. Plant Sci 217–218:63–70. https://doi.org/10.1016/j.plantsci.2013.12.002
Shiu SH, Bleecker AB (2001) Plant receptor-like kinase gene family: diversity, function, and signaling. Sci STKE 113:re22. https://doi.org/10.1126/stke.2001.113.re22
Shiu SH, Bleecker AB (2001b) Receptor-like kinases from Arabidopsis form a monophyletic gene family related to animal receptor kinases. Proc Natl Acad Sci U S A 98(19):10763–10768. https://doi.org/10.1073/pnas.181141598
Shpak ED (2013) Diverse roles of ERECTA family genes in plant development. J Integr Plant Biol 55(12):1238–1250. https://doi.org/10.1111/jipb.12108
Stamatakis A (2006) RAxML-VI-HPC: maximum likelihood-based phylogenetic analyses with thousands of taxa and mixed models. Bioinformatics 22(21):2688–2690. https://doi.org/10.1093/bioinformatics/btl446
Sun J, Li L, Wang P, Zhang S, Wu J (2017) Genome-wide characterization, evolution, and expression analysis of the leucine-rich repeat receptor-like protein kinase (LRR-RLK) gene family in Rosaceae genomes. BMC Genom 18(1):763. https://doi.org/10.1186/s12864-017-4155-y
Sun X, Wang GL (2011) Genome-wide identification, characterization and phylogenetic analysis of the rice LRR-kinases. PLoS ONE 6(3):e16079. https://doi.org/10.1371/journal.pone.0016079
Sun Y, Wang X, Liu F et al (2023) A Leucine-rich repeat receptor-like kinase TaBIR1 contributes to wheat resistance against Puccinia striiformis f. sp. tritici. Int J Mol Sci 24(7):5. https://doi.org/10.3390/ijms24076438
Wang X, Feng H, Chang Y et al (2020) Population sequencing enhances understanding of tea plant evolution. Nat Commun 11(1):4447. https://doi.org/10.1038/s41467-020-18228-8
Wang Y, Tang H, Debarry JD et al (2012) MCScanX: a toolkit for detection and evolutionary analysis of gene synteny and collinearity. Nucleic Acids Res 40(7):e49. https://doi.org/10.1093/nar/gkr1293
Wang Y, Yan X, Xu M et al (2023) Transmembrane kinase 1-mediated auxin signal regulates membrane-associated clathrin in Arabidopsis roots. J Integr Plant Biol 65(1):82–99. https://doi.org/10.1111/jipb.13366
Wei C, Yang H, Wang S et al (2018) Draft genome sequence of Camellia sinensis var. sinensis provides insights into the evolution of the tea genome and tea quality. Proc Natl Acad Sci U S A 115(18):E4151-e4158. https://doi.org/10.1073/pnas.1719622115
Xia E-H, Li F-D, Tong W et al (2019) Tea plant information archive: a comprehensive genomics and bioinformatics platform for tea plant. Plant Biotechnol J. https://doi.org/10.1111/pbi.13111
Xia E, Tong W, Hou Y et al (2020) The reference genome of tea plant and resequencing of 81 diverse accessions provide insights into its genome evolution and adaptation. Mol Plant 13(7):1013–1026. https://doi.org/10.1016/j.molp.2020.04.010
Xia EH, Zhang HB, Sheng J et al (2017) The tea tree genome provides insights into tea flavor and independent evolution of caffeine biosynthesis. Mol Plant 10(6):866–877. https://doi.org/10.1016/j.molp.2017.04.002
Yu Y, Song W, Zhai N et al (2023) PXL1 and SERKs act as receptor-coreceptor complexes for the CLE19 peptide to regulate pollen development. Nat Commun 14(1):3307. https://doi.org/10.1038/s41467-023-39074-4
Yuan N, Rai KM, Balasubramanian VK, Upadhyay SK, Luo H, Mendu V (2018a) Genome-wide identification and characterization of LRR-RLKs reveal functional conservation of the SIF subfamily in cotton (Gossypium hirsutum). BMC Plant Biol 18(1):185. https://doi.org/10.1186/s12870-018-1395-1
Yuan N, Yuan S, Li Z et al (2018b) Stress induced factor 2, a Leucine-rich repeat kinase regulates basal plant pathogen defense. Plant Physiol 176(4):3062–3080. https://doi.org/10.1104/pp.17.01266
Zan Y, Ji Y, Zhang Y, Yang S, Song Y, Wang J (2013) Genome-wide identification, characterization and expression analysis of populus leucine-rich repeat receptor-like protein kinase genes. BMC Genom 14:318. https://doi.org/10.1186/1471-2164-14-318
Zhang QJ, Li W, Li K et al (2020a) The Chromosome-level reference genome of tea tree unveils recent bursts of non-autonomous LTR retrotransposons in driving genome size evolution. Mol Plant 13(7):935–938. https://doi.org/10.1016/j.molp.2020.04.009
Zhang W, Zhang Y, Qiu H et al (2020b) Genome assembly of wild tea tree DASZ reveals pedigree and selection history of tea varieties. Nat Commun 11(1):3719. https://doi.org/10.1038/s41467-020-17498-6
Zhang X, Chen S, Shi L et al (2021) Haplotype-resolved genome assembly provides insights into evolutionary history of the tea plant Camellia sinensis. Nat Genet 53(8):1250–1259. https://doi.org/10.1038/s41588-021-00895-y
Zhang Z, Liu Z, Li S et al (2022) Effect of prior drought and heat stress on Camellia sinensis transcriptome changes to Ectropis oblique (Lepidoptera: Geometridae) resistance. Genomics 114(6):110506. https://doi.org/10.1016/j.ygeno.2022.110506
Zhao DZ, Wang GF, Speal B, Ma H (2002) The excess microsporocytes1 gene encodes a putative leucine-rich repeat receptor protein kinase that controls somatic and reproductive cell fates in the Arabidopsis anther. Genes Dev 16(15):2021–2031
Zhou F, Guo Y, Qiu LJ (2016) Genome-wide identification and evolutionary analysis of leucine-rich repeat receptor-like protein kinase genes in soybean. BMC Plant Biol 16:58. https://doi.org/10.1186/s12870-016-0744-1
Zipfel C, Kunze G, Chinchilla D et al (2006) Perception of the bacterial PAMP EF-Tu by the receptor EFR restricts Agrobacterium-mediated transformation. Cell 125(4):749–760. https://doi.org/10.1016/j.cell.2006.03.037
Acknowledgements
This work was supported by the grants from General Research Projects of Zhejiang Provincial Department of Education (Y202351039), Huzhou Science and Technology Plan Project (2023GZ44), and Research Program of Huzhou College (2023HXKM09)
Author information
Authors and Affiliations
Contributions
ZZ designed the research. ZZ and FY wrote the manuscript. FY, KH, TL and ZM performed the identification of LRR-RLKs, protein structure, evolution analyses, and expression analyses. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Table S1. Amino acid sequences of tea LRR-RLKs discovered in the current investigation. (TXT 349 KB)
12298_2024_1458_MOESM3_ESM.xlsx
Table S3. The comprehensive data on LRR-RLKs associated with tea as revealed in this current investigation. (XLSX 32 KB)
12298_2024_1458_MOESM9_ESM.pdf
Figure S1. Phylogenetic tree of LRR-RLK genes created using LRR-RLK genes sourced from Arabidopsis, rice, and tea. (PDF 878 KB)
Figure S2. The classification of groups for tea, rice and Arabidopsis LRR-RLK proteins. (PDF 150 KB)
12298_2024_1458_MOESM11_ESM.pdf
Figure S3. The conserved motifs identified in tea LRR-RLK proteins. The ten anticipated conserved structural domains are denoted by boxes of various colors (PDF 2416 KB)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhang, Z., Ye, F., Hu, K. et al. New insights into evolution and functional diversification of Camellia sinensis LRR-RLKs. Physiol Mol Biol Plants 30, 851–866 (2024). https://doi.org/10.1007/s12298-024-01458-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12298-024-01458-1