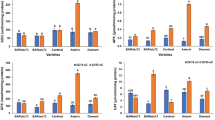
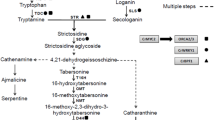
Abstract
Low temperature (cold) stress is one of the major abiotic stress conditions affecting crop productivity worldwide. Nitric oxide (NO) is a dynamic signaling molecule that interacts with various stress regulators and provides abiotic stress tolerance. Stress enhanced NO contributes to S-nitrosothiol accumulation which causes oxidation of the –SH group in proteins leading to S-nitrosation, a post-translational modification. Cold stress induced in vivo S-nitrosation of > 240 proteins majorly belonging to stress/signaling/redox (myrosinase, SOD, GST, CS, DHAR), photosynthesis (RuBisCO, PRK), metabolism (FBA, GAPDH, TPI, SBPase), and cell wall modification (Beta-xylosidases, alpha-l-arabinogalactan) in different crop plants indicated role of NO in these important cellular and metabolic pathways. NO mediated regulation of a transcription factor CBF (C-repeat Binding Factor, a transcription factor) at transcriptional and post-translational level was shown in Solanum lycopersicum seedlings. NO donor priming enhances seed germination, breaks dormancy and provides tolerance to stress in crops. Its role in averting stress, promoting seed germination, and delaying senescence paved the way for use of NO and NO releasing compounds to prevent crop loss and increase the shelf-life of fruits and vegetables. An alternative to energy consuming and expensive cold storage led to development of a storage device called “shelf-life enhancer” that delays senescence and increases shelf-life at ambient temperature (25–27 °C) using NO donor. The present review summarizes NO research in plants and exploration of NO for its translational potential to improve agricultural yield and post-harvest crop loss.



Similar content being viewed by others
References
Abat JK, Deswal R (2009) Differential modulation of S-nitrosoproteome of Brassica juncea by LT: change in S‐nitrosylation of Rubisco is responsible for the inactivation of its carboxylase activity. Proteomics 9(18):4368–4380. https://doi.org/10.1002/pmic.200800985
Abat JK, Mattoo AK, Deswal R (2008) S-nitrosylated proteins of a medicinal CAM plant Kalanchoe pinnata–ribulose‐1,5‐bisphosphate carboxylase/oxygenase activity targeted for inhibition. FEBS J 275(11):2862–2872. https://doi.org/10.1111/j.1742-4658.2008.06425.x
Ahmadi S, Vafaee Y, Koushesh Saba M (2020) Preharvest application of sodium nitroprusside improves tomato fruit quality and alleviates chilling injury during cold storage. Int J Veg Sci 26(4):364–378. https://doi.org/10.1080/19315260.2019.1636444
Ahuja I, Kissen R, Hoang L et al (2021) The imaging of Guard cells of thioglucosidase (TGG) mutants of Arabidopsis further links plant chemical defence systems with physical defence barriers. Cells 10(2):227. https://doi.org/10.3390/cells10020227
Alegre S, Pascual J, Trotta A et al (2020) Evolutionary conservation and post-translational control of S-adenosyl-L-homocysteine hydrolase in land plants. PLoS ONE 15(7):e0227466. https://doi.org/10.1371/journal.pone.0227466
Anand P, Hausladen A, Wang YJ et al (2014) Identification of S-nitroso-CoA reductases that regulate protein S-nitrosylation. Proc Natl Acad Sci 111(52):18572–18577. https://doi.org/10.1073/pnas.1417816112
Astier J, Lindermayr C (2012) Nitric oxide-dependent posttranslational modification in plants: an update. Int J Mol Sci 13(11):15193–15208. https://doi.org/10.3390/ijms131115193
Bateman RL, Rauh D, Tavshanjian B, Shokat KM (2008) Human carbonyl reductase1 is an S-nitrosoglutathione reductase. J Biol Chem 283(51):35756–35762. https://doi.org/10.1074/jbcM807125200
Bauer K, Nayem S, Lehmann M et al (2023) β-D-XYLOSIDASE 4 modulates systemic immune signaling in Arabidopsis thaliana. Front Plant Sci 13:1096800. https://doi.org/10.3389/fpls.2022.1096800
Benhar M, Forrester MT, Stamler JS (2009) Protein denitrosylation: enzymatic mechanisms and cellular functions. Nat Rev Mol Cell Biol 10(10):721–732. https://doi.org/10.1038/nrm2764
Burow M, Rice M, Hause B et al (2007) Cell-and tissue-specific localization and regulation of the epithiospecifier protein in Arabidopsis thaliana. Plant Mol Biol 64:173–185. https://doi.org/10.1007/s11103-007-9143-1
Cai B, Ning Y, Li Q et al (2022) Effects of the chloroplast fructose-1,6-bisphosphate aldolase gene on growth and low-temperature tolerance of tomato. Int J Mol Sci 23(2):728. https://doi.org/10.3390/ijms23020728
Castro-Torres E, Jiménez‐Sandoval P, Romero‐Romero S et al (2019) Structural basis for the modulation of plant cytosolic triosephosphate-isomerase activity by mimicry of redox‐based modifications. Plant J 99(5):950–964. https://doi.org/10.1111/tpj.14375
Chaki M, Shekariesfahlan A, Ageeva A et al (2015) Identification of nuclear target proteins for S-nitrosylation in pathogen-treated Arabidopsis thaliana cell cultures. Plant Sci 238:115–126. https://doi.org/10.1016/j.plantsci.2015.06.011
Chen W, Taylor NL, Chi Y et al (2014) The metabolic acclimation of Arabidopsis thaliana to arsenate is sensitized by the loss of mitochondrial LIPOAMIDE DEHYDROGENASE2, a key enzyme in oxidative metabolism. Plant Cell Env 37(3):684–695. https://doi.org/10.1111/pce.12187
Chen L, Wu R, Feng J et al (2020) Transnitrosylation mediated by the non-canonical catalase ROG1 regulates nitric oxide signaling in plants. Dev Cell 53(4):444–457. https://doi.org/10.1016/j.devcel.2020.03.020
Corpas FJ, Barroso JB (2017) Lead-induced stress, which triggers the production of nitric oxide (NO) and superoxide anion(O2·-) in Arabidopsis peroxisomes, affects catalase activity. Nitric Oxide 68:103–110. https://doi.org/10.1016/j.niox.2016.12.010
Corpas FJ, Freschi L, Rodríguez-Ruiz M et al (2018) Nitro-oxidative metabolism during fruit ripening. J Exp Bot 69(14):3449–3463. https://doi.org/10.1093/jxb/erx453
Deng L, Pan X, Chen L et al (2013) Effects of preharvest nitric oxide treatment on ethylene biosynthesis and soluble sugars metabolism in ‘Golden Delicious’ apples. Postharvest Biol Technol 84:9–15. https://doi.org/10.1016/j.postharvbio.2013.03.017
do Carmo GC, Iastrenski LF, Debiasi TV et al (2021) Nanoencapsulation improves the protective effects of a nitric oxide donor on drought-stressed Heliocarpus popayanensis seedlings. Ecotoxicol Environ Saf 225:112713. https://doi.org/10.1016/j.ecoenv.2021.112713
Dominguez R, Holmes KC (2011) Actin structure and function. Annu Rev Biophys 40:169–186. https://doi.org/10.1146/annurev-biophys-042910-155359
Dorion S, Rivoal J (2018) Plant Nucleoside diphosphate kinase1: a housekeeping enzyme with moonlighting activity. Plant Signal Behav 13(6):e1475804. https://doi.org/10.1080/15592324.2018.1475804
Edqvist J, Blomqvist K, Nieuwland J, Salminen TA (2018) Plant lipid transfer proteins: are we finally closing in on the roles of these enigmatic proteins? JLR 59(8):1374–1382. https://doi.org/10.1194/jlr.R083139
Ermakova M, Lopez-Calcagno PE, Furbank RT et al (2023) Increased sedoheptulose-1,7-bisphosphatase content in Setaria viridis does not affect C4 photosynthesis. Plant Physiol 191(2):885–893. https://doi.org/10.1093/plphys/kiac484
Falk A, Taipalensuu J, Ek B et al (1995) Characterization of rapeseed myrosinase-binding protein. Planta 195:387–395. https://doi.org/10.1007/BF00202596
Feng Y, Fu X, Han L et al (2021) Nitric oxide functions as a downstream signal for melatonin-induced cold-tolerance in cucumber seedlings. Front Plant Sci 12:686545. https://doi.org/10.3389/fpls.2021.686545
Floryszak-Wieczorek J, Arasimowicz-Jelonek M (2017) The multifunctional face of plant carbonic anhydrase. Plant Physiol Biochem 112:362–368. https://doi.org/10.1016/j.plaphy.2017.01.007
Fu L, Wang M, Han B et al (2016) Arabidopsis myrosinase genes AtTGG4 and AtTGG5 are root-tip specific and contribute to auxin biosynthesis and root-growth regulation. Int J Mol Sci 17(6):892. https://doi.org/10.3390/ijms17060892
Gai YP, Yuan SS, Liu ZY et al (2018) Integrated Phloem sap mRNA and protein expression analysis reveals phytoplasma-infection responses in mulberry. MCP 17(9):1702–1719. https://doi.org/10.1074/mcp.RA118.000670
Garai S, Bhowal B, Kaur C et al (2021) What signals the glyoxalase pathway in plants? Physiol Mol Bio Plants. https://doi.org/10.1007/s12298-021-00991-7
Gazaryan IG, Krasnikov BF, Ashby GA et al (2002) Zinc is a potent inhibitor of thiol oxidoreductase activity and stimulates reactive oxygen species production by lipoamide dehydrogenase. J Biol Chem 277(12):10064–10072. https://doi.org/10.1074/jbc.M108264200
Gomes DG, Debiasi TV, Pelegrino MT et al (2022) Soil treatment with nitric oxide-releasing chitosan-nanoparticles protects the root system and promotes the growth of Soybean plants under copper stress. Plants 11:3245. https://doi.org/10.3390/plants11233245
Gorren AC, Schrammel A, Schmidt K, Mayer B (1996) Decomposition of S-Nitrosoglutathione in the Presence of copper ions and glutathione. Arch Biochem Biophys 330(2):219–228. https://doi.org/10.1006/abbi.1996.0247
Grudkowska M, Zagdańska B (2004) Multifunctional role of plant cysteine proteinases. Acta Biochim Pol 51(3):609–624. https://doi.org/10.18388/abp.2004_3547
Guo Q, Wu B, Chen W et al (2014) Effects of nitric oxide treatment on the cell wall softening-related enzymes and several hormones of papaya fruit during storage. Food Sci Technol Int 20(4):309–317. https://doi.org/10.1177/1082013213484919
Guo X, Huang D, Jing G et al (2023) Nitric oxide-mediated DNA methylation enhances cold resistance in postharvest peach fruit. Food Chem 404:134660. https://doi.org/10.1016/j.foodchem.2022.134660
Gupta KJ (2023) System for delaying ripening of agricultural produce (Publication No. 20230087797). Retrieved from United States Patent and Trademark Office website
Gupta KJ, Hancock JT, Petrivalsky M et al (2020) Recommendations on terminology and experimental best practice associated with plant nitric oxide research. New Phytol 225(5):1828–1834. https://doi.org/10.1111/nph.16157
Gupta KJ, Kaladhar VC, Fitzpatrick TB et al (2022) Nitric oxide regulation of plant metabolism. Mol Plant 15(2):228–242. https://doi.org/10.1016/j.molp.2021.12.012
Gutiérrez-Luna FM, Hernández-Domínguez EE, Valencia-Turcotte LG, Rodríguez-Sotres R (2018) Pyrophosphate and pyrophosphatases in plants, their involvement in stress responses and their possible relationship to secondary metabolism. Plant Sci 267:11–19. https://doi.org/10.1016/j.plantsci.2017.10.016
Hasanuzzaman M, Nahar K, Gill SS et al (2017) Hydrogen peroxide pretreatment mitigates cadmium-induced oxidative stress in Brassica napus L.: an intrinsic study on antioxidant defense and glyoxalase systems. Front Plant Sci 8:115. https://doi.org/10.3389/fpls.2017.00115
Helms CC, Gladwin MT, Kim-Shapiro DB (2018) Erythrocytes and vascular function: oxygen and nitric oxide. Front Physiol 9:125. https://doi.org/10.3389/fphys.2018.00125
Hou Y, Guo Z, Li J, Wang PG (1996) Seleno compounds and glutathione peroxidase catalyzed decomposition of S-nitrosothiols. Biochem Biophys Res Commun 228(1):88–93. https://doi.org/10.1006/bbrc.1996.1620
Hwarari D, Guan Y, Ahmad B et al (2022) ICE-CBF-COR signaling cascade and its regulation in plants responding to cold stress. Int J Mol Sci 23(3):1549. https://doi.org/10.3390/ijms23031549
Islam MM, Tani C, Watanabe-Sugimoto M et al (2009) Myrosinases, TGG1 and TGG2, redundantly function in ABA and MeJA signaling in Arabidopsis guard cells. Plant Cell Physiol 50(6):1171–1175. https://doi.org/10.1093/pcp/pcp066
Ito E, Uemura T (2022) RAB GTPases and SNAREs at the trans-golgi network in plants. J Plant Res 135(3):389–403. https://doi.org/10.1007/s10265-022-01392-x
Jensen RG (2000) Activation of Rubisco regulates photosynthesis at high-temperature and CO2. PNAS 97(24):12937–12938. https://doi.org/10.1073/pnas.97.24.12937
Ji Y, Xu M, Liu Z et al (2022) Nucleocytoplasmic shuttling of ethylene response factor5 mediated by nitric oxide suppresses ethylene biosynthesis in apple fruit. New Phytol 234(5):1714. https://doi.org/10.1038/s41477-021-00944-8
Johnson KL, Jones BJ, Bacic A, Schultz CJ (2003) The fasciclin-like arabinogalactan proteins of Arabidopsis. A multigene family of putative cell adhesion molecules. Plant Physiol 133(4):1911–1925. https://doi.org/10.1104/pp.103.031237
Jourd’Heuil D, Laroux FS, Miles AM et al (1999) Effect of superoxide dismutase on the stability of S-nitrosothiols. Arch Biochem Biophys 361(2):323–330. https://doi.org/10.1006/abbi.1998.1010
Kang Y, Tong J, Liu W et al (2023) Comprehensive analysis of major latex-like protein family genes in Cucumber (Cucumis sativus L.) and their potential roles in Phytophthora Blight Resistance. Int J Mol Sci 24(1):784. https://doi.org/10.3390/ijms24010784
Kapoor I, Varshney U (2020) Diverse roles of nucleoside diphosphate kinase in genome stability and growth fitness. Curr Genet 66(4):671–682. https://doi.org/10.1007/s00294-020-01073-z
Kashyap P, Deswal R (2019) Two ICE isoforms showing differential transcriptional regulation by cold and hormones participate in Brassica juncea cold stress signaling. Gene 695:32–41. https://doi.org/10.1016/j.gene.2019.02.005
Kashyap P, Sehrawat A, Deswal R (2015) Nitric oxide modulates Lycopersicon esculentum C-repeat binding factor1(LeCBF1) transcriptionally as well as post-translationally by nitrosylation. Plant Physiol Biochem 96:115–123. https://doi.org/10.1016/j.plaphy.2015.07.032
Kato H, Takemoto D, Kawakita K (2013) Proteomic analysis of S-nitrosylated proteins in potato plant. Physiol Plant 148(3):371–386. https://doi.org/10.1111/j.1399-3054.2012.01684.x
Khan MN, Mobin M, Abbas ZK, Siddiqui MH (2017) Nitric oxide-induced synthesis of hydrogen sulfide alleviates osmotic stress in wheat seedlings through sustaining antioxidant enzymes, osmolyte accumulation and cysteine homeostasis. Nitric Oxide 68:91–102. https://doi.org/10.1016/j.niox.2017.01.001
Kochetov GA, Solovjeva ON (2014) Structure and functioning mechanism of transketolase. Biochim et Biochim Biophys Acta Proteins Proteom 1844(9):1608–1618. https://doi.org/10.1016/j.bbapap.2014.06.003
Kolbert ZS, Barroso JB, Brouquisse R et al (2019) A forty year journey: the generation and roles of NO in plants. Nitric Oxide 93:53–70. https://doi.org/10.1016/j.niox.2019.09.006
Kozuleva M, Goss T, Twachtmann M et al (2016) Ferredoxin: NADP(H) oxidoreductase abundance and location influences redox poise and stress tolerance. Plant Physiol 172(3):1480–1493. https://doi.org/10.1104/pp.16.01084
Kubienová L, Kopečný D, Tylichová M et al (2013) Structural and functional characterization of a plant S-nitrosoglutathione reductase from Solanum lycopersicum. Biochim 95(4):889–902. https://doi.org/10.1016/j.biochi.2012.12.009
Lee ES, Park JH, Wi SD et al (2021) Redox-dependent structural switch and CBF activation confer freezing-tolerance in plants. Nat Plants 7(7):914–922. https://doi.org/10.1038/s41477-021-00944-8
Li YY, Guo LN, Liang CZ et al (2022) Overexpression of Brassica napus cytosolic fructose-1,6-bisphosphatase and sedoheptulose-1,7-bisphosphatase genes significantly enhanced Tobacco growth and biomass. J Integr Agric 21(1):49–59. https://doi.org/10.1016/S2095-3119(20)63438-4
Li C, Yu W, Liao W (2022) Role of nitric oxide in postharvest senescence of fruits. Int J Mol Sci 23(17):10046. https://doi.org/10.3390/ijms231710046
Lindermayr C (2018) Crosstalk between reactive oxygen species and nitric oxide in plants: key role of S-nitrosoglutathione reductase. Free Radic Biol Med 122:110–115. https://doi.org/10.1016/j.freeradbiomed.2017.11.027
Lindermayr C, Saalbach G, Durner J (2005) Proteomic identification of S-nitrosylated proteins in Arabidopsis. Plant Physiol 137(3):921–930. https://doi.org/10.1104/pp.104.058719
Liszka A, Schimpf R, Cartuche Zaruma KI et al (2020) Three cytosolic NAD-malate dehydrogenase isoforms of Arabidopsis thaliana: on the crossroad between energy fluxes and redox signaling. Biochem J 477(19):3673–3693. https://doi.org/10.1042/BCJ20200240
Liu Y, Lu S, Liu K et al (2019) Proteomics: a powerful tool to study plant responses to biotic stress. Plant Methods 15:1–20. https://doi.org/10.1186/s13007-019-0515-8
Lopes-Oliveira PJ, Gomes DG, Pelegrino MT et al (2019) Effects of nitric oxide-releasing nanoparticles on neotropical tree seedlings submitted to acclimation under full sun in the nursery. Sci Rep 9(1):17371. https://doi.org/10.1038/s41598-019-54030-3
Lopes-Oliveira PJ, Oliveira HC, Kolbert Z, Freschi L (2021) The light and dark sides of nitric oxide: multifaceted roles of nitric oxide in plant responses to light. J Exp Bot 72(3):885–903. https://doi.org/10.1093/jxb/eraa504
Lutziger I, Oliver DJ (2001) Characterization of two cDNAs encoding mitochondrial lipoamide dehydrogenase from Arabidopsis. Plant Physiol 127(2):615–623. https://doi.org/10.1104/pp.010321
Ma Y, Fu L, Hussain Z et al (2019) Enhancement of storability and antioxidant systems of sweet cherry fruit by nitric oxide-releasing chitosan-nanoparticles (GSNO-CS NPs). Food Chem 285:10–21. https://doi.org/10.1016/j.foodchem.2019.01.156
Ma Y, Shafee T, Mudiyanselage AM et al (2023) Distinct functions of fascilin-like arabinogalactan proteins relate to domain structure. Plant Physiol 192(1):119–132. https://doi.org/10.1093/plphys/kiad097
Mao Y, Catherall E, Díaz-Ramos A et al (2023) The small subunit of Rubisco and its potential as an engineering target. J Exp Bot 74(2):543–561. https://doi.org/10.1093/jxb/erac309
Mattioli EJ, Rossi J, Meloni M et al (2022) Structural snapshots of nitrosoglutathione binding and reactivity underlying S-nitrosylation of photosynthetic GAPDH. Redox Biol 54:102387. https://doi.org/10.1016/j.redox.2022.102387
Mhamdi A, Queval G, Chaouch S et al (2010) Catalase function in plants: a focus on Arabidopsis mutants as stress-mimic models. J Exp Bot 61(15):4197–4220. https://doi.org/10.1093/jxb/erq282
Mouzo D, Bernal J, López-Pedrouso M et al (2018) Advances in the biology of seed and vegetative storage proteins based on two-dimensional electrophoresis coupled to mass spectrometry. Mol 23(10):2462. https://doi.org/10.3390/molecules23102462
Neupane P, Bhuju S, Thapa N, Bhattarai HK (2019) ATP synthase: structure, function and inhibition. Biomol Concepts 10(1):1–10. https://doi.org/10.1515/bmc-2019-0001
Niazi AK, Bariat L, Riondet C et al (2019) Cytosolic isocitrate dehydrogenase from Arabidopsis thaliana is regulated by glutathionylation. Antioxid 8(1):16. https://doi.org/10.3390/antiox8010016
Oliveira HC, Gomes BCR, Pelegrino MT, Seabra AB (2016) Nitric oxide-releasing chitosan-nanoparticles alleviate the effects of salt stress in maize plants. Nitric Oxide 61:10–19. https://doi.org/10.1016/j.niox.2016.09.010
Opassiri R, Pomthong B, Onkoksoong T et al (2006) Analysis of rice glycosyl hydrolase family 1 and expression of Os4bglu12 β-glucosidase. BMC Plant Biol 6:1–19. https://doi.org/10.1186/1471-2229-6-33
Ortega-Galisteo AP, Rodríguez-Serrano M, Pazmino DM et al (2012) S-Nitrosylated proteins in pea (Pisum sativum L.) leaf peroxisomes: changes under abiotic stress. J Exp Bot 63(5):2089–2103. https://doi.org/10.1093/jxb/err414
Oshunsanya SO, Nwosu NJ, Li Y (2019) Abiotic stress in agricultural crops under climatic conditions. In: Jhariya M, Banerjee A, Meena R, Yadav D (eds) Sustainable agriculture, forest and environmental management. Springer, Singapore, pp 71–100. https://doi.org/10.1007/978-981-13-6830-1_3
Palmieri MC, Lindermayr C, Bauwe H et al (2010) Regulation of plant glycine decarboxylase by S-nitrosylation and glutathionylation. Plant Physiol 152(3):1514–1528. https://doi.org/10.1104/pp.109.152579
Pande A, Mun BG, Khan M et al (2022) Nitric oxide signaling and its Association with ubiquitin-mediated proteasomal degradation in plants. Int J Mol Sci 23(3):1657. https://doi.org/10.3390/ijms23031657
Pareek A, Mishra D, Rathi D et al (2021) The small heat shock proteins, chaperonin 10, in plants: an evolutionary view and emerging functional diversity. EEB 182:104323. https://doi.org/10.1016/j.envexpbot.2020.104323
Pignocchi C, Fletcher JM, Wilkinson JE et al (2003) The function of ascorbate oxidase in Tobacco. Plant Physiol 132(3):1631–1641. https://doi.org/10.1104/pp.103.022798
Plohovska SH, Krasylenko YA, Yemets AI (2019) Nitric oxide modulates actin filament organization in Arabidopsis thaliana primary root cells at low-temperatures. Cell Biol Int 43(9):1020–1030. https://doi.org/10.1002/cbin.10931
Poria V, Saini JK, Singh S et al (2020) Arabinofuranosidases: characteristics, microbial production, and potential in waste valorization and industrial applications. Bioresour Technol 304:123019. https://doi.org/10.1016/j.biortech.2020.123019
Portis AR (2003) Rubisco activase–Rubisco’s catalytic chaperone. Photosynth Res 75:11–27. https://doi.org/10.1023/A:1022458108678
Puyaubert J, Fares A, Rézé N et al (2014) Identification of endogenously S-nitrosylated proteins in Arabidopsis plantlets:effect of cold stress on cysteine nitrosylation level. Plant Sci 215:150–156. https://doi.org/10.1016/j.plantsci.2013.10.014
Qu Y, Sakoda K, Fukayama H et al (2021) Overexpression of both Rubisco and Rubisco activase rescues rice photosynthesis and biomass under heat stress. Plant Cell Environ 44(7):2308–2320. https://doi.org/10.1111/pce.14051
Raines CA, Lloyd JC, Dyer TA (1999) New insights into the structure and function of sedoheptulose-1,7-bisphosphatase; an important but neglected Calvin-cycle enzyme. J Exp Bot 50(330):1–8. https://doi.org/10.1093/jxb/50.330.1
Randall SK (1992) Characterization of vacuolar calcium-binding proteins. Plant Physiol 100(2):859–867. https://doi.org/10.1104/pp1002859
Rask L, Andréasson E, Ekbom B et al (2000) Myrosinase: gene family evolution and herbivore defense in Brassicaceae. In: Doyle JJ, Gaut BS et al (eds) Plant Molecular Evolution. Springer, Dordrecht. https://doi.org/10.1007/978-94-011-4221-2_5
Romero LC, Aroca MÁ, Laureano-Marín AM et al (2014) Cysteine and cysteine-related signaling pathways in Arabidopsis thaliana. Mol Plant 7(2):264–276. https://doi.org/10.1093/mp/sst168
Roy S (2021) Role of nitric oxide as a double-edged Sword in Root Growth and Development. In: Mukherjee S, Baluška F (eds) Rhizobiology: Molecular Physiology of Plant roots. Springer, Cham. https://doi.org/10.1007/978-3-030-84985-6_11
Sako K, Nguyen HM, Seki M (2020) Advances in chemical priming to enhance abiotic stress tolerance in plants. Plant Cell Physiol 61(12):1995–2003. https://doi.org/10.1093/pcp/pcaa119
Sasikumar AN, Perez WB, Kinzy TG (2012) The many roles of the eukaryotic elongation factor1 complex. Wiley Interdiscip Rev RNA 3(4):543–555. https://doi.org/10.1002/wrna.1118
Schulze S, Westhoff P, Gowik U (2016) Glycine decarboxylase in C3, C4 and C3–C4 intermediate species. Curr Opin Plant Biol 31:29–35. https://doi.org/10.1016/j.pbi.2016.03.011
Seabra AB, Silveira NM, Ribeiro RV et al (2022) Nitric oxide-releasing nanomaterials: from basic research to potential biotechnological applications in agriculture. New Phytol 234(4):1119–1125. https://doi.org/10.1111/nph18073
Sehrawat A, Deswal R (2014) S-nitrosylation analysis in Brassica juncea apoplast highlights the importance of nitric oxide in cold-stress signaling. J Proteome Res 13(5):2599–2619. https://doi.org/10.1021/pr500082u
Sehrawat A, Abat JK, Deswal R (2013) RuBisCO depletion improved proteome coverage of cold-responsive S-nitrosylated targets in Brassica juncea. Front Plant Sci 4:342. https://doi.org/10.3389/fpls.2013.00342
Sehrawat A, Sougrakpam Y, Deswal R (2019) Cold modulated nuclear S-nitrosoproteome analysis indicates redox-modulation of novel Brassicaceae specific, myrosinase and napin in Brassica juncea. Environ Exp Bot 161:312–333. https://doi.org/10.1016/j.envexpbot.2018.10.010
Sharma RK, Parameswaran S (2018) Calmodulin-binding proteins: a journey of 40 years. Cell Calcium 75:89–100. https://doi.org/10.1016/jceca201809002
Sherameti I, Venus Y, Drzewiecki C et al (2008) PYK10, a β-glucosidase located in the endoplasmic reticulum, is crucial for the beneficial interaction between Arabidopsis thaliana and the endophytic fungus Piriformospora indica. Plant J 54(3):428–439. https://doi.org/10.1111/j.1365-313X.2008.03424.x
Silveira NM, Prataviera PJ, Pieretti JC et al (2021) Chitosan-encapsulated nitric oxide donors enhance physiological recovery of sugarcane plants after water deficit. Environ Exp Bot 190:104593. https://doi.org/10.1016/j.envexpbot.2021.104593
Skelly MJ, Malik SI, Le Bihan T et al (2019) A role for S-nitrosylation of the SUMO-conjugating enzyme SCE1 in plant immunity. PNAS 116(34):17090–17095. https://doi.org/10.1073/pnas.1900052116
Sliskovic I, Raturi A, Mutus B (2005) Characterization of the S-denitrosation activity of protein disulfide isomerase. J Biol Chem 280(10):8733–8741. https://doi.org/10.1074/jbcM408080200
Sobieszczuk-Nowicka E, Arasimowicz-Jelonek M, Tanwar UK, Floryszak-Wieczorek J (2022) Plant homocysteine, a methionine precursor and plant’s hallmark of metabolic disorders. Front Plant Sci 13:1044944. https://doi.org/10.3389/fpls.2022.1044944
Soboleva A, Frolova N, Bureiko K et al (2022) Dynamics of reactive-carbonyl species in pea Root nodules in response to polyethylene glycol (PEG)-Induced osmotic stress. Int J Mol Sci 23(5):2726. https://doi.org/10.3390/ijms23052726
Soliman M, Alhaithloul HA, Hakeem KR et al (2019) Exogenous nitric oxide mitigates nickel-induced oxidative damage in eggplant by upregulating antioxidants, osmolyte metabolism, and glyoxalase systems. Plants 8(12):562. https://doi.org/10.3390/plants8120562
Stomberski CT, Hess DT, Stamler JS (2019) Protein S-nitrosylation: determinants of specificity and enzymatic regulation of S-nitrosothiol-based signaling. Antioxid Redox Signal 30(10):1331–1351. https://doi.org/10.1089/ars.2017.7403
Stoyanovsky DA, Tyurina YY, Tyurin VA et al (2005) Thioredoxin and lipoic acid catalyze the denitrosation of low-molecular weight and protein S-nitrosothiols. J Am Chem Soc 127(45):15815–15823.
Thom SR, Bhopale VM, Milovanova TN et al (2012) Thioredoxin reductase linked to cytoskeleton by focal adhesion kinase reverses actin S-nitrosylation and restores neutrophil β2-integrin function. J Biol Chem 287(36):30346–30357. https://doi.org/10.1074/jbc.M112.355875
Timm S, Hagemann M (2020) Photorespiration-how is it regulated and how does it regulate overall plant metabolism? J Exp Bot 71(14):3955–3965. https://doi.org/10.1093/jxb/eraa183
Treffon P, Vierling E (2022) Focus on nitric oxide homeostasis: direct and indirect enzymatic regulation of protein denitrosation reactions in plants. Antioxid 11(7):1411. https://doi.org/10.3390/antiox11071411
Trujillo M, Alvarez MN, Peluffo G (1998) Xanthine oxidase-mediated decomposition of S-nitrosothiols. J Biol Chem 273(14):7828–7834. https://doi.org/10.1074/jbc.273.14.7828
Vaish S, Gupta D, Mehrotra R et al (2020) Glutathione S-transferase: a versatile protein family. 3 Biotech 10:1–19. https://doi.org/10.1007/s13205-020-02312-3
Voss I, Sunil B, Scheibe R, Raghavendra AS (2013) Emerging concept for the role of photorespiration as an important part of abiotic stress response. Plant Biol 15(4):713–722. https://doi.org/10.1111/j.1438-8677.2012.00710.x
Wang L, Tsuda K, Sato M et al (2009) Arabidopsis CaM binding protein CBP60g contributes to MAMP-induced SA accumulation and is involved in disease resistance against Pseudomonas syringae. PLoS Pathog 5(2):e1000301. https://doi.org/10.1371/journal.ppat.1000301
Ward OP (2011) Proteases. Compr Biotech. https://doi.org/10.1016/B978-0-08-088504-9.00222-1
Wei L, Zhang J, Wei S et al (2022) Nitric oxide alleviates salt stress through protein S-nitrosylation and transcriptional regulation in tomato seedlings. Planta 256(6):101. https://doi.org/10.1007/s00425-022-04015-w
Yamada K, Hasegawa T, Minami E et al (2003) Induction of myrosinase gene expression and myrosinase activity in radish hypocotyls by phototropic stimulation. J Plant Physiol 160(3):255–259. https://doi.org/10.1078/0176-1617-00950
Yokochi Y, Yoshida K, Hahn F et al (2021) Redox-regulation of NADP-malate dehydrogenase is vital for land plants under fluctuating light environment. PNAS 118(6):e2016903118. https://doi.org/10.1073/pnas.2016903118
Yu A, Xie Y, Pan X et al (2020) Photosynthetic phosphoribulokinase structures: enzymatic mechanisms and the redox regulation of the Calvin–Benson–Bassham cycle. Plant Cell 32(5):1556–1573. https://doi.org/10.1105/tpc.19.00642
Zaffagnini M, Fermani S, Costa A et al (2013) Plant cytoplasmic GAPDH: redox post-translational modifications and moonlighting properties. Front Plant Sci 4:450. https://doi.org/10.3389/fpls.2013.00450
Zaffagnini M, Michelet L, Sciabolini C et al (2014) High-resolution crystal structure and redox properties of chloroplastic triosephosphate isomerase from Chlamydomonas reinhardtii. Mol Plant 7(1):101–120. https://doi.org/10.1093/mp/sst139
Zaharah SS, Singh Z (2011) Mode of action of nitric oxide in inhibiting ethylene biosynthesis and fruit softening during ripening and cool storage of ‘Kensington Pride’ mango. Postharvest Biol Technol 62(3):258–266. https://doi.org/10.1016/j.postharvbio.2011.06.007
Zhan S, Zhang Q, Yao Y et al (2023) Cytosolic isocitrate dehydrogenase regulates plant stem cell maintenance in response to nutrient deficiency. Plant Physiol. https://doi.org/10.1093/plphys/kiad246
Zhang J, Liao W (2019) Protein S-nitrosylation in plant abiotic stresses. Funct Plant Biol 47(1):1–10. https://doi.org/10.1071/FP19071
Zhang CS, Hawley SA, Zong Y et al (2017) Fructose-1,6-bisphosphate and aldolase mediate glucose sensing by AMPK. Nature 548(7665):112–116. https://doi.org/10.1038/nature23275
Zhang T, Che F, Zhang H et al (2017) Effect of nitric oxide treatment on chilling injury, antioxidant enzymes and expression of the CmCBF1 and CmCBF3 genes in cold-stored Hami melon (Cucumis melo L.) fruit. Postharvest Biol Technol 127:88–98. https://doi.org/10.1016/j.postharvbio.2017.01.005
Zhang ZW, Luo S, Zhang GC et al (2017) Nitric oxide induces monosaccharide accumulation through enzyme S-nitrosylation. Plant Cell Environ 40(9):1834–1848. https://doi.org/10.1111/pce.12989
Zhang K, Su H, Zhou J et al (2019) Overexpressing the myrosinase gene TGG1 enhances stomatal defense against Pseudomonas syringae and delays flowering in Arabidopsis. Front Plant Sci 10:1230. https://doi.org/10.3389/fpls.2019.01230
Zhang ZW, Fu YF, Zhou YH et al (2019) Nitrogen and nitric oxide regulate Arabidopsis flowering differently. Plant Sci 284:177–184. https://doi.org/10.1016/j.plantsci.2019.04.015
Zhang S, Liu L, Wu Z et al (2023) S-nitrosylation of superoxide dismutase and catalase involved in promotion of fruit resistance to chilling stress: a case study on Ziziphus Jujube Mill. Postharvest Biol Technol 197:112210. https://doi.org/10.1016/j.postharvbio.2022.112210
Zhao R, Sheng J, Lv S et al (2011) Nitric oxide participates in the regulation of LeCBF1 gene expression and improves cold tolerance in harvested tomato fruit. Postharvest Biol Technol 62(2):121–126. https://doi.org/10.1016/j.postharvbio.2011.05.013
Zhou X, Joshi S, Khare T et al (2021) Nitric oxide, crosstalk with stress regulators and plant abiotic stress tolerance. Plant Cell Rep 40(8):1395–1414. https://doi.org/10.1007/s00299-021-02705-5
Zhu S, Liu M, Zhou J (2006) Inhibition by nitric oxide of ethylene biosynthesis and lipoxygenase activity in peach fruit during storage. Postharvest Biol Technol 42(1):41–48. https://doi.org/10.1016/j.postharvbio.2006.05.004
Acknowledgements
This work was partially supported by FRP grant provided by Institute of Eminence, University of Delhi [IOE/2021/12/FRP]. YS and PB availed senior research fellowship from UGC, India.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sougrakpam, Y., Babuta, P. & Deswal, R. Nitric oxide (NO) modulates low temperature-stress signaling via S-nitrosation, a NO PTM, inducing ethylene biosynthesis inhibition leading to enhanced post-harvest shelf-life of agricultural produce. Physiol Mol Biol Plants 29, 2051–2065 (2023). https://doi.org/10.1007/s12298-023-01371-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12298-023-01371-z