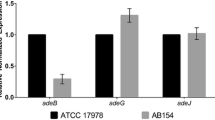
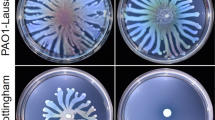
Abstract
Acinetobacter baumannii is a nosocomial bacterial pathogen, and infections attributed to this species are further complicated by a remarkable ability to acquire antimicrobial resistance genes and to survive in a desiccated state. While the antibiotic resistance and biofilm formation of A. baumannii is well-documented, less is known about the virulence attributes of this organism. Recent studies reported A. baumannii strains display a motility phenotype, which appears to be partially dependent upon Type IV pili, autoinducer molecules, and the response to blue light. In this study, we wanted to determine the prevalence of this trait in genetically diverse clinical isolates, and any additional required factors, and environmental cues that regulate motility. When strains are subjected to a wide array of stress conditions, A. baumannii motility is significantly reduced. In contrast, when extracellular iron is provided or salinity is reduced, motility is significantly enhanced. We further investigated whether the genes required for the production of lipopolysaccharide (lpsB) and K1 capsule (epsA/ptk) are required for motility as demonstrated in other Gram-negative bacteria. Transposon mutagenesis resulted in reduced motility by the insertion derivatives of each of these genes. The presence of the parental allele provided in trans, in the insertion mutant background, could only restore motility in the lpsB mutant. The production of core LPS directly contributes to the motility phenotype, while capsular polysaccharide may have an indirect effect. Further, the data suggest motility is regulated by extracellular conditions, indicating that A. baumannii is actively sensing the environment and responding accordingly.
Similar content being viewed by others
References
Adams, M.D., Goglin, K., Molyneaux, N., Hujer, K.M., Lavender, H., Jamison, J.J., MacDonald, I.J., Martin, K.M., Russo, T., Campagnari, A.A., andet al. 2008. Comparative genome sequence analysis of multidrug-resistant Acinetobacter baumannii. J. Bacteriol.190, 8053–8064.
Adams, D., Yee, L., Rimmer, J.A., Williams, R., Martin, H., and Ovington, C. 2011. Investigation and management of an A. baumannii outbreak in ICU. Br. J. Nurs.20,140, 142, 144–147.
Alarcon, I., Evans, D.J., and Fleiszig, S.M. 2009. The role of twitching motility in Pseudomonas aeruginosa exit from and translocation of corneal epithelial cells. Invest. Ophthalmol. Vis. Sci.50, 2237–2244.
Ali, A., Johnson, J.A., Franco, A.A., Metzger, D.J., Connell, T.D., Morris, J.G.Jr., and Sozhamannan, S. 2000. Mutations in the extracellular protein secretion pathway genes (eps) interfere with rugose polysaccharide production in and motility of Vibrio cholerae. Infect. Immun.68, 1967–1974.
Alsultan, A.A., Hamouda, A., Evans, B.A., and Amyes, S.G. 2009. Acinetobacter baumannii: emergence of four strains with novel bla (OXA-51-like) genes in patients with diabetes mellitus. J. Chemother.21, 290–295.
Banin, E., Vasil, M.L., and Greenberg, E.P. 2005. Iron and Pseudomonas aeruginosa biofilm formation. Proc. Natl. Acad. Sci. USA102, 11076–11081.
Beavers, S.F., Blossom, D.B., Wiemken, T.L., Kawaoka, K.Y., Wong, A., Goss, L., McCormick, M.I., Thoroughman, D., and Srinivasan, A. 2009. Comparison of risk factors for recovery of Acinetobacter baumannii during outbreaks at two Kentucky hospitals, 2006. Public Health Rep.124, 868–874.
Brisou, J. 1953. Essay on the system of the genus Achromobacter. Ann. Inst. Pasteur (Paris)84, 812–814.
Burrows, L.L. 2005. Weapons of mass retraction. Mol. Microbiol.57, 878–888.
Butler, M.T., Wang, Q., and Harshey, R.M. 2010. Cell density and mobility protect swarming bacteria against antibiotics. Proc. Natl. Acad. Sci. USA107, 3776–3781.
Caiazza, N.C., Shanks, R.M., and O’Toole, G.A. 2005. Rhamnolipids modulate swarming motility patterns of Pseudomonas aeruginosa. J. Bacteriol.187, 7351–7361.
Calhoun, J.H., Murray, C.K., and Manring, M.M. 2008. Multidrug-resistant organisms in military wounds from Iraq and Afghanistan. Clin. Orthop. Relat. Res.466, 1356–1362.
Chang, H.L., Tang, C.H., Hsu, Y.M., Wan, L., Chang, Y.F., Lin, C.T., Tseng, Y.R., Lin, Y.J., Sheu, J.J., Lin, C.W., andet al. 2009. Nosocomial outbreak of infection with multidrug-resistant Acinetobacter baumannii in a medical center in Taiwan. Infect. Control Hosp. Epidemiol.30, 34–38.
Chen, B.G., Turner, L., and Berg, H.C. 2007. The wetting agent required for swarming in Salmonella enterica serovar typhimurium is not a surfactant. J. Bacteriol.189, 8750–8753.
Choi, W.S., Kim, S.H., Jeon, E.G., Son, M.H., Yoon, Y.K., Kim, J.Y., Kim, M.J., Sohn, J.W., and Park, D.W. 2010. Nosocomial outbreak of carbapenem-resistant Acinetobacter baumannii in intensive care units and successful outbreak control program. J. Korean Med. Sci.25, 999–1004.
Choi, C.H., Lee, E.Y., Lee, Y.C., Park, T.I., Kim, H.J., Hyun, S.H., Kim, S.A., Lee, S.K., and Lee, J.C. 2005. Outer membrane protein 38 of Acinetobacter baumannii localizes to the mitochondria and induces apoptosis of epithelial cells. Cell Microbiol.7, 1127–1138.
Cisneros, J.M. and Rodriguez-Bano, J. 2002. Nosocomial bacteremia due to Acinetobacter baumannii: epidemiology, clinical features and treatment. Clin. Microbiol. Infect.8, 687–693.
Clemmer, K.M., Bonomo, R.A., and Rather, P.N. 2011. Genetic analysis of surface motility in Acinetobacter baumannii. Microbiology157, 2534–2544.
Cuthbertson, L., Mainprize, I.L., Naismith, J.H., and Whitfield, C. 2009. Pivotal roles of the outer membrane polysaccharide export and polysaccharide copolymerase protein families in export of extracellular polysaccharides in Gram-negative bacteria. Microbiol. Mol. Biol. Rev.73, 155–177.
D’Arezzo, S., Principe, L., Capone, A., Petrosillo, N., Petrucca, A., and Visca, P. 2011. Changing carbapenemase gene pattern in an epidemic multidrug-resistant Acinetobacter baumannii lineage causing multiple outbreaks in central Italy. J. Antimicrob. Chemother.66, 54–61.
Da Re, S. and Ghigo, J.M. 2006. A CsgD-independent pathway for cellulose production and biofilm formation in Escherichia coli. J. Bacteriol.188, 3073–3087.
Dalla-Costa, L.M., Coelho, J.M., Souza, H.A., Castro, M.E., Stier, C.J., Bragagnolo, K.L., Rea-Neto, A., Penteado-Filho, S.R., Livermore, D.M., and Woodford, N. 2003. Outbreak of carbapenem-resistant Acinetobacter baumannii producing the OXA-23 enzyme in Curitiba, Brazil. J. Clin. Microbiol.41, 3403–3406.
Dallo, S.F. and Weitao, T. 2010. Insights into Acinetobacter war-wound infections, biofilms, and control. Adv. Skin Wound Care23, 169–174.
Dijkshoorn, L., Nemec, A., and Seifert, H. 2007. An increasing threat in hospitals: multidrug-resistant Acinetobacter baumannii. Nat. Rev. Microbiol.5, 939–951.
Dixon, B. 2008. There’s the rub: infection control that spreads infection. Lancet Infect. Dis.8, 91–91.
Eijkelkamp, B.A., Hassan, K.A., Paulsen, I.T., and Brown, M.H. 2011. Investigation of the human pathogen Acinetobacter baumannii under iron limiting conditions. BMC Genomics12, 126.
Ellison, R.T.3rd, Giehl, T.J., and LaForce, F.M. 1988. Damage of the outer membrane of enteric Gram-negative bacteria by lactoferrin and transferrin. Infect. Immun.56, 2774–2781.
Falagas, M.E., Karveli, E.A., Kelesidis, I., and Kelesidis, T. 2007. Community-acquired Acinetobacter infections. Eur. J. Clin. Microbiol. Infect. Dis.26, 857–868.
Firehammer, B.D. 1987. Inhibition of growth and swarming of Proteus mirabilis and Proteus vulgaris by triclosan. J. Clin. Microbiol.25, 1312–1313.
Fong, J.C., Syed, K.A., Klose, K.E., and Yildiz, F.H. 2010. Role of Vibrio polysaccharide (vps) genes in VPS production, biofilm formation and Vibrio cholerae pathogenesis. Microbiology156, 2757–2769.
Forbes, S.J., Eschmann, M., and Mantis, N.J. 2008. Inhibition of Salmonella enterica serovar typhimurium motility and entry into epithelial cells by a protective antilipopolysaccharide monoclonal immunoglobulin A antibody. Infect. Immun.76, 4137–4144.
Forman, H.J. and Torres, M. 2001. Redox signaling in macrophages. Mol. Aspects Med.22, 189–216.
Gaddy, J.A., Tomaras, A.P., and Actis, L.A. 2009. The Acinetobacter baumannii 19606 OmpA protein plays a role in biofilm formation on abiotic surfaces and in the interaction of this pathogen with eukaryotic cells. Infect. Immun.77, 3150–3160.
Gootz, T.D. and Marra, A. 2008. Acinetobacter baumannii: an emerging multidrug-resistant threat. Expert Rev. Anti. Infect. Ther.6, 309–325.
Guerrero, D.M., Perez, F., Conger, N.G., Solomkin, J.S., Adams, M.D., Rather, P.N., and Bonomo, R.A. 2010. Acinetobacter baumannii-associated skin and soft tissue infections: recognizing a broadening spectrum of disease. Surg. Infect. (Larchmt)11, 49–57.
Halvorsen, J.F. 1963. Gliding Motility in the Organisms Bacterium anitratum (B5w), Moraxella lwoffi and Alkaligenes haemolysans, as compared to Moraxella nonliquefaciens. Acta. Pathol. Microbiol. Scand.59, 200–204.
Heilmann, C., Gerke, C., Perdreau-Remington, F., and Gotz, F. 1996. Characterization of Tn917 insertion mutants of Staphylococcus epidermidis affected in biofilm formation. Infect. Immun.64, 277–282.
Hod, E.A., Zhang, N., Sokol, S.A., Wojczyk, B.S., Francis, R.O., Ansaldi, D., Francis, K.P., Della-Latta, P., Whittier, S., Sheth, S., andet al. 2010. Transfusion of red blood cells after prolonged storage produces harmful effects that are mediated by iron and inflammation. Blood115, 4284–4292.
Hood, M.I., Jacobs, A.C., Sayood, K., Dunman, P.M., and Skaar, E.P. 2010. Acinetobacter baumannii increases tolerance to antibiotics in response to monovalent cations. Antimicrob. Agents Chemother.54, 1029–1041.
Huang, T.P., Somers, E.B., and Wong, A.C. 2006. Differential biofilm formation and motility associated with lipopolysaccharide/ exopolysaccharide-coupled biosynthetic genes in Stenotrophomonas maltophilia. J. Bacteriol.188, 3116–3120.
Jacobs, A.C., Hood, I., Boyd, K.L., Olson, P.D., Morrison, J.M., Carson, S., Sayood, K., Iwen, P.C., Skaar, E.P., and Dunman, P.M. 2010. Inactivation of phospholipase D diminishes Acinetobacter baumannii pathogenesis. Infect. Immun.78, 1952–1962.
Jamieson, W.D., Pehl, M.J., Gregory, G.A., and Orwin, P.M. 2009. Coordinated surface activities in Variovorax paradoxus EPS. BMC Microbiol.9, 124.
Joly-Guillou, M.L. 2005. Clinical impact and pathogenicity of Acinetobacter. Clin. Microbiol. Infect.11, 868–873.
Karthikeyan, K., Thirunarayan, M.A., and Krishnan, P. 2010. Coexistence of blaOXA-23 with blaNDM-1 and armA in clinical isolates of Acinetobacter baumannii from India. J. Antimicrob. Chemother.65, 2253–2254.
Kearns, D.B. and Losick, R. 2003. Swarming motility in undomesticated Bacillus subtilis. Mol. Microbiol.49, 581–590.
Ko, K.S., Suh, J.Y., Kwon, K.T., Jung, S.I., Park, K.H., Kang, C.I., Chung, D.R., Peck, K.R., and Song, J.H. 2007. High rates of resistance to colistin and polymyxin B in subgroups of Acinetobacter baumannii isolates from Korea. J. Antimicrob. Chemother.60, 1163–1167.
La Forgia, C., Franke, J., Hacek, D.M., Thomson, R.B.Jr., Robicsek, A., and Peterson, L.R. 2010. Management of a multidrug-resistant Acinetobacter baumannii outbreak in an intensive care unit using novel environmental disinfection: a 38-month report. Am. J. Infect. Control38, 259–263.
Lancero, H., Caberoy, N.B., Castaneda, S., Li, Y., Lu, A., Dutton, D., Duan, X.Y., Kaplan, H.B., Shi, W., and Garza, A.G. 2004. Characterization of a Myxococcus xanthus mutant that is defective for adventurous motility and social motility. Microbiology150, 4085–4093.
Lee, J., Bansal, T., Jayaraman, A., Bentley, W.E., and Wood, T.K. 2007. Enterohemorrhagic Escherichia coli biofilms are inhibited by 7-hydroxyindole and stimulated by isatin. Appl. Environ. Microbiol.73, 4100–4109.
Letunic, I. and Bork, P. 2011. Interactive tree of life v2: online annotation and display of phylogenetic trees made easy. Nucleic Acids Res.39, W475–478.
Lindberg, R.B., Wetzler, T.F., Newton, A., Howard, J.M., Davis, J.H., and Strawitz, J. 1955. The bacterial flora of the blood stream in the Korean battle casualty. Ann. Surg.141, 366–368.
Lindum, P.W., Anthoni, U., Christophersen, C., Eberl, L., Molin, S., and Givskov, M. 1998. N-Acyl-L-homoserine lactone autoinducers control production of an extracellular lipopeptide biosurfactant required for swarming motility of Serratia liquefaciens MG1. J. Bacteriol.180, 6384–6388.
Liu, J.B., Tang, T.S., and Xiao, D.S. 2004. Changes of free iron contents and its correlation with lipid peroxidation after experimental spinal cord injury. Chin. J. Traumatol.7, 229–232.
Loehfelm, T.W., Luke, N.R., and Campagnari, A.A. 2008. Identification and characterization of an Acinetobacter baumannii biofilm-associated protein. J. Bacteriol.190, 1036–1044.
Luke, N.R., Sauberan, S.L., Russo, T.A., Beanan, J.M., Olson, R., Loehfelm, T.W., Cox, A.D., St Michael, F., Vinogradov, E.V., and Campagnari, A.A. 2010. Identification and characterization of a glycosyltransferase involved in Acinetobacter baumannii lipopolysaccharide core biosynthesis. Infect. Immun.78, 2017–2023.
Marchaim, D., Navon-Venezia, S., Leavitt, A., Chmelnitsky, I., Schwaber, M.J., and Carmeli, Y. 2007. Molecular and epidemiologic study of polyclonal outbreaks of multidrug-resistant Acinetobacter baumannii infection in an Israeli hospital. Infect. Control Hosp. Epidemiol.28, 945–950.
Matsuyama, T., Bhasin, A., and Harshey, R.M. 1995. Mutational analysis of flagellum-independent surface spreading of Serratia marcescens 274 on a low-agar medium. J. Bacteriol.177, 987–991.
McQueary, C.N. and Actis, L.A. 2011. Acinetobacter baumannii biofilms: Variations among strains and correlations with other cell properties. J. Microbiol.49, 243–250.
Miller, J. 1972. Experiments in molecular genetics. Cold Spring Harbor Laboratory, Cold spring Harbor, New York, N.Y., USA.
Montefour, K., Frieden, J., Hurst, S., Helmich, C., Headley, D., Martin, M., and Boyle, D.A. 2008. Acinetobacter baumannii: an emerging multidrug-resistant pathogen in critical care. Crit. Care Nurse28, 15–25; quiz 26.
Mussi, M.A., Gaddy, J.A., Cabruja, M., Arivett, B.A., Viale, A.M., Rasia, R., and Actis, L.A. 2010. The opportunistic human pathogen Acinetobacter baumannii senses and responds to light. J. Bacteriol.192, 6336–6345.
Nwugo, C.C., Gaddy, J.A., Zimbler, D.L., and Actis, L.A. 2011. Deciphering the iron response in Acinetobacter baumannii: A proteomics approach. J. Proteomics74, 44–58.
O’Toole, G.A. and Kolter, R. 1998. Flagellar and twitching motility are necessary for Pseudomonas aeruginosa biofilm development. Mol. Microbiol.30, 295–304.
Park, Y.K., Choi, J.Y., Shin, D., and Ko, K.S. 2011. Correlation between overexpression and amino acid substitution of the PmrAB locus and colistin resistance in Acinetobacter baumannii. Int. J. Antimicrob. Agents37, 525–530.
Patriquin, G.M., Banin, E., Gilmour, C., Tuchman, R., Greenberg, E.P., and Poole, K. 2008. Influence of quorum sensing and iron on twitching motility and biofilm formation in Pseudomonas aeruginosa. J. Bacteriol.190, 662–671.
Peleg, A.Y., Seifert, H., and Paterson, D.L. 2008. Acinetobacter baumannii: emergence of a successful pathogen. Clin. Microbiol. Rev.21, 538–582.
Perez, F., Endimiani, A., Ray, A.J., Decker, B.K., Wallace, C.J., Hujer, K.M., Ecker, D.J., Adams, M.D., Toltzis, P., Dul, M.J., andet al. 2010. Carbapenem-resistant Acinetobacter baumannii and Klebsiella pneumoniae across a hospital system: impact of post-acute care facilities on dissemination. J. Antimicrob. Che mother.65, 1807–1818.
Petersen, K., Riddle, M.S., Danko, J.R., Blazes, D.L., Hayden, R., Tasker, S.A., and Dunne, J.R. 2007. Trauma-related infections in battlefield casualties from Iraq. Ann. Surg.245, 803–811.
Piskin, N., Celebi, G., Kulah, C., Mengeloglu, Z., and Yumusak, M. 2011. Activity of a dry mist-generated hydrogen peroxide disinfection system against methicillin-resistant Staphylococcus aureus and Acinetobacter baumannii. Am. J. Infect. Control.37, 525–530.
Prates, C.G., Martins, A.F., Superti, S.V., Lopes, F.S., Ramos, F., Cantarelli, V.V., and Zavascki, A.P. 2011. Risk factors for 30-day mortality in patients with carbapenem-resistant Acinetobacter baumannii during an outbreak in an intensive care unit. Epidemiol. Infect.139, 411–418.
Rashid, M.H. and Kornberg, A. 2000. Inorganic polyphosphate is needed for swimming, swarming, and twitching motilities of Pseudomonas aeruginosa. Proc. Natl. Acad. Sci. USA97, 4885–4890.
Ray, A., Perez, F., Beltramini, A.M., Jakubowycz, M., Dimick, P., Jacobs, M.R., Roman, K., Bonomo, R.A., and Salata, R.A. 2010. Use of vaporized hydrogen peroxide decontamination during an outbreak of multidrug-resistant Acinetobacter baumannii infection at a long-term acute care hospital. Infect. Control Hosp. Epidemiol.31, 1236–1241.
Russo, T.A., Luke, N.R., Beanan, J.M., Olson, R., Sauberan, S.L., Macdonald, U., Schultz, L.W., Umland, T.C., and Campagnari, A.A. 2010. The K1 capsular polysaccharide of Acinetobacter baumannii 307-0294 is a major virulence factor. Infect. Immun.78, 3993–4000.
Russo, T.A., MacDonald, U., Beanan, J.M., Olson, R., MacDonald, I.J., Sauberan, S.L., Luke, N.R., Schultz, L.W., and Umland, T.C. 2009. Penicillin-binding protein 7/8 contributes to the survival of Acinetobacter baumannii in vitro and in vivo. J. Infect. Dis.199, 513–521.
Scott, P., Deye, G., Srinivasan, A., Murray, C., Moran, K., Hulten, E., Fishbain, J., Craft, D., Riddell, S., Lindler, L., andet al. 2007. An outbreak of multidrug-resistant Acinetobacter baumannii-calcoaceticus complex infection in the US military health care system associated with military operations in Iraq. Clin. Infect. Dis.44, 1577–1584.
Smith, M.G., Gianoulis, T.A., Pukatzki, S., Mekalanos, J.J., Ornston, L.N., Gerstein, M., and Snyder, M. 2007. New insights into Acinetobacter baumannii pathogenesis revealed by high-density pyrosequencing and transposon mutagenesis. Genes Dev.21, 601–614.
Stepanovic, S., Vukovic, D., Dakic, I., Savic, B., and Svabic-Vlahovic, M. 2000. A modified microtiter-plate test for quantification of staphylococcal biofilm formation. J. Microbiol. Methods40, 175–179.
Strom, M.S. and Lory, S. 1993. Structure-function and biogenesis of the type IV pili. Annu. Rev. Microbiol.47, 565–596.
Toguchi, A., Siano, M., Burkart, M., and Harshey, R.M. 2000. Genetics of swarming motility in Salmonella enterica serovar typhimurium: critical role for lipopolysaccharide. J. Bacteriol.182, 6308–6321.
Tomaras, A.P., Dorsey, C.W., Edelmann, R.E., and Actis, L.A. 2003. Attachment to and biofilm formation on abiotic surfaces by Acinetobacter baumannii: involvement of a novel chaperone-usher pili assembly system. Microbiology149, 3473–3484.
Tong, M.J. 1972. Septic complications of war wounds. JAMA219, 1044–1047.
Wakimoto, N., Nishi, J., Sheikh, J., Nataro, J.P., Sarantuya, J., Iwashita, M., Manago, K., Tokuda, K., Yoshinaga, M., and Kawano, Y. 2004. Quantitative biofilm assay using a microtiter plate to screen for enteroaggregative Escherichia coli. Am. J. Trop. Med. Hyg.71, 687–690.
Yang, Z., Lux, R., Hu, W., Hu, C., and Shi, W. 2010. PilA localization affects extracellular polysaccharide production and fruiting body formation in Myxococcus xanthus. Mol. Microbiol.76, 1500–1513.
Yun, H.C., Branstetter, J.G., and Murray, C.K. 2008. Osteomyelitis in military personnel wounded in Iraq and Afghanistan. J. Trauma64, S163–168; discussion S168.
Zimbler, D.L., Penwell, W.F., Gaddy, J.A., Menke, S.M., Tomaras, A.P., Connerly, P.L., and Actis, L.A. 2009. Iron acquisition functions expressed by the human pathogen Acinetobacter baumannii. BioMetals22, 23–32.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplemental material for this article may be found at http://www.springer.com/content/120956
The findings and opinions expressed herein belong to the authors and do not necessarily reflect the official views of the WRAIR, the U.S. Army, or the Department of Defense.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
McQueary, C.N., Kirkup, B.C., Si, Y. et al. Extracellular stress and lipopolysaccharide modulate Acinetobacter baumannii surface-associated motility. J Microbiol. 50, 434–443 (2012). https://doi.org/10.1007/s12275-012-1555-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12275-012-1555-1