Abstract
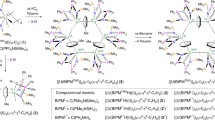
The rational reticular design of metal—organic frameworks (MOFs) from building units of known geometries is essential for enriching the diversity of MOF structures. Unexpected and intriguing structures, however, can also arise from subtle changes in the rigidity/length of organic linkers and/or synthetic conditions. Herein, we report three uranium-based MOF structures—i.e., NU-135X (X = 0, 1, 2)—synthesized from trigonal planar uranyl nodes and triptycene-based hexacarboxylate ligands with variable arm lengths. A new chiral 3,6-connected nuc net was observed in NU-1350, while the extended versions of the ligand led to 3-fold catenated MOFs (NU-1351 and NU-1352) with rare 3,6-connected cml-c3 nets. The differences in the topology of NU-1350 and NU-1351/NU-1352 could be attributed to the slight distortions of the shorter linker in the former from the ideal trigonal prism geometry to an octahedral geometry when coordinated to the trigonal planar uranyl nodes.

Similar content being viewed by others
References
Yaghi, O. M.; O’Keeffe, M.; Ockwig, N. W.; Chae, H. K.; Eddaoudi, M.; Kim, J. Reticular synthesis and the design of new materials. Nature2003, 423, 705–714.
Yaghi, O. M.; Kalmutzki, M. J.; Diercks, C. S. Introduction to Reticular Chemistry: Metal-Organic Frameworks and Covalent Organic Frameworks; Wiley-VCH Verlag GmbH & Co.: Weinheim, 2019.
Li, M.; Li, D.; O’Keeffe, M.; Yaghi, O. M. Topological analysis of metal-organic frameworks with polytopic linkers and/or multiple building units and the minimal transitivity principle. Chem. Rev.2014, 114, 1343–1370.
Chen, Z. J.; Jiang, H.; O’Keeffe, M.; Eddaoudi, M. Minimal edge-transitive nets for the design and construction of metal-organic frameworks. Faraday Discuss. 2017, 201, 127–143.
Chen, Z. J.; Weseliński, Ł. J.; Adil, K.; Belmabkhout, Y.; Shkurenko, A.; Jiang, H.; Bhatt, P. M.; Guillerm, V.; Dauzon, E.; Xue, D. X. et al. Applying the power of reticular chemistry to finding the missing alb-MOF platform based on the (6,12)-coordinated edge-transitive net. J. Am. Chem. Soc.2017, 139, 3265–3274.
Chen, Z. J.; Hanna, S. L.; Redfern, L. R.; Alezi, D.; Islamoglu, T.; Farha, O. K. Reticular chemistry in the rational synthesis of functional zirconium cluster-based MOFs. Coord. Chem. Rev.2019, 386, 32–49.
Jiang, H.; Jia, J. T.; Shkurenko, A.; Chen, Z. J.; Adil, K.; Belmabkhout, Y.; Weselinski, L. J.; Assen, A. H.; Xue, D. X.; O’Keeffe, M. et al. Enriching the reticular chemistry repertoire: Merged nets approach for the rational design of intricate mixed-linker metal-organic framework platforms. J. Am. Chem. Soc.2018, 140, 8858–8867.
Delgado-Friedrichs, O.; O’Keeffe, M.; Yaghi, O. M. Three-periodic nets and tilings: Edge-transitive binodal structures. Acta Cryst. A2006, 62, 350–355.
O’Keeffe, M.; Peskov, M. A.; Ramsden, S. J.; Yaghi, O. M. The reticular chemistry structure resource (RCSR) database of, and symbols for, crystal nets. Acc. Chem. Res.2008, 41, 1782–1789.
Zhang, X.; Huang, Z. Y.; Ferrandon, M.; Yang, D. L.; Robison, L.; Li, P.; Wang, T. C.; Delferro, M.; Farha, O. K. Catalytic chemoselective functionalization of methane in a metal-organic framework. Nat. Catal.2018, 1, 356–362.
Kim, H.; Yang, S.; Rao, S. R.; Narayanan, S.; Kapustin, E. A.; Furukawa, H.; Umans, A. S.; Yaghi, O. M.; Wang, E. N. Water harvesting from air with metal-organic frameworks powered by natural sunlight. Science2017, 356, 430–434.
Li, H.; Wang, K. C.; Sun, Y. J.; Lollar, C. T.; Li, J. L.; Zhou, H. C. Recent advances in gas storage and separation using metal-organic frameworks. Mater. Today2018, 21, 108–121.
Cui, X. L.; Chen, K. J.; Xing, H. B.; Yang, Q. W.; Krishna, R.; Bao, Z. B.; Wu, H.; Zhou, W.; Dong, X. L.; Han, Y. et al. Pore chemistry and size control in hybrid porous materials for acetylene capture from ethylene. Science2016, 353, 141–144.
Adil, K.; Belmabkhout, Y.; Pillai, R. S.; Cadiau, A.; Bhatt, P. M.; Assen, A. H.; Maurin, G.; Eddaoudi, M. Gas/vapour separation using ultra-microporous metal-organic frameworks: Insights into the structure/separation relationship. Chem. Soc. Rev.2017, 46, 3402–3430.
Chae, H. K.; Siberio-Pérez, D. Y.; Kim, J.; Go, Y.; Eddaoudi, M.; Matzger, A. J.; O’Keeffe, M.; Yaghi, O. M.; Design, M.; Group, D. A route to high surface area, porosity and inclusion of large molecules in crystals. Nature2004, 427, 523–527.
Furukawa, H.; Ko, N.; Go, Y. B.; Aratani, N.; Choi, S. B.; Choi, E.; Yazaydin, A. Ö.; Snurr, R. Q.; O’Keeffe, M.; Kim, J. et al. Ultrahigh porosity in metal-organic frameworks. Science2010, 329, 424–428.
Wang, Y. L.; Liu, Z. Y.; Li, Y. X.; Bai, Z. L.; Liu, W.; Wang, Y. X.; Xu, X. M.; Xiao, C. Q.; Sheng, D. P.; Diwu, J. et al. Umbellate distortions of the uranyl coordination environment result in a stable and porous polycatenated framework that can effectively remove cesium from aqueous solutions. J. Am. Chem. Soc.2015, 137, 6144–6147.
Gilson, S. E.; Li, P.; Szymanowski, J. E. S.; White, J.; Ray, D.; Gagliardi, L.; Farha, O. K.; Burns, P. C. In situ formation of unprecedented neptunium-oxide wheel clusters stabilized in a metal-organic framework. J. Am. Chem. Soc.2019, 141, 11842–11846.
Li, P.; Goswami, S.; Otake, K. I.; Wang, X. J.; Chen, Z. J.; Hanna, S. L.; Farha, O. K. Stabilization of an unprecedented hexanuclear secondary building unit in a thorium-based metal-organic framework. Inorg. Chem.2019, 58, 3586–3590.
Dolgopolova, E. A.; Rice, A. M.; Shustova, N. B. Actinide-based MOFs: A middle ground in solution and solid-state structural motifs. Chem. Commun.2018, 54, 6472–6483.
Hu, K. Q.; Huang, Z. W.; Zhang, Z. H.; Mei, L.; Qian, B. B.; Yu, J. P.; Chai, Z. F.; Shi, W. Q. Actinide-based porphyrinic MOF as a dehydrogenation catalyst. Chem.—Eur. J.2018, 24, 16766–16769.
Hanna, S. L.; Zhang, X.; Otake, K. I.; Drout, R. J.; Li, P.; Islamoglu, T.; Farha, O. K. Guest-dependent single-crystal-to-single-crystal phase transitions in a two-dimensional uranyl-based metal-organic framework. Cryst. Growth Des.2019, 19, 506–512.
Wang, Y. L.; Liu, W.; Bai, Z. L.; Zheng, T.; Silver, M. A.; Li, Y. X.; Wang, Y. X.; Wang, X.; Diwu, J.; Chai, Z. F. et al. Employing an unsaturated Th4+ site in a porous thorium-organic framework for Kr/Xe uptake and separation. Angew. Chem., Int. Ed.2018, 57, 5783–5787.
Li, Y. X.; Yang, Z. X.; Wang, Y. L.; Bai, Z. L.; Zheng, T.; Dai, X.; Liu, S. T.; Gui, D. X.; Liu, W.; Chen, M. et al. A mesoporous cationic thorium-organic framework that rapidly traps anionic persistent organic pollutants. Nat. Commun.2017, 8, 1354.
Wang, Y. X.; Yin, X. M.; Liu, W.; Xie, J.; Chen, J. F.; Silver, M. A.; Sheng, D. P.; Chen, L. H.; Diwu, J.; Liu, N. et al. Emergence of uranium as a distinct metal center for building intrinsic X-ray scintillators. Angew. Chem., Int. Ed.2018, 57, 7883–7887.
Xie, J.; Wang, Y. X.; Liu, W.; Yin, X. M.; Chen, L. H.; Zou, Y. M.; Diwu, J.; Chai, Z. F.; Albrecht-Schmitt, T. E.; Liu, G. K. et al. Highly sensitive detection of ionizing radiations by a photoluminescent uranyl organic framework. Angew. Chem., Int. Ed.2017, 56, 7500–7504.
Li, P.; Vermeulen, N. A.; Malliakas, C. D.; Gómez-Gualdrón, D. A.; Howarth, A. J.; Mehdi, B. L.; Dohnalkova, A.; Browning, N. D.; O’Keeffe, M.; Farha, O. K. Bottom-up construction of a superstructure in a porous uranium-organic crystal. Science2017, 356, 624–627.
Chen, Z. J.; Li, P. H.; Zhang, X.; Li, P.; Wasson, M. C.; Islamoglu, T.; Stoddart, J. F.; Farha, O. K. Reticular access to highly porous acs-MOFs with rigid trigonal prismatic linkers for water sorption. J. Am. Chem. Soc.2019, 141, 2900–2905.
Chen, Z. J.; Li, P. H.; Wang, X. J.; Otake, K. I.; Zhang, X.; Robison, L.; Atilgan, A.; Islamoglu, T.; Hall, M. G.; Peterson, G. W. et al. Ligand-directed reticular synthesis of catalytically active missing zirconium-based metal-organic frameworks. J. Am. Chem. Soc.2019, 141, 12229–12235.
Xue, D. X.; Cairns, A. J.; Belmabkhout, Y.; Wojtas, L.; Liu, Y. L.; Alkordi, M. H.; Eddaoudi, M. Tunable rare-earth fcu-MOFs: A platform for systematic enhancement of CO2 adsorption energetics and uptake. J. Am. Chem. Soc.2013, 135, 7660–7667.
Blatov, V. A.; Shevchenko, A. P.; Proserpio, D. M. Applied topological analysis of crystal structures with the program package ToposPro. Cryst. Growth Des.2014, 14, 3576–3586.
Li, P. H.; Li, P.; Ryder, M. R.; Liu, Z. C.; Stern, C. L.; Farha, O. K.; Stoddart, J. F. Interpenetration isomerism in triptycene-based hydrogen-bonded organic frameworks. Angew. Chem., Int. Ed.2019, 58, 1664–1669.
Chae, H. K.; Eddaoudi, M.; Kim, J.; Hauck, S. I.; Hartwig, J. F.; O’Keeffe, M.; Yaghi, O. M. Tertiary building units: Synthesis, structure, and porosity of a metal-organic dendrimer framework (MODF-1). J. Am. Chem. Soc.2001, 123, 11482–11483.
Jia, J. T.; Sun, F. X.; Fang, Q. R.; Liang, X. Q.; Cai, K.; Bian, Z.; Zhao, H. J.; Gao, L. X.; Zhu, G. S. A novel low density metal-organic framework with pcu topology by dendritic ligand. Chem. Commun.2011, 47, 9167–9169.
Delgado Friedrichs, O.; O’Keeffe, M.; Yaghi, O. M. Three-periodic nets and tilings: regular and quasiregular nets. Acta Cryst. A2003, 59, 22–27.
Liu, C. M.; Zuo, J. L.; Zhang, D. Q.; Zhu, D. B. Carboxylic acid-dependent assembly of neodymium-organic frameworks with attractive topologies and second-order nonlinear optical and/or magnetic properties. CrystEngComm2008, 10, 1674–1680.
Li, P. H.; Chen, Z. J.; Ryder, M. R.; Stern, C. L.; Guo, Q. H.; Wang, X. J.; Farha, O. K.; Stoddart, J. F. Assembly of a porous supramolecular polyknot from rigid trigonal prismatic building blocks. J. Am. Chem. Soc.2019, 141, 12998–13002.
Yuan, S.; Lu, W. G.; Chen, Y. P.; Zhang, Q.; Liu, T. F.; Feng, D. W.; Wang, X.; Qin, J. S.; Zhou, H. C. Sequential linker installation: Precise placement of functional groups in multivariate metal-organic frameworks. J. Am. Chem. Soc.2015, 137, 3177–3180.
Zhang, Y. Y.; Zhang, X.; Lyu, J. F.; Otake, K. I.; Wang, X. J.; Redfern, L. R.; Malliakas, C. D.; Li, Z. Y.; Islamoglu, T.; Wang, B. et al. A flexible metal-organic framework with 4-connected Zr6 nodes. J. Am. Chem. Soc.2018, 140, 11179–11183.
Guillerm, V.; Grancha, T.; Imaz, I.; Juanhuix, J.; Maspoch, D. Zigzag ligands for transversal design in reticular chemistry: Unveiling new structural opportunities for metal-organic frameworks. J. Am. Chem. Soc.2018, 140, 10153–10157.
Chandrasekhar, P.; Savitha, G.; Moorthy, J. N. Robust MOFs of “tsg” topology based on trigonal prismatic organic and metal cluster SBUs: Single crystal to single crystal postsynthetic metal exchange and selective CO2 capture. Chem.—Eur. J.2017, 23, 7297–7305.
Acknowledgements
O. K. F. acknowledges the support from the U.S. Department of Energy, National Nuclear Security Administration, under Award Number DE-NA0003763. This work made use of the EPIC facility of Northwestern University’s NUANCE Center, which has received support from the Soft and Hybrid Nanotechnology Experimental (SHyNE) Resource (NSF NNCI-1542205); the MRSEC program (NSF DMR-1720139) at the Materials Research Center; the International Institute for Nanotechnology (IIN); the Keck Foundation; and the State of Illinois, through the IIN. This work made use of the IMSERC at Northwestern University, which has received support from the NSF (CHE-1048773 and DMR0521267); Soft and Hybrid Nanotechnology Experimental (SHyNE) Resource (NSF NNCI-1542205); the State of Illinois and International Institute for Nanotechnology (IIN). P. L. and J. F. S. acknowledge the Joint Center of Excellence in Integrated Nano-Systems (JCIN) at King Abdulaziz City for Science and Technology (KACST) and Northwestern University (NU).
Author information
Authors and Affiliations
Corresponding authors
Electronic Supplementary Material
Rights and permissions
About this article
Cite this article
Chen, Z., Li, P., Zhang, X. et al. Reticular exploration of uranium-based metal—organic frameworks with hexacarboxylate building units. Nano Res. 14, 376–380 (2021). https://doi.org/10.1007/s12274-020-2690-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12274-020-2690-3