Abstract
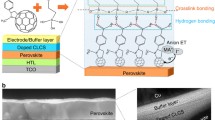
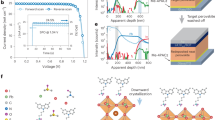
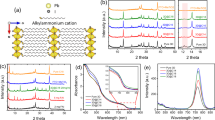
In spite of a continuous increase in their power conversion efficiency (PCE) and an economically viable fabrication process, organic–inorganic perovskite solar cells (PSCs) pose a significant problem when used in practical applications: They show fast degradation of the PCE when exposed to very humid environments. In this study, the stability of PSCs under very humid conditions is greatly enhanced by coating the surface of the PSC devices with a multi-layer film consisting of ultrahydrophobic and relatively hydrophilic layers. A hydrophobic composite of poly(methyl methacrylate) (PMMA), polyurethane (PU), and SiO2 nanoparticles successfully retards the water molecules from very humid surroundings. Also, the hydrophilic layer with moderately PMMA captures the residual moisture within the perovskite layer; subsequently, the perovskite layer recovers. This dual function of the coating film keeps the PCE of PSCs at 17.3% for 180 min when exposed to over 95% humidity.

Similar content being viewed by others
References
Son, D.-Y.; Lee, J.-W.; Choi, Y. J.; Jang, I.-H.; Lee, S.; Yoo, P. J.; Shin, H.; Ahn, N.; Choi, M.; Kim, D. et al. Selfformed grain boundary healing layer for highly efficient CH3NH3PbI3 perovskite solar cells. Nat. Energy 2016, 1, 16081.
Park, N.-G. Perovskite solar cells: An emerging photovoltaic technology. Mater. Today 2015, 18, 65–72.
Grätzel, M. The light and shade of perovskite solar cells. Nat. Mater. 2014, 13, 838–842.
Green, M. A.; Ho-Baillie, A.; Snaith, H. J. The emergence of perovskite solar cells. Nat. Photonics 2014, 8, 506–514.
Jung, H. S.; Park, N.-G. Perovskite solar cells: From materials to devices. Small 2015, 11, 10–25.
Christians, J. A.; Herrera, P. A. M.; Kamat, P. V. Transformation of the excited state and photovoltaic efficiency of CH3NH3PbI3 perovskite upon controlled exposure to humidified air. J. Am. Chem. Soc. 2015, 137, 1530–1538.
Niu, G. D.; Li, W. Z.; Meng, F. Q.; Wang, L. D.; Dong, H. P.; Qiu, Y. Study on the stability of CH3NH3PbI3 films and the effect of post-modification by aluminum oxide in all-solid-state hybrid solar cells. J. Mater. Chem. A 2014, 2, 705–710.
Yuan, Y. B.; Wang, Q.; Shao, Y. C.; Lu, H. D.; Li, T.; Gruverman, A.; Huang, J. S. Electric-field-driven reversible conversion between methylammonium lead triiodide perovskites and lead iodide at elevated temperatures. Adv. Energy Mater. 2016, 6, 1501803.
Yuan, Y. B.; Huang, J. S. Ion migration in organometal trihalide perovskite and its impact on photovoltaic efficiency and stability. Acc. Chem. Res. 2016, 49, 286–293.
Wei, D.; Wang, T. Y.; Ji, J.; Li, M. C.; Cui, P.; Li, Y. Y.; Li, G. Y.; Mbenguea, J. M.; Song, D. D. Photo-induced degradation of lead halide perovskite solar cells caused by the hole transport layer/metal electrode interface. J. Mater. Chem. A 2016, 4, 1991–1998.
D’Innocenzo, V.; Grancini, G.; Alcocer, M. J. P.; Kandada, A. R. S.; Stranks, S. D.; Lee, M. M.; Lanzani, G.; Snaith, H. J.; Petrozza, A. Excitons versus free charges in organo-lead tri-halide perovskites. Nat. Commun. 2014, 5, 3586.
Shao, Y. C.; Xiao, Z. G.; Bi, C.; Yuan, Y. B.; Huang, J. S. Origin and elimination of photocurrent hysteresis by fullerene passivation in CH3NH3PbI3 planar heterojunction solar cells. Nat. Commun. 2014, 5, 5784.
Unger, E. L.; Hoke, E. T.; Bailie, C. D.; Nguyen, W. H.; Bowring, A. R.; Heumü ller, T.; Christoforod, M. G.; McGehee, M. D. Hysteresis and transient behavior in current–voltage measurements of hybrid-perovskite absorber solar cells. Energy Environ. Sci. 2014, 7, 3690–3698.
Chen, H.-W.; Sakai, N.; Ikegami, M.; Miyasaka, T. Emergence of hysteresis and transient ferroelectric response in organolead halide perovskite solar cells. J. Phys. Chem. Lett. 2015, 6, 164–169.
Tress, W.; Marinova, N.; Moehl, T.; Zakeeruddin, S. M.; Nazeeruddina, M. K.; Grä tzel, M. Understanding the ratedependent J–V hysteresis, slow time component, and aging in CH3NH3PbI3 perovskite solar cells: The role of a compensated electric field. Energy Environ. Sci. 2015, 8, 995–1004.
Hao, F.; Stoumpos, C. C.; Cao, D. H.; Chang, R. P. H.; Kanatzidis, M. G. Lead-free solid-state organic–inorganic halide perovskite solar cells. Nat. Photonics 2014, 8, 489–494.
Noel, N. K.; Stranks, S. D.; Abate, A.; Wehrenfennig, C.; Guarnera, S.; Haghighirad, A.-A.; Sadhanala, A.; Eperon, G. E.; Pathak, S. K.; Johnston, M. B. et al. Lead-free organic–inorganic tin halide perovskites for photovoltaic applications. Energy Environ. Sci. 2014, 7, 3061–3068.
Stoumpos, C. C.; Malliakas, C. D.; Kanatzidis, M. G. Semiconducting tin and lead iodide perovskites with organic cations: Phase transitions, high mobilities, and near-infrared photoluminescent properties. Inorg. Chem. 2013, 52, 9019–9038.
Habisreutinger, S. N.; Leijtens, T.; Eperon, G. E.; Stranks, S. D.; Nicholas, R. J.; Snaith, H. J. Carbon nanotube/polymer composites as a highly stable hole collection layer in perovskite solar cells. Nano Lett. 2014, 14, 5561–5568.
Hwang, I.; Jeong, I.; Lee, J.; Ko, M. J.; Yong, K. Enhancing stability of perovskite solar cells to moisture by the facile hydrophobic passivation. ACS Appl. Mater. Interfaces 2015, 7, 17330–17336.
Li, X.; Dar, M. I.; Yi, C. Y.; Luo, J. S.; Tschumi, M.; Zakeeruddin, S. M.; Nazeeruddin, M. K.; Han, H. W.; Grä tzel, M. Improved performance and stability of perovskite solar cells by crystal crosslinking with alkylphosphonic acid ?-ammonium chlorides. Nat. Chem. 2015, 7, 703–711.
You, J. B.; Meng, L.; Song, T.-B.; Guo, T.-F.; Yang, Y.; Chang, W.-H.; Hong, Z. R.; Chen, H. J.; Zhou, H. P.; Chen, Q. et al. Improved air stability of perovskite solar cells via solution-processed metal oxide transport layers. Nat. Nanotechnol. 2016, 11, 75–81.
Han, Y.; Meyer, S.; Dkhissi, Y.; Weber, K.; Pringle, J. M.; Bach, U.; Spiccia, L.; Cheng, Y.-B. Degradation observations of encapsulated planar CH3NH3PbI3 perovskite solar cells at high temperatures and humidity. J. Mater. Chem. A 2015, 3, 8139–8147.
Leijtens, T.; Eperon, G. E.; Pathak, S.; Abate, A.; Lee, M. M.; Snaith, H. J. Overcoming ultraviolet light instability of sensitized TiO2 with meso-superstructured organometal tri-halide perovskite solar cells. Nat. Commun. 2013, 4, 2885.
Liu, Z. Y.; Sun, B.; Shi, T. L.; Tang, Z. R.; Liao, G. L. Enhanced photovoltaic performance and stability of carbon counter electrode based perovskite solar cells encapsulated by PDMS. J. Mater. Chem. A 2016, 4, 10700–10709.
Wei, Z. H.; Zheng, X. L.; Chen, H. N.; Long, X.; Wang, Z. L.; Yang, S. H. A multifunctional C + epoxy/Ag-paint cathode enables efficient and stable operation of perovskite solar cells in watery environments. J. Mater. Chem. A 2015, 3, 16430–16434.
Kim, J. H.; Liang, P.-W.; Williams, S. T.; Cho, N.; Chueh, C.-C.; Glaz, M. S.; Ginger, D. S.; Jen, A. K.-Y. Highperformance and environmentally stable planar heterojunction perovskite solar cells based on a solution-processed copperdoped nickel oxide hole-transporting layer. Adv. Mater. 2015, 27, 695–701.
Guarnera, S.; Abate, A.; Zhang, W.; Foster, J. M.; Richardson, G.; Petrozza, A.; Snaith, H. J. Improving the long-term stability of perovskite solar cells with a porous Al2O3 buffer layer. J. Phys. Chem. Lett. 2015, 6, 432–437.
Zheng, L. L.; Chung, Y.-H.; Ma, Y. Z.; Zhang, L. P.; Xiao, L. X.; Chen, Z. J.; Wang, S. F.; Qu, B.; Gong, Q. H. A hydrophobic hole transporting oligothiophene for planar perovskite solar cells with improved stability. Chem. Commun. 2014, 50, 11196–11199.
Liu, J.; Wu, Y. Z.; Qin, C. J.; Yang, X. D.; Yasuda, T.; Islam, A.; Zhang, K.; Peng, W. Q.; Chen, W.; Han, L. Y. A dopant-free hole-transporting material for efficient and stable perovskite solar cells. Energy Environ. Sci. 2014, 7, 2963–2967.
Li, R.; Xiang, X.; Tong, X.; Zou, J.; Li, Q. Wearable doubletwisted fibrous perovskite solar cell. Adv. Mater. 2015, 27, 3831–3835.
Kim, B. J.; Kim, D. H.; Kwon, S. L.; Park, S. Y.; Li, Z.; Zhu, K.; Jung, H. S. Selective dissolution of halide perovskites as a step towards recycling solar cells. Nat. Commum. 2016, 7, 11735.
Moghbelli, E.; Banyay, R.; Sue, H.-J. Effect of moisture exposure on scratch resistance of PMMA. Tribol. Int. 2014, 69, 46–51.
Tan, K. T.; Vogt, B. D.; White, C. C.; Steffens, K. L.; Goldman, J.; Satija, S. K.; Clerici, C.; Hunston, D. L. On the origins of sudden adhesion loss at a critical relative humidity: Examination of bulk and interfacial contributions. Langmuir 2008, 24, 9189–9193.
Flory, P. J.; Rehner, J. Statistical mechanics of crosslinked polymer networks I. Rubberlike elasticity. J. Chem. Phys. 1943, 11, 512–520.
Morsch, S.; Lyon, S.; Greensmith, P.; Smith, S. D.; Gibbon, S. R. Mapping water uptake in organic coatings using AFM-IR. Faraday Discuss. 2015, 180, 527–542.
Athawale, V. D.; Kolekar, S. L.; Raut, S. S. Recent developments in polyurethanes and poly(acrylates) interpenetrating polymer networks. J. Macromol. Sci. 2003, 43, 1–26.
Gupta, N.; Srivastava, A. K. Interpenetrating polymer networks: A review on synthesis and properties. Polym. Int. 1994, 35, 109–118.
Athawale, V.; Kolekar, S. Interpenetrating polymer networks based on polyol modified castor oil polyurethane and polymethyl methacrylate. Eur. Polym. J. 1998, 34, 1447–1451.
Kumar, H.; Kumar, A. A.; Siddaramaiah. Physico-mechanical, thermal and morphological behaviour of polyurethane/ poly(methyl methacrylate) semi-interpenetrating polymer networks. Polym. Degrad. Stabil. 2006, 91, 1097–1104.
Akay, M.; Rollins, S. N. Polyurethane-poly(methyl methacrylate) interpenetrating polymer networks. Polymer 1993, 34, 1865–1873.
Zhu, A. P.; Shi, Z. H.; Cai, A. Y.; Zhao, F.; Liao, T. Q. Synthesis of core–shell PMMA–SiO2 nanoparticles with suspension–dispersion–polymerization in an aqueous system and its effect on mechanical properties of PVC composite. Polym. Test. 2008, 27, 540–547.
N’Diaye, M.; Pascaretti-Grizon, F.; Massin, P.; Baslé, M. F.; Chappard, D. Water absorption of poly(methyl methacrylate) measured by vertical interference microscopy. Langmuir 2012, 28, 11609–11614.
Yang, M. J.; Di, Z. F.; Lee, J.-K. Facile control of surface wettability in TiO2/poly(methyl methacrylate) composite films. J. Colloid Interface Sci. 2012, 368, 603–607.
Acknowledgements
This work was supported from the Global Frontier R&D Program on Center for Multiscale Energy System, Republic of Korea (No. 2012M3A6A7054855) and National Science Foundation (Nos. CMMI-1333182 and EPMD-1408025).
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
12274_2017_1603_MOESM1_ESM.pdf
Dual function of a high-contrast hydrophobic–hydrophilic coating for enhanced stability of perovskite solar cells in extremely humid environments.
Rights and permissions
About this article
Cite this article
Yoo, J.S., Han, G.S., Lee, S. et al. Dual function of a high-contrast hydrophobic–hydrophilic coating for enhanced stability of perovskite solar cells in extremely humid environments. Nano Res. 10, 3885–3895 (2017). https://doi.org/10.1007/s12274-017-1603-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12274-017-1603-6