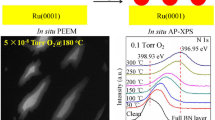
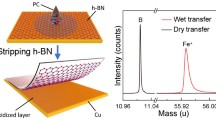
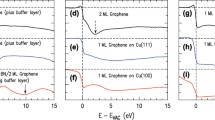
Abstract
Hexagonal boron nitride (h-BN) is believed to offer better passivation to metallic surfaces than graphene owing to its insulating nature, which facilitates blocking the flow of electrons, thereby preventing the occurrence of galvanic reactions. Nevertheless, this may not be the case when an h-BN-protected material is exposed to aqueous environments. In this work, we analyzed the stability of mono and multilayer h-BN stacks exposed to H2O2 and atmospheric conditions. Our experiments revealed that monolayer h-BN is as inefficient as graphene as a protective coating when exposed to H2O2. Multilayer h-BN offered a good degree of protection. Monolayer h-BN was found to be ineffective in an air atmosphere as well. Even a 10–15 layers-thick h-BN stack could not completely protect the surface of the metal under consideration. By combining Auger electron spectroscopy and secondary ion mass spectrometry techniques, we observed that oxygen could diffuse through the grain boundaries of the h-BN stack to reach the metallic substrate. Fortunately, because of the diffusive nature of the process, the oxidized area did not increase with time once a saturated state was reached. This makes multilayer (not monolayer) h-BN a suitable long-term oxidation barrier. Oxygen infiltration could not be observed by X-ray photoelectron spectroscopy. This technique cannot assess the chemical composition of the deeper layers of a material. Hence, the previous reports, which relied on XPS to analyze the passivating properties of h-BN and graphene, may have ignored some important subsurface phenomena. The results obtained in this study provide new insights into the passivating properties of mono and multilayer h-BN in aqueous media and the degradation kinetics of h-BN-coated metals exposed to an air environment.

Similar content being viewed by others
References
Böhm, S. Graphene against corrosion. Nat. Nanotechnol. 2014, 9, 741–742.
Segarra, M.; Miralles, L.; Díaz, J.; Xuriguera, H.; Chimenos, J. M.; Espiell, F.; Piñol, S. Copper and CuNi alloys substrates for HTS coated conductor applications protected from oxidation. Mater. Sci. Forum 2003, 426–432, 3511–3516.
Merkula, D. M.; Novikov, P. D.; Ivanenkov, V. N.; Sapozhnikov, V. V.; Lyakhin, Y. I. Utilization of EDN varnish for protection of metal sea-water sampling bottles against corrosion. Oceanology 1974, 14, 299–300.
Mittal, V. K.; Bersa, S.; Saravanan, T.; Sumathi, S.; Krishnan, R.; Rangarajan, S.; Velmurugan, S.; Narasimhan, S. V. Formation and characterization of bi-layer oxide coating on carbon-steel for improving corrosion resistance. Thin Solid Films 2009, 517, 1672–1676.
Redondo, M. I.; Breslin, C. B. Polypyrrole electrodeposited on copper from an aqueous phosphate solution: Corrosion protection properties. Corros. Sci. 2007, 49, 1765–1776.
Cho, H. B.; Tokoi, Y.; Tanaka, S.; Suematsu, H.; Suzuki, T.; Jiang, W.; Niihara, K.; Nakayama, T. Modification of BN nanosheets and their thermal conducting properties in nanocomposite film with polysiloxane according to the orientation of BN. Compos. Sci. Technol. 2011, 71, 1046–1052.
Kim, S. J.; Choi, K.; Lee, B.; Kim, Y.; Hong, B. H. Materials for flexible, stretchable electronics: Graphene and 2D materials. Annu. Rev. Mater. Res. 2015, 45, 63–84.
Zhi, C. Y.; Bando, Y.; Tang, C. C.; Kuwahara, H.; Golberg, D. Large-scale fabrication of boron nitride nanosheets and their utilization in polymeric composites with improved thermal and mechanical properties. Adv. Mater. 2009, 21, 2889–2893.
Balandin, A. A.; Ghosh, S.; Bao, W. Z.; Calizo, I.; Teweldebrhan, D.; Miao, F.; Lau, C. N. Superior thermal conductivity of single-layer graphene. Nano Lett. 2008, 8, 902–907.
Sattler, K. D. Handbook of Nanophysics: Functional Nanomaterials; CRC Press: Boca Raton, 2010.
Giovannetti, G.; Khomyakov, P. A.; Brocks, G.; Karpan, V. M.; van den Brink, J.; Kelly, P. J. Doping graphene with metal contacts. Phys. Rev. Lett. 2008, 101, 026803.
Sachdev, H. Influence of impurities on the morphology and Raman spectra of cubic boron nitride. Diamond Relat. Mater. 2003, 12, 1275–1286.
Novoselov, K. S.; Fal’ko, V. I.; Colombo, L.; Gellert, P. R.; Schwab, M. G.; Kim, K. A roadmap for graphene. Nature 2012, 490, 192–200.
Eichler, J.; Lesniak, C. Boron nitride (BN) and BN composites for high-temperature applications. J. Eur. Ceram. Soc. 2008, 28, 1105–1109.
Jan, R.; May, P.; Bell, A. P.; Habib, A.; Khan, U.; Coleman, J. N. Enhancing the mechanical properties of BN nanosheet–polymer composites by uniaxial drawing. Nanoscale 2014, 6, 4889–4895.
Lanza, M.; Wang, Y.; Gao, T.; Bayerl, A.; Porti, M.; Nafria, M.; Zhou, Y. B.; Jing, G. Y.; Zhang, Y. F.; Liu, Z. F. et al. Electrical and mechanical performance of graphene sheets exposed to oxidative environments. Nano Res. 2013, 6, 485–495.
Shi, Y. Y.; Ji, Y. F.; Hui, F.; Wu, H. H.; Lanza, M. Ageing mechanisms and reliability of graphene-based electrodes. Nano Res. 2014, 7, 1820–1831.
Chen, S. S.; Brown, L.; Levendorf, M.; Cai, W. W.; Ju, S. Y.; Edgeworth, J.; Li, X. S.; Magnuson, C. W.; Velamakanni, A.; Piner, R. D. et al. Oxidation resistance of graphene-coated Cu and Cu/Ni alloy. ACS Nano 2011, 5, 1321–1327.
Nilsson, L.; Andersen, M.; Balog, R.; Læegsgaard, E.; Hofmann, P.; Besenbacher, F.; Hammer, B.; Stensgaard, I.; Hornekær, L. Graphene coatings: Probing the limits of the one atom thick protection layer. ACS Nano 2012, 6, 10258–10266.
Duong, D. L.; Han, G. H.; Lee, S. M.; Gunes, F.; Kim, E. S.; Kim, S. T.; Kim, H.; Ta, Q. H.; So, K. P.; Yoon S. J. et al. Probing graphene grain boundaries with optical microscopy. Nature 2012, 490, 235–239.
Kang, D.; Kwon, J. Y.; Cho, H.; Sim, J. H.; Hwang, H. S.; Kim, C. S.; Kim, Y. J.; Ruoff, R. S.; Shin, H. S. Oxidation resistance of iron and copper foils coated with reduced graphene oxide multilayers. ACS Nano 2012, 6, 7763–7769.
Nemes-Incze, P.; Yoo, K. J.; Tapasztό, L.; Dobrik, G.; Lábár, J.; Horváth, Z. E.; Hwang, C. Y.; Biró, L. P. Revealing the grain structure of graphene grown by chemical vapor deposition. App. Phys. Lett. 2011, 99, 023104.
Raman, R. K. S.; Banerjee, P. C.; Lobo, D. E.; Gullapallli, H.; Sumandasa, M.; Kumar, A.; Choudhary, L.; Tkacz, R.; Ajayan, P. M.; Majumder, M. Protecting copper from electrochemical degradation by graphene coating. Carbon 2012, 50, 4040–4045.
Won, M. S.; Penkov, O. V.; Kim, D. E. Durability and degradation mechanism of graphene coatings deposited on Cu substrates under dry contact sliding. Carbon 2013, 54, 472–481.
David, L.; Bhandavat, R.; Kulkarni, G.; Pahwa, S.; Zhong, Z.; Singh, G. Synthesis of graphene films by rapid heating and quenching at ambient pressures and their electrochemical characterization. ACS Appl. Mater. Interfaces 2013, 5, 546–552.
Prasai, D.; Tuberquia, J. C.; Harl, R. R.; Jennings, G. K.; Bolotin, K. I. Graphene: Corrosion-inhibiting coating. ACS Nano 2012, 6, 1102–1108.
Hui, F.; Shi, Y. Y.; Ji, Y. F.; Lanza, M.; Duan, H. L. Mechanical properties of locally oxidized graphene electrodes. Arch. Appl. Mech. 2015, 85, 339–345.
Hu, J. C.; Ji, Y. F.; Shi, Y. Y.; Hui, F.; Duan, H. L.; Lanza, M. A review on the use of graphene as a protective coating against corrosion. Ann. J. Materials Sci. Eng. 2014, 1, 16.
Yu, Q.; Jauregui, L. A.; Wu, W.; Colby, R.; Tian, J.; Su, Z.; Cao, H.; Liu, Z.; Pandey, D.; Wei, D.; Chung, T. F.; Peng, P.; Guisinger, N. P.; Stach, E. A.; Bao, J.; Pei, S. S.; Chen, Y. P. Control and characterization of individual grains and grain boundaries in graphene grown by chemical vapour deposition. Nat. Mater. 2011, 10, 443–449.
Schriver, M.; Regan, W.; Gannett, W. J.; Zaniewski, A. M.; Crommie, M. F.; Zettl, A. Graphene as a long-term metal oxidation barrier: Worse than nothing. ACS Nano 2013, 7, 5763–5768.
Zhou, F.; Li, Z. T.; Shenoy, G. J.; Li, L.; Liu, H. T. Enhanced room-temperature corrosion of copper in the presence of graphene. ACS Nano 2013, 7, 6939–6947.
Hui, F.; Pan, C. B.; Shi, Y. Y.; Ji, Y. F.; Grustan-Gutierrez, E.; Lanza, M. On the use of two dimensional hexagonal boron nitride as dielectric. Microelectron. Eng. 2016, 163, 119–133.
Ji, Y. F.; Pan, C. B.; Zhang, M. Y.; Long, S. B.; Lian, X. J.; Miao, F.; Hui, F.; Shi, Y. Y.; Larcher, L.; Wu, E. et al. Boron nitride as two dimensional dielectric: Reliability and dielectric breakdown. Appl. Phys. Lett. 2016, 108, 012905.
Li, X. M.; Yin, J.; Zhou, J. X.; Guo, W. L. Large area hexagonal boron nitride monolayer as efficient atomically thick insulating coating against friction and oxidation. Nanotechnology 2014, 25, 105701.
Yin, J.; Li, J. D.; Hang, Y.; Yu, J.; Tai, G. A.; Li, X. M.; Zhang, Z. H.; Guo, W. L. Boron nitride nanostructures: Fabrication, functionalization and applications. Small 2016, 12, 2942–2968.
Shen, L. T.; Zhao, Y. D.; Wang, Y.; Song, R. B.; Yao, Q.; Chen, S. S.; Chai, Y. A long-term corrosion barrier with an insulating boron nitride monolayer. J. Mater. Chem. A 2016, 4, 5044–5050.
Li, L. H.; Cervenka, J.; Watanabe, K.; Taniguchi, T.; Chen, Y. Strong oxidation resistance of atomically thin boron nitride nanosheets. ACS Nano 2014, 8, 1457–1462.
Liu, Z.; Gong, Y. J.; Zhou, W.; Ma, L. L.; Yu, J. J.; Idrobo, J. C.; Jung, J.; MacDonald, A. H.; Vajtai, R.; Lou, J. et al. Ultrathin high-temperature oxidation-resistant coatings of hexagonal boron nitride. Nat. Commun. 2013, 4, 2541.
Li, L. H.; Xing, T.; Chen, Y.; Jones, R. Boron nitride nanosheets for metal protection. Adv. Mater. Interfaces 2014, 1, 1300132.
Haasch, R. T. X-ray photoelectron spectroscopy (XPS) and auger electron spectroscopy (AES). In Practical Materials Characterization; Sardela, M., Ed.; Springer: New York, 2014; pp 93–132.
Hofmann, S. Auger- and X-Ray Photoelectron Spectroscopy in Materials Science; Springer: Berlin Heidelberg, 2013.
Kubicek, M.; Holzlechner, G.; Opitz, A. K.; Larisegger, S.; Hutter, H.; Fleig, J. A novel ToF-SIMS operation mode for sub 100 nm lateral resolution: Application and performance. Appl. Surf. Sci. 2014, 289, 407–416.
Kim, K. K.; Hsu, A.; Jia, X. T.; Kim, S. M.; Shi, Y. M.; Dresselhaus, M.; Palacios, T.; Kong, J. Synthesis and characterization of hexagonal boron nitride film as a dielectric layer for graphene devices. ACS Nano 2012, 6, 8583–8590.
Lu, G. Y.; Wu, T. R.; Yuan, Q. H.; Wang, H. S.; Wang, H. M.; Ding, F.; Xie, X. M.; Jiang, M. H. Synthesis of large single-crystal hexagonal boron nitride grains on Cu–Ni alloy. Nat. Commun. 2015, 6, 6160.
Kaemmer, S. B. Introduction to Bruker’s ScanAsyst and PeakForce Tapping AFM Technology [Online]. AFM application notes. AN133, DS090, Rev. A0. http://www.bruker.com/products/surface-and-dimensional-analysis/atomic-force-microscopes/afm-application-notes/an133-introduction-to-brukers-scanasyst-and-peakforce-tapping.html (accessed Aug 23, 2016).
Moelwyn-Hughes, E. A. The Kinetics of Reactions in Solution, 2nd ed.; Clarendon Press: Oxford, 1947.
Senese, F. Will Hydrogen Peroxide Blacken Copper? [Online]. General Chemistry. http://antoine.frostburg.edu/chem/senese/ 101/redox/faq/h2o2-cu (accessed Aug 17, 2015).
Xing, W. L.; Lalwani, G.; Rusakova, I.; Sitharaman, B. Degradation of graphene by hydrogen peroxide. Part. Part. Syst. Charact. 2014, 31, 745–750.
Huang, P. Y.; Ruiz-Vargas, C. S.; van der Zande, A. M.; Whitney, W. S.; Levendorf, M. P., Kevek, J. W.; Garg, S.; Alden, J. S.; Hustedt, C. J.; Zhu, Y. et al. Grains and grain boundaries in single-layer graphene atomic patchwork quilts. Nature 2011, 469, 389–392.
Li, Q. C.; Zou, X. L.; Liu, M. X.; Sun, J. Y.; Gao, Y. B.; Qi, Y.; Zhou, X. B.; Yakobson, B. I.; Zhang, Y. F.; Liu, Z. F. Grain boundary structures and electronic properties of hexagonal boron nitride on Cu(111). Nano Lett. 2015, 15, 5804–5810.
Szalkowski, F. J.; Somorjai, G. A. Auger electron spectroscopy investigations of the surface chemical composition of vanadium, the vanadium oxides, and oxidized vanadium: Chemical shift and peak intensity analysis. J. Chem. Phys. 1972, 56, 6097–6103.
Jing, X.; Panholzer, E.; Song, X. X.; Grustan-Gutierrez, E.; Hui, F.; Shi, Y. Y.; Benstetter, G.; Illarionov, Y.; Grasser, T.; Lanza, M. Fabrication of scalable and ultra low power photodetectors with high light/dark current ratios using polycrystalline monolayer MoS2 sheets. Nano Energy 2016, 30, 494–502.
Kidambi, P. R.; Blume, R.; Kling, J.; Wagner, J. B.; Baehtz, C.; Weatherup, R. S.; Schloegl, R.; Bayer, B. C.; Hofmann, S. In situ observations during chemical vapor deposition of hexagonal boron nitride on polycrystalline copper. Chem. Mater. 2014, 26, 6380–6392.
Acknowledgements
This work has been supported by the Young 1000 Global Talent Recruitment Program of the Ministry of Education of China, the National Natural Science Foundation of China (Nos. 61502326, 41550110223, and 11661131002), the Jiangsu Government (No. BK20150343), the Ministry of Finance of China (No. SX21400213) and the National Basic Research Program of China (No. 2015CB932700). The Collaborative Innovation Center of Suzhou Nano Science & Technology, the Jiangsu Key Laboratory for Carbon-Based Functional Materials & Devices and the Priority Academic Program Development of Jiangsu Higher Education Institutions are also acknowledged. Part of this work was carried out with the excellent support of the Nanofabrication and Nanocharacterization Core Facility of CEITEC under CEITEC Nano project (No. LM2015041). P. B. and M. K. acknowledge support of Ministry of Education, Youth and Sports of the Czech Republic under the project CEITEC 2020 (No. LQ1601).
Author information
Authors and Affiliations
Corresponding author
Additional information
These authors contributed to this work equally.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Jiang, L., Xiao, N., Wang, B. et al. High-resolution characterization of hexagonal boron nitride coatings exposed to aqueous and air oxidative environments. Nano Res. 10, 2046–2055 (2017). https://doi.org/10.1007/s12274-016-1393-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12274-016-1393-2