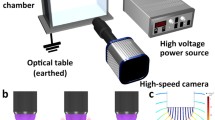
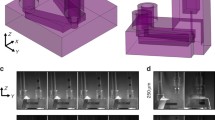
Abstract
The capability to design and modulate materials, shapes, heat transfer, and mass mixing during the process of developing chemical reactors has allowed researchers to explore millions of chemical reactions and assays. However, despite the advantages in engineering array-based microreactors or microfluidic systems, the wetting attachment between solutions of reagents/products and the glass or polymer substrates of containers leads to difficulties in collecting products effectively and preventing channel blockage. Herein we present a miniature droplet reactor which takes advantage of the anti-wetting and low-adhesive properties of nanoparticle-derived superhydrophobic pedestals, allowing aqueous droplets to be manipulated freely but also providing a confined environment for performing a series of aqueous phase chemical reactions on a small scale. Gas- or precipitate-forming reactions can also be performed inside this miniature reactor. Most importantly, reaction products in liquid, solid or gaseous states can be collected effectively, which allows the harvesting of valuable products formed in limited amounts. Such a miniature reactor built on superhydrophobic pedestals provides a new way of performing common chemical reactions and may open the door to the design of next-generation microreaction systems.

Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Crosland, M. Lavoisier: Chemist, biologist, economist—Poirier, JP. Nature 1997, 387, 250–251.
Chertok, L.; Stengers, I. Therapy and the ideal of chemistry. Nature 1987, 329, 768.
Lopez-Quintela, M. A.; Tojo, C.; Blanco, M. C.; Rio, L. G.; Leis, J. R. Microemulsion dynamics and reactions in microemulsions. Curr. Opin. Colloid. In. 2004, 9, 264–278.
Gross, G. A.; Wurziger, H.; Schlingloff, G.; Schober, A. Microreactor array assembly, designed for diversity oriented synthesis using a multiple core structure library on solid support. Qsar. Comb. Sci. 2006, 25, 1055–1062.
deMello, A. J. Control and detection of chemical reactions in microfluidic systems. Nature 2006, 442, 394–402.
Kobayashi, J.; Mori, Y.; Okamoto, K.; Akiyama, R.; Ueno, M.; Kitamori, T.; Kobayashi, S. A microfluidic device for conducting gas-liquid-solid hydrogenation reactions. Science 2004, 304, 1305–1308.
Haswell, S. J. Chemical technology—All together now. Nature 2006, 441, 705.
Song, H.; Chen, D. L.; Ismagilov, R. F. Reactions in droplets in microflulidic channels. Angew. Chem. Int. Edit. 2006, 45, 7336–7356.
Adams, J. B.; Hector, L. G.; Siegel, D. J.; Yu, H. L.; Zhong, J. Adhesion, lubrication, and wear on the atomic scale. Surf. Interface. Anal. 2001, 31, 619–626.
Cognard, J. Some recent progress in adhesion technology and science. C. R. Chim. 2006, 9, 13–24.
Marshall, S. J.; Bayne, S. C.; Baier, R.; Tomsia, A. P.; Marshall, G. W. A review of adhesion science. Dent. Mater. 2010, 26, E11–E16.
Blossey, R. Self-cleaning surfaces—virtual realities. Nat. Mater. 2003, 2, 301–306.
Chaudhury, M. K.; Whitesides, G. M. How to make water run uphill. Science 1992, 256, 1539–1541.
Gao, X. F.; Jiang, L. Water-repellent legs of water striders. Nature 2004, 432, 36.
Li, X. M.; Reinhoudt, D.; Crego-Calama, M. What do we need for a superhydrophobic surface? A review on the recent progress in the preparation of superhydrophobic surfaces. Chem. Soc. Rev. 2007, 36, 1350–1368.
Zheng, Y. M.; Bai, H.; Huang, Z. B.; Tian, X. L.; Nie, F. Q.; Zhao, Y.; Zhai, J.; Jiang, L. Directional water collection on wetted spider silk. Nature 2010, 463, 640–643.
Daniel, S.; Chaudhury, M. K.; Chen, J. C. Past drop movements resulting from the phase change on a gradient surface. Science 2001, 291, 633–636.
Yang, J. T.; Yang, Z. H.; Chen, C. Y.; Yao, D. J. Conversion of surface energy and manipulation of a single droplet across micropatterned surfaces. Langmuir 2008, 24, 9889–9897.
Sun, T. L.; Feng, L.; Gao, X. F.; Jiang, L. Bioinspired surfaces with special wettability. Accounts. Chem. Res. 2005, 38, 644–652.
Liu, M. J.; Zheng, Y. M.; Zhai, J.; Jiang, L. Bioinspired super-antiwetting interfaces with special liquid-solid adhesion. Accounts. Chem. Res. 2009, 43, 368–377.
Roach, P.; Shirtcliffe, N. J.; Newton, M. I. Progess in superhydrophobic surface development. Soft Matter. 2008, 4, 224–240.
van Oss, C. J.; Giese, R. F. Role of the properties and structure of liquid water in colloidal and interfacial systems. J. Disper. Sci. Technol. 2004, 25, 631–655.
Trudeau, T. G.; Jena, K. C.; Hore, D. K. Water structure at solid surfaces of varying hydrophobicity. J. Phys. Chem. C. 2009, 113, 20002–20008.
Matta, C. F.; Hernandez-Trujillo, J.; Tang, T. H.; Bader, R. F. W. Hydrogen-hydrogen bonding: A stabilizing interaction in molecules and crystals. Chem. Eur. J. 2003, 9, 1940–1951.
Abraham, F. F. The interfacial density profile of a Lennard-Jones fluid in contact with a (100) Lennard-Jones wall and its relationship to idealized fluid/wall systems: A Monte Carlo simulation. J. Chem. Phys. 1978, 68, 3713–3716.
Kim, S. H.; Lee, S. Y.; Yang, S. M. Janus microspheres for a highly flexible and impregnable water-repelling interface. Angew. Chem. Int. Edit. 2010, 49, 2535–2538.
Hong, X.; Gao, X. F.; Jiang, L. Application of superhydrophobic surface with high adhesive force in no lost transport of superparamagnetic microdroplet. J. Am. Chem. Soc. 2007, 129, 1478–1479.
Zhao, Y.; Fang, J.; Wang, H. X.; Wang, X. G.; Lin, T. Magnetic liquid marbles: Manipulation of liquid droplets using highly hydrophobic Fe3O4 nanoparticles. Adv. Mater. 2010, 22, 707–710.
Zhang, K.; Liang, Q. L.; Ma, S.; Mu, X. A.; Hu, P.; Wang, Y. M.; Luo, G. A. On-chip manipulation of continuous picoliter-volume superparamagnetic droplets using a magnetic force. Lab. Chip. 2009, 9, 2992–2999.
Velev, O. D.; Prevo, B. G.; Bhatt, K. H. On-chip manipulation of free droplets. Nature 2003, 426, 515–516.
Hunt, T. P.; Issadore, D.; Westervelt, R. M. Integrated circuit/microfluidic chip to programmably trap and move cells and droplets with dielectrophoresis. Lab. Chip 2008, 8, 81–87.
Ichimura, K.; Oh, S. K.; Nakagawa, M. Light-driven motion of liquids on a photoresponsive surface. Science 2000, 288, 1624–1626.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Rights and permissions
Open Access This is an open access article distributed under the terms of the Creative Commons Attribution Noncommercial License ( https://creativecommons.org/licenses/by-nc/2.0 ), which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
About this article
Cite this article
Su, B., Wang, S., Song, Y. et al. A miniature droplet reactor built on nanoparticle-derived superhydrophobic pedestals. Nano Res. 4, 266–273 (2011). https://doi.org/10.1007/s12274-010-0078-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12274-010-0078-5