Abstract
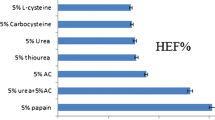
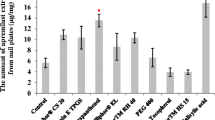
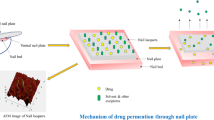
Onychomycosis is a prevailing disease caused by fungal infection of nails that mostly affects athletes and the elderly. Ciclopirox is approved by the US Food and Drug Administration for the topical treatment of onychomycosis. However, the desired penetration of ciclopirox into the nail bed has not been achieved via topical application for efficient treatment. Therefore, the main aim of this study was to enhance ciclopirox permeation and retention in nail by the development of a new nail lacquer formulation. We screened the effects of different solvents, alkalizing agents, and permeation enhancers on the permeation of bovine hooves by ciclopirox and its retention in human nail clippings. The results suggest that isopropyl alcohol, potassium hydroxide, and urea as the solvent, alkalizing agent, and permeation enhancer, respectively, improved the permeation of the ciclopirox nail lacquer formulation the most with high flux rates. Comparison of the final formulation and marketed product revealed enhanced retention of ciclopirox from our developed formulation in human nail clippings. Therefore, our newly developed nail lacquer may be a potentially effective formulation for the treatment of onychomycosis in humans.






Similar content being viewed by others
References
Baden HP, Goldsmith LA, Fleming B (1973) A comparative study of the physicochemical properties of human keratinized tissues. Biochim Biophys Acta 322:269–278
Baran R, Kaoukhov A (2005) Topical antifungal drugs for the treatment of onychomycosis: an overview of current strategies for monotherapy and combination therapy. J Eur Acad Dermatol Venereol 19:21–29
Baran R, Tosti A, Hartmane I, Altmeyer P, Hercogova J, Koudelkova V, Ruzicka T, Combemale P, Mikazans I (2009) An innovative water-soluble biopolymer improves efficacy of ciclopirox nail lacquer in the management of onychomycosis. J Eur Acad Dermatol Venereol 23:773–781
Brown MB, Khengar RH, Turner RB, Forbes B, Traynor MJ, Evans CRG, Jones SA (2009) Overcoming the nail barrier: a systematic investigation of ungual chemical penetration enhancement. Int J Pharm 370:61–67
Caputo R, De Boulle K, Del Rosso J, Nowicki R (2001) Prevalence of superficial fungal infections among sports-active individuals: results from the Achilles survey, a review of the literature. J Eur Acad Dermatol Venereol 15:312–316
Einarson TR, Gupta AK, Shear NH, Arikian S (1996) Clinical and economic factors in the treatment of onychomycosis. Pharmacoeconomics 9:307–320
Elkeeb R, Alikhan A, Elkeeb L, Hui X, Maibach HI (2010) Transungual drug delivery: current status. Int J Pharm 384:1–8
Elsayed MMA (2015) Development of topical therapeutics for management of onychomycosis and other nail disorders: a pharmaceutical perspective. J Control Release 199:132–144
Faergemann J, Baran R (2003) Epidemiology, clinical presentation and diagnosis of onychomycosis. Br J Dermatol 149:1–4
Farran L, Ennos AR, Eichhorn SJ (2008) The effect of humidity on the fracture properties of human fingernails. J Exp Biol 211:3677–3681
Ghannoum MA, Long L, Isham N, Bulgheroni A, Setaro M, Caserini M, Palmieri R, Mailland F (2015) Ability of hydroxypropyl chitosan nail lacquer to protect against dermatophyte nail infection. Antimicrob Agents Chemother 59:1844–1848
Gupta AK, Fleckman P, Baran R (2000) Ciclopirox nail lacquer topical solution 8 % in the treatment of toenail onychomycosis. J Am Acad Dermatol 43:S70–S80
Hargreaves AE (2003) Formulation chemicals. In: Hargreaves T (ed) Chemical formulation: an overview of surfactant based chemical preparations used in everyday life, 1st edn. The royal society of chemistry, Cambridge, pp 7–47
Hay R (2005) Literature review. onychomycosis. J Eur Acad Dermatol Venereol 19:1–7
Hui X, Wester RC, Barbadillo S, Lee C, Patel B, Wortzmman M, Gans EH, Maibach HI (2004) Ciclopirox delivery into the human nail plate. J Pharm Sci 93:2545–2548
Jacobi A, Mayer A, Augustin M (2015) Keratolytics and emollients and their role in the therapy of psoriasis: a systematic review. Dermatol Ther 5:1–18
Khengar RH, Jones SA, Turner RB, Forbes B, Brown MB (2007) Nail swelling as a pre-formulation screen for the selection and optimisation of ungual penetration enhancers. Pharm Res 24:2207–2212
Kobayashi Y, Miyamoto M, Sugibayashi K, Morimoto Y (1999) Drug ermeation through the three layers of the human nail plate. J Pharm Pharmacol 51:271–278
Lusiana Reichl S, Müller-Goymann CC (2011) Keratin film made of human hair as a nail plate model for studying drug permeation. Eur J Pharm Biopharm 78:432–440
Monti D, Saccomani L, Chetoni P, Burgalassi S, Tampucci S, Mailland F (2011) Validation of bovine hoof slices as a model for infected human toenails: in vitro ciclopirox transungual permeation. Br J Dermatol 165:99–105
Murdan S (2002) Drug delivery to the nail following topical application. Int J Pharm 236:1–26
Nogueiras-Nieto L, Begoña Delgado-Charro M, Otero-Espinar FJ (2013) Thermogelling hydrogels of cyclodextrin/poloxamer polypseudorotaxanes as aqueous-based nail lacquers: application to the delivery of triamcinolone acetonide and ciclopirox olamine. Eur J Pharm Biopharm 83:370–377
Palliyil B, Lebo DB, Patel PR (2013) A preformulation strategy for the selection of penetration enhancers for a transungual formulation. AAPS PharmSciTech 14:682–691
Repka MA, Mididoddi PK, Stodghill SP (2004) Influence of human nail etching for the assessment of topical onychomycosis therapies. Int J Pharm 282:95–106
Roberts DT (1999) Onychomycosis: current treatment and future challenges. Br J Dermatol 141:1–4
Shokri J, Azarmi S, Fasihi Z, Hallaj-Nezhadi S, Nokhodchi A, Javadzadeh Y (2012) Effects of various penetration enhancers on percutaneous absorption of piroxicam from emulgels. Res Pharm Sci 7:225–234
Smith KA, Hao J, Li SK (2010) Influence of pH on transungual passive and iontophoretic transport. J Pharm Sci 99:1955–1967
Tabara K, Szewczyk AE, Bienias W, Wojciechowska A, Pastuszka M, Oszukowska M, Kaszuba A (2015) Amorolfine versus ciclopirox–lacquers for the treatment of onychomycosis. Adv Dermatol Allergol/Postȩp Dermatologii i Alergologii 32:40–45
Thomas J, Jacobson GA, Narkowicz CK, Peterson GM, Burnet H, Sharpe C (2010) Toenail onychomycosis: an important global disease burden. J Clin Pharm Ther 35:497–519
Trottet L, Merly C, Mirza M, Hadgraft J, Davis AF (2004) Effect of finite doses of propylene glycol on enhancement of in vitro percutaneous permeation of loperamide hydrochloride. Int J Pharm 274:213–219
Walters KA, Flynn GL (1983) Permeability characteristics of the human nail plate. Int J Cosmet Sci 5:231–246
Acknowledgments
This research study was supported by the Yeungnam University in 2015.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Kang MH, Han SD, Jun JH, and Son MW are employees of Dong-A ST Research Institute while all the other authors have no conflict of interest to declare.
Additional information
R. K. Thapa and J. Y. Choi have contributed equally.
Rights and permissions
About this article
Cite this article
Thapa, R.K., Choi, J.Y., Go, T.G. et al. Development of ciclopirox nail lacquer with enhanced permeation and retention. Arch. Pharm. Res. 39, 953–959 (2016). https://doi.org/10.1007/s12272-016-0774-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12272-016-0774-0