Abstract
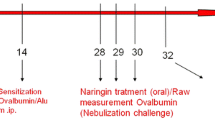
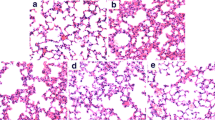
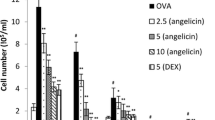
Homoegonol is a lignan derived from styraxlignolide A, which was isolated from Styrax japonica, a medicinal plant widely used for treatment of inflammatory diseases in Korea. We investigated the efficacy of homoegonol for the treatment of allergic asthma using an ovalbumin (OVA)-induced murine asthma model. The mice were sensitized through intraperitoneal injections of OVA on days 0 and 14. On days 21, 22 and 23 after the initial OVA sensitization, the mice were received OVA airway challenge. Homoegonol was administered by oral gavage at a dose of 30 mg/kg 1 h prior to the OVA challenge. The homoegonol-treated mice exhibited reduced inflammatory cell counts and Th2 cytokines in BALF, AHR, and IgE in the serum compared with the OVA-sensitized/challenged mice. The histological analysis of the lung tissue revealed that the administration of homoegonol attenuated the airway inflammation and the mucus overproduction in airway epithelial lesions induced by OVA through a reduction in expression of inducible nitric oxide synthase and matrix metalloproteinase-9. These findings indicate that homoegonol effectively suppresses the asthmatic responses induced by OVA challenge and suggests that homoegonol exhibits potential as therapeutic drug for allergic asthma.








Similar content being viewed by others
References
Adcock, I.M., P.A. Ford, P. Bhavsar, T. Ahmad, and K.F. Chung. 2008. Steroid resistance in asthma: Mechanisms and treatment options. Current Allergy and Asthma Reports 8: 171–178.
Bertanha, C.S., C.G. Braguine, A.C. Moraes, V.M. Gimenez, M. Groppo, M.L. Silva, W.R. Cunha, A.H. Januario, and P.M. Pauletti. 2012. Cyclooxygenase inhibitory properties of nor-neolignans from Styrax pohlii. Natural Product Research 26: 2323–2329.
Bosnjak, B., B. Stelzmueller, K.J. Erb, and M.M. Epstein. 2011. Treatment of allergic asthma: Modulation of Th2 cells and their responses. Respiratory Research 12: 114.
Chibana, K., J.B. Trudeau, A.T. Mustovich, H. Hu, J. Zhao, S. Balzar, H.W. Chu, and S.E. Wenzel. 2008. IL-13 induced increases in nitrite level are primarily driven by increases in inducible nitric oxide synthase as compared with effects on arginases in human primary bronchial epithelial cells. Clinical and Experimental Allergy 36: 936–946.
Di Girolamo, N., I. Indoh, N. Jackson, D. Wakefield, H.P. McNeil, W. Yan, C. Geczy, J.P. Arm, and N. Tedla. 2006. Human mast cell-derived gelatinase B (matrix metalloproteinase-9) is regulated by inflammatory cytokines: Role in cell migration. Journal of Immunology 177: 2638–2650.
Durham, A., I.M. Adcock, and O. Tliba. 2011. Steroid resistance in severe asthma: Current mechanisms and future treatment. Current Pharmaceutical Design 17: 674–684.
Gueders, M.M., J.M. Foidart, A. Noel, and D.D. Cataldo. 2006. Matrix metalloproteinase (MMPs) and tissue inhibitors of MMPs in the respiratory tract: Potential implications in asthma and other lung diseases. European Journal of Pharmacology 533: 133–144.
Hamid, Q., and M. Tulic. 2009. Immunobiology of asthma. Annual Review of Physiology 71: 489–507.
Hansbro, P.M., G.E. Kaiko, and P.S. Foster. 2011. Cytokine/anti-cytokine therapy—novel treatments for asthma? British Journal of Pharmacology 163: 81–95.
Hansbro, P.M., G.V. Scott, A.T. Essilfie, R.Y. Kim, M.R. Starkey, D.H. Nguyen, P.D. Allen, G.E. Kaiko, M. Yang, J.C. Horvat, and P.S. Foster. 2013. Th2 cytokine antagonists: Potential treatments for severe asthma. Expert Opinion on Investigational Drugs 22: 49–69.
Hart, T.K., M.N. Blackbum, M. Brigham-Burke, K. Dede, N. Al-Mahdi, P. Zia-Amirhosseini, and R.M. Cook. 2002. Preclinical efficacy and safety of pascolizumab (SB 240683): A humanized anti-interleukin-4 antibody with therapeutic potential in asthma. Clinical and Experimental Immunology 130: 93–100.
Harikrishnan, R., J.S. Kim, M.C. Kim, C. Balasundaram, and M.S. Heo. 2011. Styrax japonica supplementation diet enhances the innate immune response in Epinephelus bruneus against bacterial and protozoan infections. Experimental Parasitology 129: 260–265.
Hart, T.K., R.M. Cook, P. Zia-Amirhosseini, B.E. Maleeff, S. Eustis, L.W. Schwartz, P. Tsui, E.R. Appelbaum, E.C. Martin, P.J. Bugelski, and D.S. Herzyk. 2001. Preclinical efficacy and safety of mepolizumab (SB-240563), a humanized monoclonal antibody to IL-5, in cynomolgus monkeys. Journal of Allergy and Clinical Immunology 108: 250–257.
Heussen, C., and E.B. Dowdel. 1980. Electrophoretic analysis of plasminogen activators in polyacrylamide gels containing sodium dodecyl sulfate and copolymerized substrate. Analytical Biochemistry 102: 196–202.
Kanda, A., V. Driss, V. Hornez, M. Abdallah, T. Roumier, G. Abboud, F. Legrand, D. Staumont-Salle, S. Queant, J. Bertout, S. Fleury, P. Remy, J.P. Papin, V. Julia, M. Capron, and D. Dombrowicz. 2009. Eosinophil-derived IFN-gamma induces airway hyperresponsiveness and lung inflammation in the absence of lymphocytes. Journal of Allergy and Clinical Immunology 124: 573–582.
Leckie, M.J., A. ten Brinke, J. Khan, Z. Diamant, B.J. O’Connor, C.M. Walls, A.K. Mathur, H.C. Cowley, K.F. Chung, R. Djukanovic, T.T. Hansel, S.T. Holgate, P.J. Sterk, and P.J. Barnes. 2000. Effects of an interleukin-5 blocking monoclonal antibody on eosinophils airway hyperresponsiveness, and the late asthmatic response. Lancet 356: 23–30.
Lee, K.S., S.M. Jin, H.J. Kim, and Y.C. Lee. 2003. Matrix metalloproteinase inhibitor regulates inflammatory cell migration by reducing ICAM-1 and VCAM-1 expression in a murine model of toluene diisocyanate-induced asthma. Journal of Allergy and Clinical Immunology 111: 1278–1284.
Lee, J., and K.T. Lim. 2010. Apoptotic activity of ethanol extract from Styrax Japonica Siebold et al Zuccarini in HepG2 cells. Journal of Ethnopharmacology 131: 210–215.
Lee, J., and K.T. Lim. 2013. SJSZ glycoprotein (38 kDa) inhibits cell cycle and oxidative stress in N-methyl-N′-nitro-N-nitrosoguanidine-induced ICR mice. Anticancer Agents in Medicinal Chemistry 13: 647–653.
Lee, M.Y., J.A. Lee, C.S. Seo, H. Ha, N.H. Lee, and H.K. Shin. 2011. Protective effects of Mentha haplocalyx ethanol extract (MH) in a mouse model of allergic asthma. Phytotherapy Research 25: 863–869.
Liu, L.Y., S.K. Mathur, J.B. Sedgwick, N. Jarjour, W.W. Busse, and E.A. Kelly. 2006. Human airway and peripheral blood eosinophils enhance Th1 and Th2 cytokine secretion. Allergy 61: 689–697.
Locke, N.R., S.G. Royce, J.S. Wanewright, C.S. Samuel, and M.L. Tang. 2007. Comparison of airway remodeling in acute, subacute, and chronic models of allergic airways disease. American Journal of Respiratory Cell and Molecular Biology 36: 625–632.
Martin, P.L., D. Fisher, W. Glass, K. O’Neil, A. Das, E.C. Martin, and L. Li. 2008. Preclinical safety and pharmacology of an anti-human interleukin-13 monoclonal antibody in normal macaques and in macaques with allergic asthma. International Journal of Toxicology 27: 351–358.
Min, B.S., S.R. Oh, K.S. Ahn, J.H. Kim, J. Lee, D.Y. Kim, E.H. Kim, and H.K. Lee. 2004a. Anti-complement activity of norlignans and terpenes from the stem bark of Styrax japonica. Planta Medica 70: 1210–1215.
Min, B.S., M.K. Na, S.R. Oh, K.S. Ahn, G.S. Jeong, G. Li, S.K. Lee, H. Joung, and H.K. Lee. 2004b. New furofuran and butyrolactone lignans with antioxidant activity from the stem bark of Styrax japonica. Journal of Natural Products 67: 1980–1984.
Possa, S.S., E.A. Leick, C.M. Prado, M.A. Martins, and I.F. Tiberio. 2013. Eosinophilic inflammation in allergic asthma. Frontiers in Pharmacology 4: 46.
Rachelefsky, G. 2009. Inhaled corticosteroids and asthma control in children: Assessing impairment and risk. Pediatrics 123: 353–366.
Singh, D., B. Kane, N.A. Molfino, R. Faggioni, L. Roskos, and A. Woodcock. 2010. A phase 1 study evaluating the pharmacokinetics, safety and tolerability of repeat dosing with a human IL-13 antibody (CAT-354) in subjects with asthma. BMC Pulmonary Medicine 10: 3.
Uhm, T.G., B.S. Kim, and I.Y. Chung. 2012. Eosinophil development, regulation of eosinophil-specific genes, and role of eosinophils in the pathogenesis of asthma. Allergy Asthma & Immunology Research 4: 68–79.
Voehringer, D., K. Shinkai, and R. Locksley. 2004. Type 2 immunity reflects orchestrated recruitment of cells committed to IL-4 production. Immunity 20: 267–277.
Wang, W., J.J. Li, P.S. Foster, P.M. Hansbro, and M. Yang. 2010. Potential therapeutic targets for steroid-resistant asthma. Current Drug Targets 11: 957–970.
Williams, C.M., S. Rahman, C. Hubeau, and H.L. Ma. 2012. Cytokine pathways in allergic asthma. Toxicologic Pathology 40: 205–215.
Yun, K.J., B.S. Min, J.Y. Kim, and K.T. Lee. 2007. Styraxoside A isolated from the stem bark of Styrax japonica inhibits lipopolysaccharide-induced expression of inducible nitric oxide synthase and cyclooxygenase-2 in RAW264.7 cells by suppressing nuclear factor-kappa B activation. Biological and Pharmaceutical Bulletin 30: 139–144.
Zhou, D.Y., Q. Du, R.R. Li, M. Huang, Q. Zhang, and G.Z. Wei. 2011. Grape seed proanthocyanidin extract attenuates airway inflammation and hyperresponsiveness in a murine model of asthma by downregulating inducible nitric oxide synthase. Planta Medica 77: 1575–1581.
Acknowledgments
This work was supported by grants from Aju Pharmaceutics Co. (IGM0181211) and from the KRIBB Research Initiative Program (KGM1221312) of the Republic of Korea.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Shin, IS., Ahn, KS., Shin, NR. et al. Homoegonol attenuates the asthmatic responses induced by ovalbumin challenge. Arch. Pharm. Res. 37, 1201–1210 (2014). https://doi.org/10.1007/s12272-013-0327-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12272-013-0327-8