Abstract
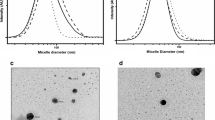
In this study, we prepared amphotericin B (AmpB)-encapsulated polymeric micelle of poly(DL-lactideco-glycolide) (PLGA) grafted-dextran (DexLG) copolymer for the cytotoxicity test. The average particle size of AmpB-encapsulated DexLG polymeric micelles was around 30∼70 nm and their morphology showed spherical shapes. Since aggregation states of AmpB are related to intrinsic cytotoxicity, prevention of AmpB aggregation in aqueous solution will provide low cytotoxicity and increased antimicrobial activity for the infectious disease. At UV/VIS spectrum measurement, polymeric micelle prepared from methanol/water mixture (method B) showed a monomeric state of AmpB while polymeric micelle prepared from DMSO (method A) showed an aggregated state. During the hemolysis activity test, polymeric micelle from method B showed reduced hemolysis activity compared to AmpB itself and polymeric micelle from method A. These results indicated that AmpB-incorporated polymeric micelle prepared from methanol/water mixture has low cytotoxicity and favorable antimicrobial activity.
Similar content being viewed by others
Refernces
Adams, M. L. and Kwon, G. S., Relative aggregation state and hemolytic activity of amphotericin B encapsulated by poly (ethylene oxide)-block-poly(N-hexyl-L-aspartamide)-acyl conjugate micelles: effects of acyl chain length. J. Control. Release, 87, 23–32 (2003).
Barwicz, J., Christian S., and Gruda, I., Effects of aggregation of amphotericin B on its toxicity to mice. Antimicrob. Agents Chemother., 36, 2310–2315 (1992).
Berman, J. D., Human leishmaniasis: clinical, diagnostic and chemotherapeutic developments in past 10 years. Clin. Infect. Dis., 24, 684–703 (1997).
Bolard, J., Legrand, P., Heitz, F., and Cybulska, B., One-sided action of amphotericin B on cholesterol-containing membranes is determined by its self-association in the medium. Biochemistry, 30, 5707–5715 (1991).
Choi, K. C., Bang, J. Y., Kim, P. I., Kim, C., and Song, C. E., Amphotericin B-incorporated polymeric micelles composed of poly(d,l-lactide-co-glycolide)/dextran graft copolymer. Int. J. Pharm., 355, 224–230 (2008).
Deray, G., Amphotericin B nephrotoxicity. J. Antimicrob. Chemother., 49, 37–41 (2002).
Dupont, B., Overview of the lipid formulations of amphotericin B. J. Antimicrob. Chemother. 49(suppl S1), 31–36 (2002).
Gallis, H. A., Drew, R. H., and Pickard, W. W., Amphotericin B: 30 years of clinical experience. Rev. Infect. Dis., 12, 308–329 (1990).
Gref, R., Minamitake, Y., Peracchia, M. T., Trubetskoy, V., Torchilin, V., and Langer, R., Biodegradable long-circulating polymeric nanospheres. Science, 263, 1600–1603 (1994).
Groll, A. H. and Walsh, T. J., Uncommon opportunistic fungi: new nosocomial threats. Clin. Microbiol. Infect.,z 7(Suppl 2), 8–24 (2001).
Ichinose, K., Tomiyama, N., Nakashima, M., Ohya, Y., Ichikawa, M., Ouchi, T., and Kanematsu, T., Antitumor activity of dextran derivatives immobilizing platinum complex (II). Anticancer Drugs, 11, 33–38 (2000).
Kataoka, K., Kwon, G. S., Yokohama, M., Okano, T., and Sakurai, Y., Block copolymer micelles as vehicles for drug delivery. Journal of Controlled Release, 24, 119–132 (1993).
Larabi, M., Yardley, V., Loiseau, P. M., Appel, M., Legrand, P., Gulik, A., Bories, C., Croft, S. L., and Barrat, G., Toxicity and Antileishmanial activity of a new stable lipid suspension of amphotericin B. Antimicrob. Agents Chemother., 47, 3774–3779 (2003).
Lavasanifar, A., Samuel, J., Sattari, S., and Kwon, G. S., Block copolymer micelles for the encapsulation and delivery of amphotericin B. Pharm Res., 19, 418–422 (2002).
Legrand, P., Romero, E., Devissaguet, J. P., Eleazar Cohen, B., and Bolard, J. Effects of aggregation and solvent on toxicity of amphotericin B to human erythrocytes. Antimicrob. Agents Chemother., 36, 2518–2522 (1992).
Mehvar, R., Recent trends in the use of polysaccharides for improved delivery of therapeutic agents: pharmacokinetic and pharmacodynamic perspectives. Current Pharmaceutical Biotechnology, 4, 283–302 (2003).
Walsh, T. J., Hiemenz, J., and Anaissie, E., Recent progress and current problems in treatment of invasive fungal infections in neuropenic patients. Infect. Dis. Clin. North Am., 10, 365–400 (1996).
Vandermeulen, G., Rouxhet, L., Arien, A., Brewster, M. E., and Preat, V., Encapsulation of amphotericin B in poly (ethylene glycol)-block-poly(ɛ-caprolactone-co-trimethylenecarbonate) polymeric micelles. Int. J. Pharm., 309, 234–240 (2006).
Yardley, V. and Croft, S. J., Activity of liposomal amphotericin B against experimental cutaneous leishmaniasis. Antimicrob. Agents Chemother., 41, 752–756 (1997).
Yu, B., Okano, T., Kataoka, K., Sardari, S., and Kwon, G., In vitro dissociation of antifungal efficacy and toxicity for amphotericin B-loaded poly(ethylene oxide)-block-poly(β-benzyl-L-aspartate) micelles. J. control. Release, 56, 285–291 (1998).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bang, JY., Song, CE., Kim, C. et al. Cytotoxicity of amphotericin B-incorporated polymeric micelles composed of poly(DL-lactide-co-glycolide)/dextran graft copolymer. Arch. Pharm. Res. 31, 1463–1469 (2008). https://doi.org/10.1007/s12272-001-2131-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12272-001-2131-0