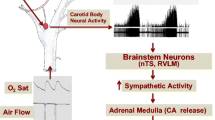
Abstract
The increased incidence of hypertension associated with obstructive sleep apnea (OSA) presents significant physical, psychological, and economic challenges. Peroxisome proliferator-activated receptor gamma (PPARγ) plays a role in both OSA and hypertension, yet the therapeutic potential of PPARγ agonists and antagonists for OSA-related hypertension remains unexplored. Therefore, we constructed a chronic intermittent hypoxia (CIH)-induced hypertension rat model that mimics the pathogenesis of OSA-related hypertension in humans. The model involved administering PPARγ agonist rosiglitazone (RSG), PPARγ antagonist GW9662, or normal saline, followed by regular monitoring of blood pressure and thoracic aorta analysis using staining and electron microscopy. Intriguingly, our results indicated that both RSG and GW9662 appeared to potently counteract CIH-induced hypertension. In silico study suggested that GW9662's antihypertensive effect might mediated through angiotensin II receptor type 1 (AGTR1). Our findings provide insights into the mechanisms of OSA-related hypertension and propose novel therapeutic targets.
Graphical abstract






Similar content being viewed by others
Data Availability
The datasets used and/or analysed during the current study available from the corresponding author on reasonable request.
Abbreviations
- AGTR1:
-
Angiotensin II receptor type 1
- CIH:
-
Chronic intermittent hypoxia
- DBP:
-
Diastolic blood pressure
- GW:
-
GW9662
- H&E:
-
Hematoxylin–eosin
- NS:
-
Normal saline
- Nx:
-
Normoxia
- OSA:
-
Obstructive sleep apnea
- PPARγ:
-
Peroxisome proliferator-activated receptor gamma
- MPAP:
-
Pulmonary arterial pressure
- Rg:
-
Radius of gyration
- RMSD:
-
Root mean square deviation
- RSG:
-
Rosiglitazone
- MSAP:
-
Systemic arterial pressure
- SBP:
-
Systolic blood pressure
- TEM:
-
Transmission electron microscopy
- TZDs:
-
Thiazolidinediones
- EVG:
-
Verhoeff–Van Gieson elastic
References
Lavie P. Obstructive sleep apnoea syndrome as a risk factor for hypertension: population study. BMJ. 2000;320(7233):479–82. https://doi.org/10.1136/bmj.320.7233.479.
Durán J, Esnaola S, Rubio R, Iztueta Á. Obstructive sleep apnea-hypopnea and related clinical features in a population-based sample of subjects aged 30 to 70 yr. Am J Respir Crit Care Med. 2001;163(3):685–9. https://doi.org/10.1164/ajrccm.163.3.2005065.
Young T, Peppard P, Palta M, et al. Population-based study of sleep-disordered breathing as a risk factor for hypertension. Arch Intern Med. 1997;157(15):1746–52.
Hou H, Zhao Y, Yu W, et al. Association of obstructive sleep apnea with hypertension: A systematic review and meta-analysis. J Glob Health. 2018;8(1): 010405. https://doi.org/10.7189/jogh.08.010405.
Yeghiazarians Y, Jneid H, Tietjens JR, et al. Obstructive Sleep Apnea and Cardiovascular Disease: A Scientific Statement From the American Heart Association. Circulation. 2021;144(3). https://doi.org/10.1161/CIR.0000000000000988
Redline S, Azarbarzin A, Peker Y. Obstructive sleep apnoea heterogeneity and cardiovascular disease. Nat Rev Cardiol. 2023;20(8):560–73. https://doi.org/10.1038/s41569-023-00846-6.
Seravalle G, Grassi G. Sleep Apnea and Hypertension. High Blood Press Cardiovasc Prev. 2022;29(1):23–31. https://doi.org/10.1007/s40292-021-00484-4.
Somers VK, Dyken ME, Clary MP, Abboud FM. Sympathetic neural mechanisms in obstructive sleep apnea. J Clin Invest. 1995;96(4):1897–904. https://doi.org/10.1172/JCI118235.
Lima-Junior JGA, Martins AVV, Drager LF. What is the Best Antihypertensive Treatment for OSA? Arch Bronconeumol. 2023;59(9):548–9. https://doi.org/10.1016/j.arbres.2023.03.006.
Diogo LN, Monteiro EC. The efficacy of antihypertensive drugs in chronic intermittent hypoxia conditions. Front Physiol. 2014;5. https://doi.org/10.3389/fphys.2014.00361
Pedrosa RP, Drager LF, De Paula LKG, Amaro ACS, Bortolotto LA, Lorenzi-Filho G. Effects of OSA treatment on BP in patients with resistant hypertension: a randomized trial. Chest. 2013;144(5):1487–94. https://doi.org/10.1378/chest.13-0085.
Muxfeldt ES, Margallo V, Costa LMS, et al. Effects of continuous positive airway pressure treatment on clinic and ambulatory blood pressures in patients with obstructive sleep apnea and resistant hypertension: a randomized controlled trial. Hypertension. 2015;65(4):736–42. https://doi.org/10.1161/HYPERTENSIONAHA.114.04852.
Martínez-García MA, Capote F, Campos-Rodríguez F, et al. Effect of CPAP on blood pressure in patients with obstructive sleep apnea and resistant hypertension: the HIPARCO randomized clinical trial. JAMA. 2013;310(22):2407. https://doi.org/10.1001/jama.2013.281250.
Barbé F, Durán-Cantolla J, Capote F, et al. Long-term effect of continuous positive airway pressure in hypertensive patients with sleep apnea. Am J Respir Crit Care Med. 2010;181(7):718–26. https://doi.org/10.1164/rccm.200901-0050OC.
Scholtes C, Giguère V. Transcriptional control of energy metabolism by nuclear receptors. Nat Rev Mol Cell Biol. 2022;23(11):750–70. https://doi.org/10.1038/s41580-022-00486-7.
Miyachi H. Structural Biology Inspired Development of a Series of Human Peroxisome Proliferator-Activated Receptor Gamma (PPARγ) Ligands: From Agonist to Antagonist. Int J Mol Sci. 2023;24(4):3940. https://doi.org/10.3390/ijms24043940.
Mannan A, Garg N, Singh TG, Kang HK. Peroxisome Proliferator-Activated Receptor-Gamma (PPAR-ɣ): Molecular Effects and Its Importance as a Novel Therapeutic Target for Cerebral Ischemic Injury. Neurochem Res. 2021;46(11):2800–31. https://doi.org/10.1007/s11064-021-03402-1.
Pizcueta P, Vergara C, Emanuele M, Vilalta A, Rodríguez-Pascau L, Martinell M. Development of PPARγ Agonists for the Treatment of Neuroinflammatory and Neurodegenerative Diseases: Leriglitazone as a Promising Candidate. Int J Mol Sci. 2023;24(4):3201. https://doi.org/10.3390/ijms24043201.
Ivanova EA, Parolari A, Myasoedova V, Melnichenko AA, Bobryshev YV, Orekhov AN. Peroxisome proliferator-activated receptor (PPAR) gamma in cardiovascular disorders and cardiovascular surgery. J Cardiol. 2015;66(4):271–8. https://doi.org/10.1016/j.jjcc.2015.05.004.
Nesti L, Tricò D, Mengozzi A, Natali A. Rethinking pioglitazone as a cardioprotective agent: a new perspective on an overlooked drug. Cardiovasc Diabetol. 2021;20(1):109. https://doi.org/10.1186/s12933-021-01294-7.
Huang JV, Greyson CR, Schwartz GG. PPAR-γ as a therapeutic target in cardiovascular disease: evidence and uncertainty. J Lipid Res. 2012;53(9):1738–54. https://doi.org/10.1194/jlr.R024505.
Montaigne D, Butruille L, Staels B. PPAR control of metabolism and cardiovascular functions. Nat Rev Cardiol. 2021;18(12):809–23. https://doi.org/10.1038/s41569-021-00569-6.
Füllert S, Schneider F, Haak E, et al. Effects of pioglitazone in nondiabetic patients with arterial hypertension: a double-blind, placebo-controlled study. J Clin Endocr Metab. 2002;87(12):5503–6 (2020071616072507200).
Komajda M, Curtis P, Hanefeld M, et al. Effect of the addition of rosiglitazone to metformin or sulfonylureas versus metformin/sulfonylurea combination therapy on ambulatory blood pressure in people with type 2 diabetes: a randomized controlled trial (the RECORD study). Cardiovasc Diabetol. 2008;7(1):10. https://doi.org/10.1186/1475-2840-7-10.
Yoshii H, Onuma T, Yamazaki T, et al. Effects of pioglitazone on macrovascular events in patients with type 2 diabetes mellitus at high risk of stroke: the PROFIT-J study. J Atheroscler Thromb. 2014;21(6):563–73.
Dormandy JA, Charbonnel B, Eckland DJ, et al. Secondary prevention of macrovascular events in patients with type 2 diabetes in the PROactive Study (PROspective pioglitAzone Clinical Trial In macroVascular Events): a randomised controlled trial. Lancet. 2005;366(9493):1279–89. https://doi.org/10.1016/S0140-6736(05)67528-9.
Ogihara T, Rakugi H, Ikegami H, Mikami H, Masuo K. Enhancement of insulin sensitivity by troglitazone lowers blood pressure in diabetic hypertensives. Am J Hypertens. 1995;8(3):316–20. https://doi.org/10.1016/0895-7061(95)96214-5.
Gharib SA, Hayes AL, Rosen MJ, Patel SR. A Pathway-Based Analysis on the Effects of Obstructive Sleep Apnea in Modulating Visceral Fat Transcriptome. SLEEP. Published online January 1, 2013. https://doi.org/10.5665/sleep.2294
Li X, Zhang X, Hou X, et al. Obstructive sleep apnea-increased DEC1 regulates systemic inflammation and oxidative stress that promotes development of pulmonary arterial hypertension. Apoptosis. 2023;28(3–4):432–46. https://doi.org/10.1007/s10495-022-01797-y.
Wang H, Xiong W, Hang S, Wang Y, Zhang S, Liu S. Depletion of SENP1-mediated PPARγ SUMOylation exaggerates intermittent hypoxia-induced cognitive decline by aggravating microglia-mediated neuroinflammation. Aging (milano). 2021;13(11):15240–54. https://doi.org/10.18632/aging.203084.
Lian N, Chen M, Zhang S, Chen L, Huang J, Lin Q. Decreased expression of PPARγ is associated with aortic endothelial cell apoptosis in intermittently hypoxic rats. Sleep Breath. 2021;25(4):2241–50. https://doi.org/10.1007/s11325-021-02319-x.
Ning-fang L, Yong-xu J, Jia C, Cai-yun W, Jie-feng H, Qi-chang L. The role of PPARγ in intermittent hypoxia-related human umbilical vein endothelial cell injury. Sleep Breath. 2023;27(3):1155–64. https://doi.org/10.1007/s11325-022-02696-x.
Tache V, Ciric A, Moretto-Zita M, et al. Hypoxia and Trophoblast Differentiation: A Key Role for PPARγ. Stem Cells Dev. 2013;22(21):2815–24. https://doi.org/10.1089/scd.2012.0596.
Rashid J, Alobaida A, Al-Hilal TA, et al. Repurposing rosiglitazone, a PPAR-γ agonist and oral antidiabetic, as an inhaled formulation, for the treatment of PAH. J Controlled Release. 2018;280:113–23. https://doi.org/10.1016/j.jconrel.2018.04.049.
McGuiness JA, Scheinert RB, Asokan A, et al. Indomethacin Increases Neurogenesis across Age Groups and Improves Delayed Probe Trial Difference Scores in Middle-Aged Rats. Front Aging Neurosci. 2017;9:280. https://doi.org/10.3389/fnagi.2017.00280.
Norris S, Carson S, Roberts C. Comparative Effectiveness of Pioglitazone and Rosiglitazone in Type 2 Diabetes, Prediabetes, and the Metabolic Syndrome: A Meta-Analysis. Curr Diabetes Rev. 2007;3(2):127–40. https://doi.org/10.2174/157339907780598216.
Younis F, Stern N, Limor R, Oron Y, Zangen S, Rosenthal T. Telmisartan ameliorates hyperglycemia and metabolic profile in nonobese Cohen-Rosenthal diabetic hypertensive rats via peroxisome proliferator activator receptor–γ activation. Metabolis. 2010;59(8):1200–9. https://doi.org/10.1016/j.metabol.2009.11.013.
Prabhakar NR, Peng Y, Kumar GK, Nanduri J. Peripheral Chemoreception and Arterial Pressure Responses to Intermittent Hypoxia. Prakash YS, ed. Compr Physiol. 2015;5(2):561–77. https://doi.org/10.1002/cphy.c140039.
Dumitrascu R, Heitmann J, Seeger W, Weissmann N, Schulz R. Obstructive Sleep Apnea, Oxidative Stress and Cardiovascular Disease: Lessons from Animal Studies. Oxid Med Cell Longev. 2013;2013:1–7. https://doi.org/10.1155/2013/234631.
Le XT, Thi Nguyen LT, Nguyen PT, et al. Anti-hypertensive effects of Callisia fragrans extract on Reno-vascular hypertensive rats. Clin Exp Hypertens. 2022;44(5):411–8. https://doi.org/10.1080/10641963.2022.2065286.
Cao et al Prenatal Lipopolysaccharides Exposure Induces Transgenerational Inheritance of Hypertension.pdf. 2022 https://doi.org/10.1161/CIRCULATIONAHA.122.059891
Masters L, Eagon S, Heying M. Evaluation of consensus scoring methods for AutoDock Vina, smina and idock. J Mol Graph Model. 2020;96: 107532. https://doi.org/10.1016/j.jmgm.2020.107532.
Van Der Spoel D, Lindahl E, Hess B, Groenhof G, Mark AE, Berendsen HJC. GROMACS: Fast, flexible, and free. J Comput Chem. 2005;26(16):1701–18. https://doi.org/10.1002/jcc.20291.
Mukohda M, Ozaki H. Anti-inflammatory mechanisms of the vascular smooth muscle PPARγ. J Smooth Muscle Res. 2021;57:1–7. https://doi.org/10.1540/jsmr.57.1.
Nikolic D, Castellino G, Banach M, et al. PPAR Agonists, Atherogenic Dyslipidemia and Cardiovascular Risk. Curr Pharm Des. 2017;23(6):894–902. https://doi.org/10.2174/1381612822666161006151134.
Fang S, Livergood MC, Nakagawa P, Wu J, Sigmund CD. Role of the Peroxisome Proliferator Activated Receptors in Hypertension. Circ Res. 2021;128(7):1021–39. https://doi.org/10.1161/CIRCRESAHA.120.318062.
Barroso I, Gurnell M, Crowley VEF, et al. Dominant negative mutations in human PPARgamma associated with severe insulin resistance, diabetes mellitus and hypertension. Nature. 1999;402(6764):880–3. https://doi.org/10.1038/47254.
Auclair M, Vigouroux C, Boccara F, et al. Peroxisome proliferator-activated receptor-γ mutations responsible for lipodystrophy with severe hypertension activate the cellular renin-angiotensin system. Arterioscler, Thromb, Vasc Biol. 2013;33(4):829–38. https://doi.org/10.1161/ATVBAHA.112.300962.
Horio T, Suzuki M, Suzuki K, et al. Pioglitazone Improves Left Ventricular Diastolic Function in Patients With Essential Hypertension. Am J Hypertens. 2005;18(7):949–57. https://doi.org/10.1016/j.amjhyper.2005.02.003.
Horio T, Suzuki M, Takamisawa I, et al. Pioglitazone-Induced Insulin Sensitization Improves Vascular Endothelial Function in Nondiabetic Patients With Essential Hypertension. Am J Hypertens. 2005;18(12):1626–30. https://doi.org/10.1016/j.amjhyper.2005.05.035.
Sobolev VV, Tchepourina E, Korsunskaya IM, et al. The Role of Transcription Factor PPAR-γ in the Pathogenesis of Psoriasis, Skin Cells, and Immune Cells. Int J Mol Sci. 2022;23(17):9708. https://doi.org/10.3390/ijms23179708.
Nissen SE, Wolski K. Effect of rosiglitazone on the risk of myocardial infarction and death from cardiovascular causes. N Engl J Med. 2007;356(24):2457–71. https://doi.org/10.1056/NEJMoa072761.
Lazar MA. Reversing the curse on PPARγ. J Clin Invest. 2018;128(6):2202–4. https://doi.org/10.1172/JCI121392.
Suzuki G, Khanal S, Rastogi S, et al. Long-term Pharmacological Activation of PPARγDoes not Prevent Left Ventricular Remodeling in Dogs with Advanced Heart Failure. Cardiovasc Drugs Ther. 2007;21(1):29–36. https://doi.org/10.1007/s10557-007-6003-9.
Zaki HA, Iftikhar H, Shallik NA, et al. A Systematic Review and Meta-Analysis of Randomized Controlled Trials Comparing the Effects of Biguanides (Metformin) and Thiazolidinediones on Glucose Tolerance and Insulin Sensitivity in Patients With Type II Diabetes Mellitus. Cureus. 2023;15(5): e39445. https://doi.org/10.7759/cureus.39445.
Rieusset J, Touri F, Michalik L, et al. A new selective peroxisome proliferator-activated receptor gamma antagonist with antiobesity and antidiabetic activity. Mol Endocrinol. 2002;16(11):2628–44 (2016092613584000132).
Santini E, Fallahi P, Ferrari SM, Masoni A, Antonelli A, Ferrannini E. Effect of PPAR-␥ Activation and Inhibition on Glucose-Stimulated Insulin Release in INS-1e Cells. 2004;53. https://doi.org/10.2337/diabetes.53.suppl_3.S79
Kwak HJ, Choi HE, Jang J, et al. Suppression of Adipocyte Differentiation by Foenumoside B from Lysimachia foenum-graecum Is Mediated by PPARγ Antagonism. Peterson JM, ed. PLOS One. 2016;11(5):e0155432. https://doi.org/10.1371/journal.pone.0155432.
Zhang J, Liu X, Xie XB, Cheng XC, Wang RL. Multitargeted bioactive ligands for PPARs discovered in the last decade. Chem Biol Drug Des. 2016;88(5):635–63. https://doi.org/10.1111/cbdd.12806.
Mehta RG, Peng X, Roy S, et al. PPARγ antagonist GW9662 induces functional estrogen receptor in mouse mammary organ culture: potential translational significance. Mol Cell Biochem. 2013;372(1–2):249–56. https://doi.org/10.1007/s11010-012-1466-9.
Zhang Y, Yu L, Cai W, et al. Protopanaxatriol, a novel PPARγ antagonist from Panax ginseng, alleviates steatosis in mice. Sci Rep. 2014;4(1):7375. https://doi.org/10.1038/srep07375.
Singh A, Nunes JJ, Ateeq B. Role and therapeutic potential of G-protein coupled receptors in breast cancer progression and metastases. Eur J Pharmacol. 2015;763(Pt B):178–83. https://doi.org/10.1016/j.ejphar.2015.05.011.
Zhuang Y, Niu F, Liu D, et al. Association between AGTR1 A1166C polymorphism and the susceptibility to diabetic nephropathy. Medicine (Baltimore). 2018;97(41): e07689. https://doi.org/10.1097/MD.0000000000007689.
Wang Q, You L, Li Z, Zhang L, Li X, Yang X. Influence of AGTR1 and ABCB1 Gene Polymorphism on the Curative Effect of Irbesartan. Katsuya T, ed. Int J Hypertens. 2022;2022:1–8. https://doi.org/10.1155/2022/4278675.
Fletcher EC, Orolinova N, Bader M. Blood pressure response to chronic episodic hypoxia: the renin-angiotensin system. J Appl Physiol. 2002;92(2):627–33. https://doi.org/10.1152/japplphysiol.00152.2001.
Guan Y, Hao C, Cha DR, et al. Thiazolidinediones expand body fluid volume through PPARgamma stimulation of ENaC-mediated renal salt absorption. Nat Med. 2005;11(8):861–6. https://doi.org/10.1038/nm1278.
Nesto RW, Bell D, Bonow RO, et al. Thiazolidinedione use, fluid retention, and congestive heart failure: a consensus statement from the American Heart Association and American Diabetes Association. Diabetes Care. 2004;27(1):256–63. https://doi.org/10.2337/diacare.27.1.256.
Kriska T, Cepura C, Gauthier KM, Campbell WB. Role of macrophage PPARγ in experimental hypertension. Am J Physiol-heart C. 2014;306(1):H26–32. https://doi.org/10.1152/ajpheart.00287.2013.
Del Rio R, Moya EA, Iturriaga R. Carotid body and cardiorespiratory alterations in intermittent hypoxia: the oxidative link. Eur Respir J. 2010;36(1):143–50. https://doi.org/10.1183/09031936.00158109.
Kumar GK, Rai V, Sharma SD, et al. Chronic intermittent hypoxia induces hypoxia-evoked catecholamine efflux in adult rat adrenal medulla via oxidative stress. J Physiol. 2006;575(1):229–39. https://doi.org/10.1113/jphysiol.2006.112524.
Kanagy NL, Walker BR, Nelin LD. Role of in intermittent hypoxia-induced hypertension. Hypertension. 2001;37(2):511–5. https://doi.org/10.1161/01.hyp.37.2.511.
Belaidi E, Joyeux-Faure M, Ribuot C, Launois SH, Levy P, Godin-Ribuot D. Major role for hypoxia inducible factor-1 and the endothelin system in promoting myocardial infarction and hypertension in an animal model of obstructive sleep apnea. J Am Coll Cardiol. 2009;53(15):1309–17. https://doi.org/10.1016/j.jacc.2008.12.050.
Guo C, Zhang M, Su W, Xu M, Zhao S. miR-199a-5p Relieves Obstructive Sleep Apnea Syndrome-Related Hypertension by Targeting HIF-1α. Wang F, ed. J Immunol Res. 2022;2022:1–11. https://doi.org/10.1155/2022/7236647.
He L, Liao X, Zhu G, Kuang J. miR-126a-3p targets HIF-1α and alleviates obstructive sleep apnea syndrome with hypertension. Hum Cell. 2020;33(4):1036–45. https://doi.org/10.1007/s13577-020-00404-z.
Rodriguez J, Escobar JB, Cheung EC, et al. Hypothalamic Oxytocin Neuron Activation Attenuates Intermittent Hypoxia-Induced Hypertension and Cardiac Dysfunction in an Animal Model of Sleep Apnea. Hypertension. 2023;80(4):882–94. https://doi.org/10.1161/HYPERTENSIONAHA.122.20149.
Lu D, Wang J, Zhang H, Shan Q, Zhou B. Renal denervation improves chronic intermittent hypoxia induced hypertension and cardiac fibrosis and balances gut microbiota. Life Sci. 2020;262: 118500. https://doi.org/10.1016/j.lfs.2020.118500.
Funding
Funding This work was supported by the National Natural Science Foundation of China (82270456, 81970445).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Ethical Approval
No human studies were carried out by the authors for this article.
All institutional and national guidelines for the care and use of laboratory animals were followed and approved by Fudan University's Animal Ethics Committee, China.
Conflict of Interest
The authors declared no competing interests.
Additional information
Editor-in-Chief Enrique Lara-Pezzi oversaw the review of this article
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhang, N., Wei, F., Ning, S. et al. PPARγ Agonist Rosiglitazone and Antagonist GW9662: Antihypertensive Effects on Chronic Intermittent Hypoxia-Induced Hypertension in Rats. J. of Cardiovasc. Trans. Res. (2024). https://doi.org/10.1007/s12265-024-10499-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12265-024-10499-6