Abstract
Irisin, a myokine mainly secreted by skeletal and cardiac muscles, is actively involved in cardiovascular diseases. However, whether irisin is associated with aortic stenosis remains unknown. Two hundred ninety-three severe AS patients who underwent transcatheter aortic valve implantation were enrolled and followed-up for 35 months on average. Enzyme-linked immunosorbent assay (ELISA) was applied to measure circulating irisin levels. Patients were divided into two groups based on the median plasma irisin level. We found that high plasma irisin levels were independently associated with pure aortic stenosis (PAS) after adjusting for age, body mass index, history of peripheral vascular disease, and creatinine (OR = 3.015, 95% CI 1.775–5.119, P < 0.001). ROC curve analysis showed a significant predictive value of irisin for PAS (AUC = 0.647, 95% CI 0.583–0.711, P < 0.001). The severity of aortic valve calcification was negatively associated with plasma irisin levels (P < 0.05). In conclusion, irisin is an independent predictor for PAS and is negatively associated with the severity of aortic valve calcification.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Aortic stenosis (AS) is the most common valvular heart disease in elderly people. The prevalence of AS increases with age, accounting for only 0.2% in people aged 50 to 59 years, and increasing to 9.8% in people aged ≥ 80 years [1]. Progressive narrowing of the aortic valve leads to an increased pressure gradient, left ventricular hypertrophy, and left ventricular diastolic dysfunction, which subsequently leads to a decrease in exercise tolerance and even heart failure. Studies have shown that patients with AS have a long asymptomatic period during which their survival rates are similar to those of healthy people. Once typical symptoms appear, such as dyspnea, syncope, and angina, survival rates are sharply reduced [2]. Patients with severe aortic stenosis with symptoms have an annual mortality rate of approximately 25%, and 75% of patients die within 3 years [3]. Current academic guidelines recommend intervention in patients with severe aortic stenosis who have symptoms, significantly reduced left ventricular ejection fraction (< 50%), or rapid progression [4]. Since its first clinical application in 2002, transcatheter aortic valve implantation (TAVI) has provided a new option for patients who cannot tolerate surgery or who are at high risk of surgery [5], and its efficacy has been fully recognized in multiple clinical studies. Thus, there is a trend to expand TAVI to patients with moderate- to low-risk of surgery. A systematic preoperative assessment of TAVI, including anatomy, hemodynamic status, and surgical risk assessment, is critical to determine the suitability and benefit of TAVI treatment in patients with AS. Currently available mainstream surgical risk scoring system includes the Society of Thoracic Surgeons (STS) score and the European System for Cardiac Operative Risk Evaluation (EuroSCORE) score. However, their association with the peri-operative risk of interventional operation was not as significant as that noted for surgery [6]. In addition, frailty (muscle weakness, muscle atrophy, malnutrition, and slow gait) and neurocognitive function are also major factors influencing TAVI postoperative outcomes [7], and these variables were incorporated into the preoperative risk assessment system for TAVI in patients with AS. The above assessment scoring system is computationally complex, especially for the assessment of frailty, which is susceptible to subjective factors. Thus, more objective and reliable biomarkers are needed to create an ideal tool for interventional risk assessment and prognosis analysis in patients with AS.
Irisin, a 112-amino acid myokine cleaved from the plasma membrane protein fibronectin type III domain containing protein 5 (FNDC5), is mainly secreted in response to exercise [8]. In addition to skeletal muscle, cardiac muscle can also secrete large amounts of irisin [9]. Previous studies demonstrated that this myokine-regulated metabolism, affected energy supply, and was associated with multiple metabolic diseases, such as diabetes mellitus (DM), chronic kidney disease, and macrovascular disease [10]. Some studies reported that plasma irisin levels were significantly reduced and negatively correlated with creatine phosphokinase-myocardial band isoenzyme (CK-MB) levels in infarct rats and patients with myocardial infarction (MI)[11,12,13]. Plasma irisin level could be applied to predict mortality in patients with acute heart failure (AHF)[14]. However, whether irisin is associated with aortic stenosis remains unknown. Reduced muscle mass is common in AS patients due to decreased activity tolerance. Studies have demonstrated that sarcopenia is strongly associated with prognosis after TAVI in AS patients [15]. Low levels of circulating irisin are a sensitive marker of muscle weakness and atrophy[16]. Combined with the strong association between frailty state and postoperative prognosis of AS patients after TAVI, we believe that plasma irisin levels may be easier to measure and quantify than frailty state or muscle content, and may also be related to AS patients prognosis after TAVI.
In the current study, a total of 293 severe AS patients who underwent TAVI were consecutively enrolled to explore the association between circulating irisin levels and baseline characteristics in AS patients and to assess whether baseline irisin levels could predict mortality in symptomatic AS patients who underwent TAVI.
Methods
This study consecutively enrolled symptomatic AS patients who underwent TAVI in the Second Affiliated Hospital, Zhejiang University School of Medicine (SAHZU) from March 2013 to November 2018. The inclusion criteria were as follows: hospitalization for symptomatic AS, underwent TAVI, 18 years or older, severe AS (mean gradient evaluated by echocardiography). We excluded those who previously performed coronary artery bypass grafting surgery. Finally, 293 patients who completed the TAVI procedure were included in our study. Patients were followed up at 1 month and every year after the TAVI procedure. The last follow-up visit for the current study occurred in January 2020. The median follow-up period was 35 months. Baseline characteristics and echocardiographic and computed tomography (CT) parameters were collected before the TAVI procedure. We also obtained survival data on all-cause mortality, cardiovascular mortality, or noncardiovascular mortality (Valve Academic Research Consortium (VARC)- 2 definition) during the follow-up. The plasma irisin level was measured from blood samples obtained before the TAVI procedure using an enzyme-linked immunosorbent assay (ELISA) kit (Catalog No. EK-067–29, Phoenix Biotech, TX, USA), according to the manufacturer’s manual. The study complied with the principles of the Declaration of Helsinki regarding investigation in humans and this study protocol was approved by the Human Research Ethics Committee at SAHZU. All patients provided written informed consent.
For the present analysis, the patient population was divided into 2 groups based on the median pre-TAVI plasma irisin level reading (13.70 ng/mL). The primary study endpoint was overall survival after TAVI treatment. Cardiovascular death and noncardiovascular death were considered to be the secondary study endpoints. Death was confirmed by inspection of the death certificate or verified with a family member.
SPSS Statistics for Windows (IBM, NY, USA), version 25.0 was used to perform all statistical analyses. Continuous variables were tested for normality of distribution with the Shapiro–Wilk test and are presented as the mean ± SD or median and interquartile range (IQR). Differences between two groups of patients were analyzed using Student’s t test (normally distributed variables) or the Wilcoxon rank-sum test (nonnormally distributed variables). Categorical variables were expressed as numbers (percentage) and were compared using χ2 tests or Fisher’s exact test when appropriate. Spearman rank correlation analysis and logistic regression analysis were conducted to explore the associations of baseline variables and clinical outcomes with circulating irisin levels. Kaplan‒Meier analysis was used to evaluate and compare the time-to-event curve between the two groups. All analyses were considered statistically significant at a two-tailed P value of less than 0.05.
Variables were selected based on overall clinical relevance. Clinical characteristics including age, sex, body mass index (BMI), smoking status, frailty (KATZ score < 6) [17], Society of Thoracic Surgeons (STS) score, pure aortic stenosis (PAS) or mixed aortic valve disease (MAVD), NYHA class, medical history of dyslipidemia, DM, hypertension, stroke, MI, peripheral vascular disease (PVD), and previous percutaneous coronary intervention (PCI) were recorded. Serum or plasma creatinine levels, estimated glomerular filtration rate (eGFR), pro-brain natriuretic peptide (pro-BNP), CK-MB, and cardiac troponin-T (cTnT) were measured from venous blood samples prior to TAVI procedures. Echocardiography was performed in all patients to record the left ventricular ejection fraction (LVEF), left atrium size (LA), pulmonary artery systolic pressure (PASP), left ventricular end-diastolic dimension (LVEDd), mean pressure gradient across the aortic valve (mPG), aortic valve area (AVA), maximum transaortic velocity (Vmax), aortic regurgitation (AR) grade, mitral stenosis (MS) grade, mitral regurgitation (MR) grade, tricuspid regurgitation (TR) grade, and bicuspid or tricuspid aortic valve (BAV or TAV) at baseline. Aortic valve type (BAV versus TAV) and calcification grade were assessed by CT using 3mensio software (3mensio Medical Imaging BV, Bilthoven, the Netherlands). The AR/MS/MR/TR grade was categorized as follows: grade 0—none, 1—mild, 2—moderate, 3—moderate to severe, 4—severe. Aortic valve calcifications were graded in a 5-scale semiquantitative system: grade 0—noncalcification; grade 1—mild calcification (small isolated spots); grade 2—moderate calcification (multiple large spots); grade 3—severe calcification (extensive calcification of all cusps with fusion); and grade 4—massive calcification (large calcifications outreaching the annulus level) [18].
Results
Study Population
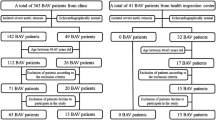
A total of 293 severe AS patients who underwent TAVI in SAHZU were included. The baseline clinical characteristics, laboratory findings, and echocardiographic or CT parameters of the study population are shown in Tables 1 and 2. The average age of the study population was 77 years and 58% were male. The median irisin level reading was 13.70 (IQR 10.95–17.96) ng/mL. The differences in age, frailty, CK-MB level, creatinine level, PAS, medical history of PVD, and dyslipidemia between the two groups all reached statistical significance (P < 0.05). Participants with high irisin levels were older and had relatively lower CK-MB levels, lower creatinine levels, and less frailty, PVD or dyslipidemia medical history. These patients also had a higher prevalence of PAS. Regarding echocardiographic features, a smaller LVEDd, lower PASP, and less aortic calcification were found, whereas a higher prevalence of BAV was observed in the high irisin level group (P < 0.05). Diverse aortic valve calcification grades and aortic regurgitation grades all reached significant differences between the two groups of AS patients in general (P < 0.05).
However, no significant disparities in sex, BMI, eGFR, pro-BNP, cTnT, smoking status, history of previous PCI, STS score, or NYHA class were noted between the two groups. In addition, there was no association between plasma irisin levels and comorbidities of severe AS patients, including DM, hypertension, stroke, or MI (P > 0.05 for all). Furthermore, baseline echocardiographic parameters including LVEF, LA size, AVA, mPG, Vmax, MS grade, MR grade, and TR grade were comparable between the two groups.
Clinical Associations of Plasma Irisin Levels in AS
To further uncover the associations between baseline variables and irisin levels, Spearman rank correlation analysis analyses were performed (Table 3). Frailty, PAS or MAVD, and aortic valve calcification grade were strongly associated with plasma irisin levels (P < 0.001). In addition, age, creatinine, CK-MB, medical history of PVD, and dyslipidemia were also significantly different between the two irisin groups (P < 0.05). In addition, several imaging characteristics, such as LVEDd, PASP, BAV or TAV, and AR grade, also exhibited statistically significant differences in patients with different plasma irisin levels (P < 0.05). A scatter plot of the correlation between irisin and continuous variables is presented in Fig. 1.
Then we conducted logistic regression analysis to certify associations among frailty, PAS or MAVD, aortic valve calcification grade, and plasma irisin level. After adjusting for age, sex, medical history of DM, and creatinine level, a low plasma irisin level was independently associated with frailty (OR = 0.091, 95% CI 0.043–0.195, P < 0.001). In addition, multivariate logistic regression analysis revealed that a high plasma irisin level was independently associated with PAS when adjusting for age, BMI, medical history of PVD, and creatinine level (OR = 3.015, 95% CI 1.775–5.119, P < 0.001). Further receiver operating characteristic (ROC) curve analysis showed a significant predictive value of plasma irisin levels for PAS (AUC = 0.647, 95% CI 0.582–0.711, P < 0.001) (Fig. 2). The cutoff value of irisin for the diagnosis of PAS was 14.20 ng/mL. Univariate logistic regression analysis demonstrated that patients with severe and massive aortic valve calcification had significantly lower plasma irisin levels than patients with no aortic valve calcification (Table 4). After adjusting for age, sex, BMI, creatinine, and medical history of hyperlipidemia, hypertension, and DM, plasma irisin levels remained significantly different between patients with massive aortic valve calcification and no aortic valve calcification (OR = 0.158, 95% CI 0.060–0.415, P < 0.001).
Plasma Irisin Level and Clinical Outcomes in AS
During the whole follow-up period, 52 (17.7%) patients suffered from all-cause death, and 23 (7.8%) patients died due to cardiovascular events. Compared with the high irisin level group, the all-cause death rate at the whole follow-up was significantly higher in patients with lower plasma irisin levels (23.1% versus 12.3%, P = 0.016). However, cardiovascular mortality at the whole follow-up was comparable in the two groups. Similarly, no significant difference in all-cause or cardiovascular mortality at the 1-month or the 1-year follow-up was noted between plasma irisin dichotomous groups. To further confirm the association between plasma irisin levels and all-cause mortality in AS patients, we conducted logistic regression analysis. Univariate logistic analysis indicated that all-cause mortality at the whole follow-up was significantly different between irisin dichotomous groups (OR = 0.467, 95% CI 0.250–0.873, P = 0.017). After adjusting for age, sex, BMI, creatinine, PAS or MAVD, and the calcification grade of the aortic valve in multivariate logistic analysis, the all-cause mortality of AS patients at the whole follow-up was no longer significantly different between the two groups (OR = 0.591, 95% CI 0.287–1.217, P = 0.153). The clinical outcomes of the dichotomous plasma irisin groups are shown in Table 5.
To compare the time-to-event curve between different plasma irisin levels, Kaplan–Meier survival analysis with the log-rank test of all-cause and cardiovascular mortality was performed among all patients. The results indicated similar death rates of all-cause mortality between patients in the dichotomous irisin group throughout the entire follow-up (log-rank P = 0.507). Similarly, cardiovascular mortality rates exhibited no significant difference between the dichotomous irisin groups (log-rank P = 0.545) (Fig. 3).
Discussion
Our study was the first to pay close attention to exploring the association of plasma irisin with baseline characteristics in AS patients and evaluating the implication of baseline plasma irisin levels on post-TAVI clinical outcomes of AS patients. Spearman rank correlation analysis indicated that frailty, PAS or MAVD, and the calcification grade of the aortic valve were strongly associated with plasma irisin levels. Further analysis demonstrated that the low plasma irisin level was independently associated with frailty, and the high plasma irisin level was independently associated with PAS. ROC curve analysis showed a significant predictive value of plasma irisin levels for PAS (AUC = 0.647, 95% CI 0.582–0.711, P < 0.001). Participants with massive aortic valve calcification had lower plasma irisin levels than patients with no aortic valve calcification. Plasma irisin levels were inversely associated with all-cause death rates during the whole follow-up period, whereas this association disappeared after adjusting for confounding variables. Additionally, survival analysis demonstrated similar time-to-event curves between patients in the dichotomous irisin groups during follow-up. We also explored the associations of other risk factors including pro-BNP, frailty scoring, and troponin with clinical outcomes (see supplemental materials).
Previous studies have demonstrated that cardiac muscles are capable of producing a large amount of irisin [9]. However, the role of irisin in the pathogenesis of cardiovascular diseases remains controversial. In the heart tissue and circulation of the murine MI model, irisin expression was lower and negatively correlated with cardiac damage markers, such as troponin and CK-MB [19]. Similarly, circulating irisin levels were decreased in patients with either stable coronary artery disease or MI versus healthy controls [12]. Researchers hypothesized that to resist reduced energy availability, the myocardium may release less irisin to restrain the metabolic demands in the state of deficient blood and oxygen supply [12]. According to this hypothesis, we suppose prolonged left ventricular ejection time and decreased cardiac output caused by AS results in myocardial ischemia and increased myocardial oxygen consumption, which subsequently leads to decreased irisin level in the heart tissue and circulation of AS patients. Shen et al. provided evidence that plasma irisin levels were significantly higher in AHF patients who died at the 1-year follow-up [14]. Increased irisin expression in hypertrophic murine heart and plasma improved cardiac function and reduced pressure overload-induced cardiac hypertrophy and fibrosis [20]. Therefore, we hypothesized that irisin expression may temporarily and reflexively increase as a compensatory mechanism when severe AS patients suffer acute heart failure or stress. Once severe AS is improved after TAVI, cardiac irisin expression levels may change. In addition, decreased activity tolerance caused by frailty in severe AS patients could reduce circulating irisin secreted by skeletal muscle, which may also change the intrinsic irisin balance of the body and influence the prognostic value of irisin. Therefore, baseline plasma irisin levels measured before TAVI may not strongly predict the prognosis of severe AS patients. Hospitalized patients we included had already been in a state of cardiac stress and heart failure due to severe AS, so further studies could consider measuring circulating irisin levels in AS patients with no symptoms or mild symptoms to exclude the influence of heart failure and cardiac stress state. In addition, we can also explore the relationship between irisin and AS by measuring postoperative circulating irisin levels before discharge as a baseline and monitoring changes in circulating irisin levels during postoperative follow-up. As a metabolically related biomarker, irisin may better reflect body status, which could replace subjective biomarkers such as frailty in risk evaluation and stratification of AS.
Previous studies demonstrated that patients with PAS who underwent TAVR had worse survival than patients with MAVD [21,22,23]. In addition, PAS without regurgitation is a risk factor for early stroke after TAVR [24]. However, patients with MAVD exhibited more frequent prosthetic valve regurgitation, a higher operative risk, more severe adverse cardiac remodeling, and worse functional status than patients with PAS [25]. Our study demonstrates that the plasma irisin level has a strong predictive value for PAS, suggesting that irisin could aid in the risk stratification of interventional therapy and prognosis evaluation for AS patients.
Calcific aortic stenosis is the most common cardiac valve lesion in developed countries [26]. The process of cardiac valvular calcification shares a similar pathophysiological mechanism with bone formation including promotion of osteogenesis and loss of mineralization inhibitors, which lead to the deposition of extracellular matrix and calcium phosphate crystals in cardiac valves [27]. Colaianni et al. indicated that irisin could induce osteoblast differentiation: increase the expression of osteoblastic transcription regulators, such as Runt-related transcription factor-2 and osterix; and upregulate osteoblast differentiation markers, including alkaline phosphatase, collagen type 1 alpha-1, osteocalcin, and osteopontin [28]. Consistent with our findings, a previous study also demonstrated that the irisin level was significantly lower in patients with vascular calcification than in those without vascular calcification. A lower irisin level was an independent risk factor for vascular calcification in hemodialysis patients [29]. More studies are needed to elucidate the pathophysiological mechanisms of the interaction between irisin and valve calcification in AS. It has been demonstrated that aortic valve calcification is significantly associated with an increase in LA dilation, left ventricular hypertrophy, peak velocity, and the mean gradient, and a decrease in AVA [30]. However, our study did not identify an association between circulating irisin and these hemodynamic parameters of AS severity. A large portion of our patients had AS combined with AR. Hemodynamic parameters of AS, such as valve diameter and peak velocity, are influenced by anatomical structure and systemic circulation pressure, especially when combined with AR. Therefore, low flow and a low gradient of the aortic valve caused by AR and decreased myocardial contractility would largely influence circulating irisin levels. The peak velocity did not accordingly increase in severe aortic valve stenosis with low flow and a low gradient, which would influence the study result. Therefore, we hypothesize that irisin may have a greater effect on metabolism than hemodynamic parameters.
It is well described that men present more calcification than women when assessing the physiopathology of AS in patients with the same AS severity [31]. Although our study also showed sex differences in aortic valve calcification, no sex difference in irisin levels was observed. Similarly, a cross-sectional study indicated no sex-specific difference in circulating irisin levels in a healthy cohort aged between 20 and 80 years old [32]. However, our study found that elderly men have lower irisin levels than elderly women. An animal study showed that the removal of sex hormones in ovariectomized rats resulted in increased circulating irisin [33], suggesting that a compensatory mechanism may exist in postmenopausal women.
The study was subjected to several limitations as follows. First, our analysis was a retrospective follow-up study. We only measured plasma irisin levels at baseline, which may not accurately reflect their distribution and change during the follow-up period. Second, this study was restricted to 293 AS patients who underwent TAVI in one institution without randomization and healthy population controls. Although we adjusted the outcome for diverse parameters, potential residual confounding factors may confuse the results. Undoubtedly, our findings need to be validated in a larger population and multiple institutions.
Conclusion
Plasma irisin levels are negatively associated with the severity of aortic valve calcification, and high plasma irisin levels are an independent predictor for pure aortic stenosis in the context of mixed aortic valve disease. These findings imply an important role of irisin in aortic stenosis.
Data Availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- AS:
-
Aortic stenosis
- TAVI:
-
Transcatheter aortic valve implantation
- TAVR:
-
Transcatheter aortic valve replacement
- PAS:
-
Pure aortic stenosis
- MAVD:
-
Mixed aortic valve disease
- STS:
-
The Society of Thoracic Surgeons
- EuroSCORE:
-
The European System for Cardiac Operative Risk Evaluation
- FNDC5:
-
Fibronectin type III domain containing protein 5
- MI:
-
Myocardial infarction
- CK-MB:
-
Creatine phosphokinase-myocardial band isoenzyme
- AHF:
-
Acute heart failure
- CT:
-
Computed tomography
- VARC:
-
Valve Academic Research Consortium
- ELISA:
-
Enzyme-linked immunosorbent assay
- BMI:
-
Body mass index
- DM:
-
Diabetes mellitus
- PVD:
-
Peripheral vascular disease
- PCI:
-
Percutaneous coronary intervention
- eGFR:
-
Estimated glomerular filtration rate
- pro-BNP:
-
Pro-brain natriuretic peptide
- cTnT:
-
Cardiac troponin-T
- LVEF:
-
Left ventricular ejection fraction
- LA:
-
Left atrium
- PASP:
-
Pulmonary artery systolic pressure
- LVEDd:
-
Left ventricular end-diastolic dimension
- mPG:
-
Mean pressure gradient
- AVA:
-
Aortic valve area
- Vmax :
-
Maximum transaortic velocity
- AR:
-
Aortic regurgitation
- MS:
-
Mitral stenosis
- MR:
-
Mitral regurgitation
- TR:
-
Tricuspid regurgitation
- BAV:
-
Bicuspid aortic valve
- TAV:
-
Tricuspid aortic valve
- NYHA:
-
New York Heart Association
- IQR:
-
Interquartile range
- ROC:
-
Receiver operating characteristic
- IL-6:
-
Interleukin-6
References
Eveborn GW, Schirmer H, Heggelund G, Lunde P, Rasmussen K. The evolving epidemiology of valvular aortic stenosis. the Tromsø study. Heart (British Cardiac Soc). 2013;99:396–400.
Ross J Jr, Braunwald E. Aortic stenosis. Circulation. 1968;38:61–7.
Carabello BA, Paulus WJ. Aortic stenosis. Lancet (London England). 2009;373:956–66.
Otto CM, Nishimura RA, Bonow RO, Carabello BA, Erwin JP 3rd, Gentile F, Jneid H, Krieger EV, Mack M, McLeod C, O’Gara PT, Rigolin VH, Sundt TM 3rd, Thompson A, Toly C. 2020 ACC/AHA guideline for the management of patients with valvular heart disease: executive summary: a report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. J Am Coll Cardiol. 2021;77:450–500.
Makkar RR, Fontana GP, Jilaihawi H, Kapadia S, Pichard AD, Douglas PS, Thourani VH, Babaliaros VC, Webb JG, Herrmann HC, Bavaria JE, Kodali S, Brown DL, Bowers B, Dewey TM, Svensson LG, Tuzcu M, Moses JW, Williams MR, Siegel RJ, Akin JJ, Anderson WN, Pocock S, Smith CR, Leon MB. Transcatheter aortic-valve replacement for inoperable severe aortic stenosis. N Engl J Med. 2012;366:1696–704.
Dewey TM, Brown D, Ryan WH, Herbert MA, Prince SL, Mack MJ. Reliability of risk algorithms in predicting early and late operative outcomes in high-risk patients undergoing aortic valve replacement. J Thorac Cardiovasc Surg. 2008;135:180–7.
Hinterbuchner L, Strohmer B, Hammerer M, Prinz E, Hoppe UC, Schernthaner C. Frailty scoring in transcatheter aortic valve replacement patients. European J Cardiovasc Nursing. 2016;15:384–97.
Boström P, Wu J, Jedrychowski MP, Korde A, Ye L, Lo JC, Rasbach KA, Boström EA, Choi JH, Long JZ, Kajimura S, Zingaretti MC, Vind BF, Tu H, Cinti S, Højlund K, Gygi SP, Spiegelman BM. A PGC1-α-dependent myokine that drives brown-fat-like development of white fat and thermogenesis. Nature. 2012;481:463–8.
Aydin S, Kuloglu T, Aydin S, Eren MN, Celik A, Yilmaz M, Kalayci M, Sahin İ, Gungor O, Gurel A, Ogeturk M, Dabak O. Cardiac, skeletal muscle and serum irisin responses to with or without water exercise in young and old male rats: cardiac muscle produces more irisin than skeletal muscle. Peptides. 2014;52:68–73.
Gouveia MC, Vella JP, Cafeo FR, Affonso Fonseca FL, Bacci MR. Association between irisin and major chronic diseases: a review. Eur Rev Med Pharmacol Sci. 2016;20:4072–7.
Bashar SM, Samir El-Sherbeiny SM, Boraie MZ. Correlation between the blood level of irisin and the severity of acute myocardial infarction in exercise-trained rats. J Basic Clin Physiol Pharmacol. 2018;30:59–71.
Anastasilakis AD, Koulaxis D, Kefala N, Polyzos SA, Upadhyay J, Pagkalidou E, Economou F, Anastasilakis CD, Mantzoros CS. Circulating irisin levels are lower in patients with either stable coronary artery disease (CAD) or myocardial infarction (MI) versus healthy controls, whereas follistatin and activin A levels are higher and can discriminate MI from CAD with similar to CK-MB accuracy. Metab Clin Exp. 2017;73:1–8.
Abd El-Mottaleb NA, Galal HM, El Maghraby KM, Gadallah AI. Serum irisin level in myocardial infarction patients with or without heart failure. Can J Physiol Pharmacol. 2019;97:932–8.
Shen S, Gao R, Bei Y, Li J, Zhang H, Zhou Y, Yao W, Xu D, Zhou F, Jin M, Wei S, Wang K, Xu X, Li Y, Xiao J, Li X. Serum irisin predicts mortality risk in acute heart failure patients. Cell Physiol Biochem. 2017;42:615–22.
Heidari B, Al-Hijji MA, Moynagh MR, Takahashi N, Welle G, Eleid M, Singh M, Gulati R, Rihal CS, Lerman A. Transcatheter aortic valve replacement outcomes in patients with sarcopaenia. EuroIntervention. 2019;15:671–7.
Chang JS, Kim TH, Nguyen TT, Park KS, Kim N, Kong ID. Circulating irisin levels as a predictive biomarker for sarcopenia: a cross-sectional community-based study. Geriatr Gerontol Int. 2017;17:2266–73.
Puls M, Sobisiak B, Bleckmann A, Jacobshagen C, Danner BC, Hünlich M, Beißbarth T, Schöndube F, Hasenfuß G, Seipelt R, Schillinger W. Impact of frailty on short- and long-term morbidity and mortality after transcatheter aortic valve implantation: risk assessment by Katz Index of activities of daily living. EuroIntervention. 2014;10:609–19.
John D, Buellesfeld L, Yuecel S, Mueller R, Latsios G, Beucher H, Gerckens U, Grube E. Correlation of device landing zone calcification and acute procedural success in patients undergoing transcatheter aortic valve implantations with the self-expanding CoreValve prosthesis. JACC Cardiovasc Interv. 2010;3:233–43.
Kuloglu T, Aydin S, Eren MN, Yilmaz M, Sahin I, Kalayci M, Sarman E, Kaya N, Yilmaz OF, Turk A, Aydin Y, Yalcin MH, Uras N, Gurel A, Ilhan S, Gul E, Aydin S. Irisin: a potentially candidate marker for myocardial infarction. Peptides. 2014;55:85–91.
Yu Q, Kou W, Xu X, Zhou S, Luan P, Xu X, Li H, Zhuang J, Wang J, Zhao Y, Xu Y, Peng W. FNDC5/Irisin inhibits pathological cardiac hypertrophy. Clin Sci (London England: 1979). 2019;133:611–27.
Chahine J, Kadri AN, Gajulapalli RD, Krishnaswamy A, Mick S, Perez O, Lak H, Nair RM, Montane B, Tak J, Tuzcu EM, Griffin B, Svensson LG, Harb SC, Kapadia SR. Outcomes of transcatheter aortic valve replacement in mixed aortic valve disease. JACC Cardiovasc Interv. 2019;12:2299–306.
Patel HJ, Likosky DS, Pruitt AL, Murphy ET, Theurer PF, Prager RL. Aortic valve replacement in the moderately elevated risk patient: a population-based analysis of outcomes. Ann Thorac Surg. 2016;102:1466–72.
Heidari B, Al-Hijji MA, Alkhouli MA, Egbe A, Welle G, Eleid MF, Singh M, Gulati R, Rihal C, Lerman A. Transcatheter aortic valve replacement outcomes in mixed aortic valve disease compared to predominant aortic stenosis. Int J Cardiol. 2020;299:209–14.
Kapadia S, Agarwal S, Miller DC, Webb JG, Mack M, Ellis S, Herrmann HC, Pichard AD, Tuzcu EM, Svensson LG, Smith CR, Rajeswaran J, Ehrlinger J, Kodali S, Makkar R, Thourani VH, Blackstone EH, Leon MB. Insights into timing, risk factors, and outcomes of stroke and transient ischemic attack after transcatheter aortic valve replacement in the PARTNER Trial (Placement of Aortic Transcatheter Valves). Circ Cardiovasc Interv. 2016;9(9):e002981.
Abdelghani M, Cavalcante R, Miyazaki Y, de Winter RJ, Tijssen JG, Sarmento-Leite R, Mangione JA, Abizaid A, Lemos PA, Serruys PW, de Brito FS Jr. Transcatheter aortic valve implantation for mixed versus pure stenotic aortic valve disease. EuroIntervention. 2017;13:1157–65.
Thaden JJ, Nkomo VT, Enriquez-Sarano M. The global burden of aortic stenosis. Prog Cardiovasc Dis. 2014;56:565–71.
Carrai P, Camarri S, Pondrelli CR, Gonnelli S, Caffarelli C. Calcification of cardiac valves in metabolic bone disease: an updated review of clinical studies. Clin Interv Aging. 2020;15:1085–95.
Qiao X, Nie Y, Ma Y, Chen Y, Cheng R, Yin W, Hu Y, Xu W, Xu L. Irisin promotes osteoblast proliferation and differentiation via activating the MAP kinase signaling pathways. Sci Rep. 2016;6:18732.
He L, He WY, LT A, Yang WL, Zhang AH. Lower serum irisin levels are associated with increased vascular calcification in hemodialysis patients. Kidney Blood Press Res. 2018;43:287–95.
Khurrami L, Møller JE, Lindholt JS, Dahl JS, Fredgart MH, Obel LM, Steffensen FH, Urbonaviciene G, Lambrechtsen J, Diederichsen ACP. Aortic valve calcification among elderly males from the general population, associated echocardiographic findings, and clinical implications. Eur Heart J Cardiovasc Imaging. 2022;23:177–84.
Stewart BF, Siscovick D, Lind BK, Gardin JM, Gottdiener JS, Smith VE, Kitzman DW, Otto CM. Clinical factors associated with calcific aortic valve disease Cardiovascular Health Study. J Am College Cardiol. 1997;29:630–4.
Biniaminov N, Bandt S, Roth A, Haertel S, Neumann R, Bub A. Irisin, physical activity and fitness status in healthy humans: no association under resting conditions in a cross-sectional study. PLoS ONE. 2018;13:e0189254.
Zügel M, Qiu S, Laszlo R, Bosnyák E, Weigt C, Müller D, Diel P, Steinacker JM, Schumann U. The role of sex, adiposity, and gonadectomy in the regulation of irisin secretion. Endocrine. 2016;54:101–10.
Funding
This study was funded by the Zhejiang Province Science and Technology Department Key R&D Program (2021C03097) and by the Natural Science Foundation of Zhejiang Province (LQ21H020005).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics Approval
All the procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1975, as revised in 2000 (5). Informed consent was obtained from all the patients for being included in the study.
Additional information
Associate Editor Craig M. Stolen oversaw the review of this article
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Wang, Ss., Li, Jm., Hu, P. et al. Circulating Irisin Level as a Biomarker for Pure Aortic Stenosis and Aortic Valve Calcification. J. of Cardiovasc. Trans. Res. 16, 443–452 (2023). https://doi.org/10.1007/s12265-022-10327-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12265-022-10327-9