Abstract
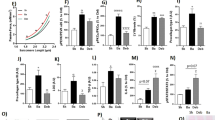
The development of left ventricular hypertrophy and dysfunction in aortic regurgitation (AR) has only been sparsely studied experimentally. In a new model of chronic AR in rats, we examined activation of molecular pathways involved in myocardial hypertrophy. Chronic AR was produced by damaging one or two valve cusps, resulting in eccentric remodeling and left ventricular dysfunction, with no increase in overall fibrosis. Western blotting showed increased activation of Akt and p38 at 12 weeks and of c-Jun amino-terminal kinase at 2 weeks, decreased activation of extracellular regulated kinase 5 at both 2 and 12 weeks, while activation of calcium/calmodulin-dependent protein kinase II and extracellular regulated kinase 1/2 was unchanged. Expression of calcineurin and ANF was also unchanged. Eccentric hypertrophy and early cardiac dysfunction in experimental AR are associated with a pattern of activation of intracellular pathways different from that seen with pathological hypertrophy in pressure overload, and more similar to that associated with benign physiological hypertrophy.






Similar content being viewed by others
References
Mudd, J. O., & Kass, D. A. (2008). Tackling heart failure in the twenty-first century. Nature, 451(7181), 919–928.
Bernardo, B. C., Weeks, K. L., Pretorius, L., & McMullen, J. R. (2010). Molecular distinction between physiological and pathological cardiac hypertrophy: experimental findings and therapeutic strategies. Pharmacology & Therapeutics, 128(1), 191–227.
Plante, E., Gaudreau, M., Lachance, D., Drolet, M. C., Roussel, E., Gauthier, C., et al. (2004). Angiotensin-converting enzyme inhibitor captopril prevents volume overload cardiomyopathy in experimental chronic aortic valve regurgitation. Canadian Journal of Physiology and Pharmacology, 82(3), 191–199.
Plante, E., Lachance, D., Gaudreau, M., Drolet, M. C., Roussel, E., Arsenault, M., et al. (2004). Effectiveness of beta-blockade in experimental chronic aortic regurgitation. Circulation, 110(11), 1477–1483.
Evangelista, A., Tornos, P., Sambola, A., Permanyer-Miralda, G., & Soler-Soler, J. (2005). Long-term vasodilator therapy in patients with severe aortic regurgitation. The New England Journal of Medicine, 353(13), 1342–1349.
Miyamoto, T., Takeishi, Y., Takahashi, H., Shishido, T., Arimoto, T., Tomoike, H., et al. (2004). Activation of distinct signal transduction pathways in hypertrophied hearts by pressure and volume overload. Basic Research in Cardiology, 99(5), 328–337.
Toischer, K., Rokita, A. G., Unsold, B., Zhu, W., Kararigas, G., Sossalla, S., et al. (2010). Differential cardiac remodeling in preload versus afterload. Circulation, 122(10), 993–1003.
Bekeredjian, R., & Grayburn, P. A. (2005). Valvular heart disease: aortic regurgitation. Circulation, 112(1), 125–134.
Arsenault, M., Plante, E., Drolet, M. C., & Couet, J. (2002). Experimental aortic regurgitation in rats under echocardiographic guidance. The Journal of Heart Valve Disease, 11(1), 128–134.
Yin, F. C. (1981). Ventricular wall stress. Circulation Research, 49(4), 829–842.
Sullivan, L. M. (2008). Repeated measures. Circulation, 117(9), 1238–1243.
Abassi, Z., Goltsman, I., Karram, T., Winaver, J., & Hoffman, A. (2011). Aortocaval fistula in rat: a unique model of volume-overload congestive heart failure and cardiac hypertrophy. Journal of Biomedicine and Biotechnology, 2011, 729497.
Uematsu, T., Yamazaki, T., Matsuno, H., Hayashi, Y., & Nakashima, M. (1989). A simple method for producing graded aortic insufficiencies in rats and subsequent development of cardiac hypertrophy. Journal of Pharmacological Methods, 22(4), 249–257.
Magid, N. M., Opio, G., Wallerson, D. C., Young, M. S., & Borer, J. S. (1994). Heart failure due to chronic experimental aortic regurgitation. The American Journal of Physiology, 267(2 Pt 2), H556–H562.
Borer, J. S., Truter, S., Herrold, E. M., Falcone, D. J., Pena, M., Carter, J. N., et al. (2002). Myocardial fibrosis in chronic aortic regurgitation: molecular and cellular responses to volume overload. Circulation, 105(15), 1837–1842.
Truter, S. L., Catanzaro, D. F., Supino, P. G., Gupta, A., Carter, J., Ene, A. R., et al. (2009). Fibronectin gene expression in aortic regurgitation: relative roles of mitogen-activated protein kinases. Cardiology, 113(4), 291–298.
Truter, S. L., Catanzaro, D. F., Supino, P. G., Gupta, A., Carter, J., Herrold, E. M., et al. (2009). Differential expression of matrix metalloproteinases and tissue inhibitors and extracellular matrix remodeling in aortic regurgitant hearts. Cardiology, 113(3), 161–168.
Magid, N. M., Wallerson, D. C., Borer, J. S., Mukherjee, A., Young, M. S., Devereux, R. B., et al. (1992). Left ventricular diastolic and systolic performance during chronic experimental aortic regurgitation. The American Journal of Physiology, 263(1 Pt 2), H226–H233.
Florenzano, F., & Glantz, S. A. (1987). Left ventricular mechanical adaptation to chronic aortic regurgitation in intact dogs. The American Journal of Physiology, 252(5 Pt 2), H969–H984.
Gaynor, J. W., Feneley, M. P., Gall, S. A., Jr., Savitt, M. A., Silvestry, S. C., Davis, J. W., et al. (1997). Left ventricular adaptation to aortic regurgitation in conscious dogs. The Journal of Thoracic and Cardiovascular Surgery, 113(1), 149–158.
Olsen, N. T., Sogaard, P., Larsson, H. B., Goetze, J. P., Jons, C., Mogelvang, R., et al. (2011). Speckle-tracking echocardiography for predicting outcome in chronic aortic regurgitation during conservative management and after surgery. JACC Cardiovascular Imaging, 4(3), 223–230.
Shioi, T., McMullen, J. R., Kang, P. M., Douglas, P. S., Obata, T., Franke, T. F., et al. (2002). Akt/protein kinase B promotes organ growth in transgenic mice. Molecular and Cellular Biology, 22(8), 2799–2809.
McMullen, J. R., Shioi, T., Huang, W. Y., Zhang, L., Tarnavski, O., Bisping, E., et al. (2004). The insulin-like growth factor 1 receptor induces physiological heart growth via the phosphoinositide 3-kinase(p110alpha) pathway. The Journal of Biological Chemistry, 279(6), 4782–4793.
McMullen, J. R., Shioi, T., Zhang, L., Tarnavski, O., Sherwood, M. C., Kang, P. M., et al. (2003). Phosphoinositide 3-kinase(p110alpha) plays a critical role for the induction of physiological, but not pathological, cardiac hypertrophy. Proceedings of the National Academy of Sciences of the United States of America, 100(21), 12355–12360.
DeBosch, B., Treskov, I., Lupu, T. S., Weinheimer, C., Kovacs, A., Courtois, M., et al. (2006). Akt1 is required for physiological cardiac growth. Circulation, 113(17), 2097–2104.
McMullen, J. R., Amirahmadi, F., Woodcock, E. A., Schinke-Braun, M., Bouwman, R. D., Hewitt, K. A., et al. (2007). Protective effects of exercise and phosphoinositide 3-kinase(p110alpha) signaling in dilated and hypertrophic cardiomyopathy. Proceedings of the National Academy of Sciences of the United States of America, 104(2), 612–617.
Bouchard-Thomassin, A. A., Lachance, D., Drolet, M. C., Couet, J., & Arsenault, M. (2011). A high-fructose diet worsens eccentric left ventricular hypertrophy in experimental volume overload. American Journal of Physiology. Heart and Circulatory Physiology, 300(1), H125–H134.
Shimoyama, M., Hayashi, D., Takimoto, E., Zou, Y., Oka, T., Uozumi, H., et al. (1999). Calcineurin plays a critical role in pressure overload-induced cardiac hypertrophy. Circulation, 100(24), 2449–2454.
Lim, H. W., De Windt, L. J., Steinberg, L., Taigen, T., Witt, S. A., Kimball, T. R., et al. (2000). Calcineurin expression, activation, and function in cardiac pressure-overload hypertrophy. Circulation, 101(20), 2431–2437.
Braun, M. U., LaRosée, P., Simonis, G., Borst, M. M., & Strasser, R. H. (2004). Regulation of protein kinase C isozymes in volume overload cardiac hypertrophy. Molecular and Cellular Biochemistry, 262(1), 135–143.
Zhang, R., Khoo, M. S., Wu, Y., Yang, Y., Grueter, C. E., Ni, G., et al. (2005). Calmodulin kinase II inhibition protects against structural heart disease. Nature Medicine, 11(4), 409–417.
Backs, J., Backs, T., Neef, S., Kreusser, M. M., Lehmann, L. H., Patrick, D. M., et al. (2009). The delta isoform of CaM kinase II is required for pathological cardiac hypertrophy and remodeling after pressure overload. Proceedings of the National Academy of Sciences of the United States of America, 106(7), 2342–2347.
Rose, B. A., Force, T., & Wang, Y. (2010). Mitogen-activated protein kinase signaling in the heart: angels versus demons in a heart-breaking tale. Physiological Reviews, 90(4), 1507–1546.
Nicol, R. L., Frey, N., Pearson, G., Cobb, M., Richardson, J., & Olson, E. N. (2001). Activated MEK5 induces serial assembly of sarcomeres and eccentric cardiac hypertrophy. The EMBO Journal, 20(11), 2757–2767.
Lachance, D., Plante, E., Roussel, E., Drolet, M. C., Couet, J., & Arsenault, M. (2008). Early left ventricular remodeling in acute severe aortic regurgitation: insights from an animal model. The Journal of Heart Valve Disease, 17(3), 300–308.
Kehat, I., Davis, J., Tiburcy, M., Accornero, F., Saba-El-Leil, M. K., Maillet, M., et al. (2011). Extracellular signal-regulated kinases 1 and 2 regulate the balance between eccentric and concentric cardiac growth. Circulation Research, 108(2), 176–183.
Acknowledgments
This work was supported in part by a National Institute of Health grant to Dr. Abraham (AG 022554). Dr. Olsen was supported by a grant from the Danish Heart Foundation, Copenhagen, Denmark.
Conflict of Interest
The authors have no conflict of interest to declare.
Author information
Authors and Affiliations
Corresponding author
Additional information
Associate Editor Angela Taylor oversaw the review of this article
Rights and permissions
About this article
Cite this article
Olsen, N.T., Dimaano, V.L., Fritz-Hansen, T. et al. Hypertrophy Signaling Pathways in Experimental Chronic Aortic Regurgitation. J. of Cardiovasc. Trans. Res. 6, 852–860 (2013). https://doi.org/10.1007/s12265-013-9503-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12265-013-9503-y