Abstract
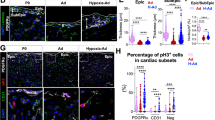
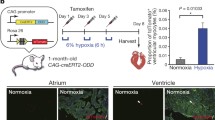
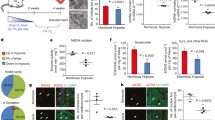
Recent reports indicate that the adult mammalian heart is capable of limited, but measurable, cardiomyocyte turnover. While the lineage origin of the newly formed cardiomyocytes is not entirely understood, mounting evidence suggest that the epicardium and subepicardium may represent an important source of cardiac stem or progenitor cells. Stem cell niches are characterized by low oxygen tension, where stem cells preferentially utilize cytoplasmic glycolysis to meet their energy demands. However, it is unclear if the heart harbors similar hypoxic regions, or whether these regions house metabolically distinct cardiac progenitor populations. Here we identify the epicardium and subepicardium as the cardiac hypoxic niche-based capillary density quantification, and localization of Hif-1α in the uninjured heart. We further demonstrate that this hypoxic microenvironment houses a metabolically distinct population of glycolytic progenitor cells. Finally, we show that Hif-1α regulates the glycolytic phenotype and progenitor properties of these cells. These findings highlight important anatomical and functional properties of the epicardial and subepicardial microenvironment, and the potential role of hypoxia signaling in regulation of cardiac progenitors.




Similar content being viewed by others
References
Laflamme, M. A., & Murry, C. E. (2011). Heart regeneration. Nature, 473, 326–335.
Segers, V. F., & Lee, R. T. (2008). Stem-cell therapy for cardiac disease. Nature, 451, 937–942.
Bergmann, O., Bhardwaj, R. D., Bernard, S., Zdunek, S., Barnabe-Heider, F., Walsh, S., Zupicich, J., Alkass, K., Buchholz, B. A., Druid, H., Jovinge, S., & Frisen, J. (2009). Evidence for cardiomyocyte renewal in humans. Science (New York, N.Y.), 324, 98–102.
Laflamme, M. A., Myerson, D., Saffitz, J. E., & Murry, C. E. (2002). Evidence for cardiomyocyte repopulation by extracardiac progenitors in transplanted human hearts. Circulation Research, 90, 634–640.
Loffredo, F. S., Steinhauser, M. L., Gannon, J., & Lee, R. T. (2011). Bone marrow-derived cell therapy stimulates endogenous cardiomyocyte progenitors and promotes cardiac repair. Cell Stem Cell, 8, 389–398.
Oh, H., Bradfute, S. B., Gallardo, T. D., Nakamura, T., Gaussin, V., Mishina, Y., Pocius, J., Michael, L. H., Behringer, R. R., Garry, D. J., Entman, M. L., & Schneider, M. D. (2003). Cardiac progenitor cells from adult myocardium: Homing, differentiation, and fusion after infarction. Proceedings of the National Academy of Sciences of the United States of America, 100, 12313–12318.
Matsuura, K., Nagai, T., Nishigaki, N., Oyama, T., Nishi, J., Wada, H., Sano, M., Toko, H., Akazawa, H., Sato, T., Nakaya, H., Kasanuki, H., & Komuro, I. (2004). Adult cardiac sca-1-positive cells differentiate into beating cardiomyocytes. Journal of Biological Chemistry, 279, 11384–11391.
Barile, L., Messina, E., Giacomello, A., & Marban, E. (2007). Endogenous cardiac stem cells. Progress in Cardiovascular Diseases, 50, 31–48.
Passier, R., van Laake, L. W., & Mummery, C. L. (2008). Stem-cell-based therapy and lessons from the heart. Nature, 453, 322–329.
Slukvin, I. I. (2011). Epicardial origin of cardiac CFU-Fs. Cell Stem Cell, 9, 492–493.
Chong, J. J., Chandrakanthan, V., Xaymardan, M., Asli, N. S., Li, J., Ahmed, I., Heffernan, C., Menon, M. K., Scarlett, C. J., Rashidianfar, A., Biben, C., Zoellner, H., Colvin, E. K., Pimanda, J. E., Biankin, A. V., Zhou, B., Pu, W. T., Prall, O. W., & Harvey, R. P. (2011). Adult cardiac-resident MSC-like stem cells with a proepicardial origin. Cell Stem Cell, 9, 527–540.
Lepilina, A., Coon, A. N., Kikuchi, K., Holdway, J. E., Roberts, R. W., Burns, C. G., & Poss, K. D. (2006). A dynamic epicardial injury response supports progenitor cell activity during zebrafish heart regeneration. Cell, 127, 607–619.
Kikuchi, K., Holdway, J. E., Werdich, A. A., Anderson, R. M., Fang, Y., Egnaczyk, G. F., Evans, T., Macrae, C. A., Stainier, D. Y., & Poss, K. D. (2010). Primary contribution to zebrafish heart regeneration by gata4(+) cardiomyocytes. Nature, 464, 601–605.
Zhou, B., Honor, L. B., He, H., Ma, Q., Oh, J. H., Butterfield, C., Lin, R. Z., Melero-Martin, J. M., Dolmatova, E., Duffy, H. S., Gise, A., Zhou, P., Hu, Y. W., Wang, G., Zhang, B., Wang, L., Hall, J. L., Moses, M. A., McGowan, F. X., & Pu, W. T. (2011). Adult mouse epicardium modulates myocardial injury by secreting paracrine factors. The Journal of Clinical Investigation, 121, 1894–1904.
Smart, N., Bollini, S., Dube, K. N., Vieira, J. M., Zhou, B., Davidson, S., Yellon, D., Riegler, J., Price, A. N., Lythgoe, M. F., Pu, W. T., & Riley, P. R. (2011). De novo cardiomyocytes from within the activated adult heart after injury. Nature, 474, 640–644.
Urbanek, K., Cesselli, D., Rota, M., Nascimbene, A., De Angelis, A., Hosoda, T., Bearzi, C., Boni, A., Bolli, R., Kajstura, J., Anversa, P., & Leri, A. (2006). Stem cell niches in the adult mouse heart. Proceedings of the National Academy of Sciences of the United States of America, 103, 9226–9231.
Walker, M. R., Patel, K. K., & Stappenbeck, T. S. (2009). The stem cell niche. The Journal of Pathology, 217, 169–180.
Popescu, L. M., Gherghiceanu, M., Manole, C. G., & Faussone-Pellegrini, M. S. (2009). Cardiac renewing: Interstitial Cajal-like cells nurse cardiomyocyte progenitors in epicardial stem cell niches. Journal of Cellular and Molecular Medicine, 13, 866–886.
Urbanek, K., Cesselli, D., Rota, M., Nascimbene, A., De Angelis, A., Hosoda, T., Bearzi, C., Boni, A., Bolli, R., Kajstura, J., Anversa, P., & Leri, A. (2006). Stem cell niches in the adult mouse heart. Proceedings of the National Academy of Sciences of the United States of America, 103, 9226–9231.
Morrison, S. J., & Spradling, A. C. (2008). Stem cells and niches: Mechanisms that promote stem cell maintenance throughout life. Cell, 132, 598–611.
Nakada, D., Levi, B. P., & Morrison, S. J. (2011). Integrating physiological regulation with stem cell and tissue homeostasis. Neuron, 70, 703–718.
Parmar, K., Mauch, P., Vergilio, J. A., Sackstein, R., & Down, J. D. (2007). Distribution of hematopoietic stem cells in the bone marrow according to regional hypoxia. Proceedings of the National Academy of Sciences of the United States of America, 104, 5431–5436.
Mohyeldin, A., Garzon-Muvdi, T., & Quinones-Hinojosa, A. (2010). Oxygen in stem cell biology: A critical component of the stem cell niche. Cell Stem Cell, 7, 150–161.
Simsek, T., Kocabas, F., Zheng, J., Deberardinis, R. J., Mahmoud, A. I., Olson, E. N., et al. (2010). The distinct metabolic profile of hematopoietic stem cells reflects their location in a hypoxic niche. Cell Stem Cell, 7, 380–390.
Cai, C. L., Martin, J. C., Sun, Y., Cui, L., Wang, L., Ouyang, K., Yang, L., Bu, L., Liang, X., Zhang, X., Stallcup, W. B., Denton, C. P., McCulloch, A., Chen, J., & Evans, S. M. (2008). A myocardial lineage derives from Tbx18 epicardial cells. Nature, 454, 104–108.
Smart, N., Risebro, C. A., Melville, A. A., Moses, K., Schwartz, R. J., Chien, K. R., & Riley, P. R. (2007). Thymosin beta4 induces adult epicardial progenitor mobilization and neovascularization. Nature, 445, 177–182.
Olivey, H. E., & Svensson, E. C. (2010). Epicardial-myocardial signaling directing coronary vasculogenesis. Circulation Research, 106, 818–832.
Limana, F., Zacheo, A., Mocini, D., Mangoni, A., Borsellino, G., Diamantini, A., De Mori, R., Battistini, L., Vigna, E., Santini, M., Loiaconi, V., Pompilio, G., Germani, A., & Capogrossi, M. C. (2007). Identification of myocardial and vascular precursor cells in human and mouse epicardium. Circulation Research, 101, 1255–1265.
Zhou, B., Honor, L. B., He, H., Ma, Q., Oh, J. H., Butterfield, C., Lin, R. Z., Melero-Martin, J. M., Dolmatova, E., Duffy, H. S., Gise, A., Zhou, P., Hu, Y. W., Wang, G., Zhang, B., Wang, L., Hall, J. L., Moses, M. A., McGowan, F. X., & Pu, W. T. (2011). Adult mouse epicardium modulates myocardial injury by secreting paracrine factors. The Journal of Clinical Investigation, 121, 1894–1904.
Mikawa, T., & Gourdie, R. G. (1996). Pericardial mesoderm generates a population of coronary smooth muscle cells migrating into the heart along with ingrowth of the epicardial organ. Developmental Biology, 174, 221–232.
Dettman, R. W., Denetclaw, W., Jr., Ordahl, C. P., & Bristow, J. (1998). Common epicardial origin of coronary vascular smooth muscle, perivascular fibroblasts, and intermyocardial fibroblasts in the avian heart. Developmental Biology, 193, 169–181.
Reimer, K. A., Lowe, J. E., Rasmussen, M. M., & Jennings, R. B. (1977). The wavefront phenomenon of ischemic cell death. 1. Myocardial infarct size vs duration of coronary occlusion in dogs. Circulation, 56, 786–794.
Semenza, G. L. (2007). Hypoxia-inducible factor 1 (HIF-1) pathway. Science's STKE, 2007, cm8.
Qi, J., Nakayama, K., Gaitonde, S., Goydos, J. S., Krajewski, S., Eroshkin, A., Bar-Sagi, D., Bowtell, D., & Ronai, Z. (2008). The ubiquitin ligase Siah2 regulates tumorigenesis and metastasis by HIF-dependent and -independent pathways. Proceedings of the National Academy of Sciences of the United States of America, 105, 16713–16718.
Nakayama, K., Frew, I. J., Hagensen, M., Skals, M., Habelhah, H., Bhoumik, A., Kadoya, T., Erdjument-Bromage, H., Tempst, P., Frappell, P. B., Bowtell, D. D., & Ronai, Z. (2004). Siah2 regulates stability of prolyl-hydroxylases, controls HIF1alpha abundance, and modulates physiological responses to hypoxia. Cell, 117, 941–952.
Li, Z., Wang, D., Messing, E. M., & Wu, G. (2005). VHL protein-interacting deubiquitinating enzyme 2 deubiquitinates and stabilizes HIF-1alpha. EMBO Reports, 6, 373–378.
Maxwell, P. H., Wiesener, M. S., Chang, G. W., Clifford, S. C., Vaux, E. C., Cockman, M. E., Wykoff, C. C., Pugh, C. W., Maher, E. R., & Ratcliffe, P. J. (1999). The tumour suppressor protein VHL targets hypoxia-inducible factors for oxygen-dependent proteolysis. Nature, 399, 271–275.
Kamura, T., Sato, S., Iwai, K., Czyzyk-Krzeska, M., Conaway, R. C., & Conaway, J. W. (2000). Activation of HIF1alpha ubiquitination by a reconstituted von Hippel-Lindau (VHL) tumor suppressor complex. Proceedings of the National Academy of Sciences of the United States of America, 97, 10430–10435.
Hagg, M., & Wennstrom, S. (2005). Activation of hypoxia-induced transcription in normoxia. Experimental Cell Research, 306, 180–191.
Maxwell, P. J., Gallagher, R., Seaton, A., Wilson, C., Scullin, P., Pettigrew, J., Stratford, I. J., Williams, K. J., Johnston, P. G., & Waugh, D. J. (2007). HIF-1 and NF-kappaB-mediated upregulation of CXCR1 and CXCR2 expression promotes cell survival in hypoxic prostate cancer cells. Oncogene, 26, 7333–7345.
Smith, R. R., Barile, L., Cho, H. C., Leppo, M. K., Hare, J. M., Messina, E., Giacomello, A., Abraham, M. R., & Marban, E. (2007). Regenerative potential of cardiosphere-derived cells expanded from percutaneous endomyocardial biopsy specimens. Circulation, 115, 896–908.
Zhou, B., Ma, Q., Rajagopal, S., Wu, S. M., Domian, I., Rivera-Feliciano, J., Jiang, D., von Gise, A., Ikeda, S., Chien, K. R., & Pu, W. T. (2008). Epicardial progenitors contribute to the cardiomyocyte lineage in the developing heart. Nature, 454, 109–113.
Beltrami, A. P., Barlucchi, L., Torella, D., Baker, M., Limana, F., Chimenti, S., Kasahara, H., Rota, M., Musso, E., Urbanek, K., Leri, A., Kajstura, J., Nadal-Ginard, B., & Anversa, P. (2003). Adult cardiac stem cells are multipotent and support myocardial regeneration. Cell, 114, 763–776.
Laugwitz, K. L., Moretti, A., Lam, J., Gruber, P., Chen, Y., Woodard, S., Lin, L. Z., Cai, C. L., Lu, M. M., Reth, M., Platoshyn, O., Yuan, J. X., Evans, S., & Chien, K. R. (2005). Postnatal isl1+ cardioblasts enter fully differentiated cardiomyocyte lineages. Nature, 433, 647–653.
Martin-Puig, S., Wang, Z., & Chien, K. R. (2008). Lives of a heart cell: Tracing the origins of cardiac progenitors. Cell Stem Cell, 2, 320–331.
Martin, C. M., Ferdous, A., Gallardo, T., Humphries, C., Sadek, H., Caprioli, A., Garcia, J. A., Szweda, L. I., Garry, M. G., & Garry, D. J. (2008). Hypoxia-inducible factor-2alpha transactivates Abcg2 and promotes cytoprotection in cardiac side population cells. Circulation Research, 102, 1075–1081.
Martin, C. M., Meeson, A. P., Robertson, S. M., Hawke, T. J., Richardson, J. A., Bates, S., Goetsch, S. C., Gallardo, T. D., & Garry, D. J. (2004). Persistent expression of the ATP-binding cassette transporter, Abcg2, identifies cardiac SP cells in the developing and adult heart. Developmental Biology, 265, 262–275.
Zhang, C. C., & Lodish, H. F. (2005). Murine hematopoietic stem cells change their surface phenotype during ex vivo expansion. Blood, 105, 4314–4320.
O'Connell, T. D., Rodrigo, M. C., & Simpson, P. C. (2007). Isolation and culture of adult mouse cardiac myocytes. Methods in Molecular Biology, 357, 271–296.
Arminan, A., Gandia, C., Garcia-Verdugo, J. M., Lledo, E., Mullor, J. L., Montero, J. A., & Sepulveda, P. (2010). Cardiac transcription factors driven lineage-specification of adult stem cells. Journal of Cardiovascular Translational Research, 3, 61–65.
Qian, Q., Qian, H., Zhang, X., Zhu, W., Yan, Y., Ye, S., et al. (2012). 5-Azacytidine induces cardiac differentiation of human umbilical cord-derived mesenchymal stem cells by activating extracellular regulated kinase. Stem Cells and Development, 21, 67–75.
Acknowledgments
The authors would like to acknowledge Drs. Eric Olson and James Richardson for their valuable input and John Shelton for his assistance with histology. Work in Hesham Sadek's laboratory is supported by grants from the American Heart Association (Fellow to Faculty Award) and the Gilead Sciences Research Scholars Program in Cardiovascular Disease.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Figure S1
Endomucin staining. Adult heart sections were stained with the endothelial marker endomucin and detected using DAB chromogen. Images of four-chamber view adult heart sections were quantified for capillaries for cell layers at five different zones in the coronal plane (base to apex) and seven different zones in the horizontal plane (endocardium to epicardium). (JPEG 33 kb)
Figure S2
Quantification of Hif-1α expression in ventricular epicardium and subepicardium. The number of Hif-1α-expressing cells in the epicardium and subepicardium was quantified. The majority of ventricular epicardial cells expressed Hif-1α, while only a small percentage of subepicardial and myocardial myocytes and non-myocytes expressed Hif-1α. (JPEG 34 kb)
Figure S3
Atrial Hif-1α expression. The majority of atrial cells express Hif-1α. Hif-1α (green), Tnnt2 (red), and DAPI (blue). (JPEG 88 kb)
Rights and permissions
About this article
Cite this article
Kocabas, F., Mahmoud, A.I., Sosic, D. et al. The Hypoxic Epicardial and Subepicardial Microenvironment. J. of Cardiovasc. Trans. Res. 5, 654–665 (2012). https://doi.org/10.1007/s12265-012-9366-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12265-012-9366-7